Evaluating AmpliSeq Childhood Cancer Panel Performance: A Deep Dive into Mean Read Depth and Key Sequencing Metrics
This article provides a comprehensive technical evaluation of the AmpliSeq for Illumina Childhood Cancer Panel, a targeted NGS solution for pediatric and young adult cancers.
Evaluating AmpliSeq Childhood Cancer Panel Performance: A Deep Dive into Mean Read Depth and Key Sequencing Metrics
Abstract
This article provides a comprehensive technical evaluation of the AmpliSeq for Illumina Childhood Cancer Panel, a targeted NGS solution for pediatric and young adult cancers. Tailored for researchers and drug development professionals, it explores foundational concepts of sequencing depth and coverage, details the panel's methodology and validation data, offers troubleshooting guidance for optimization, and presents comparative analyses with other NGS approaches. The focus is on critical performance metrics, particularly a mean read depth greater than 1000x, and their direct impact on assay sensitivity, specificity, and ultimately, clinical utility in refining diagnosis and enabling precision medicine for acute leukemia and other pediatric cancers.
Understanding NGS Performance Metrics: Why Mean Read Depth is Critical for Pediatric Cancer Profiling
In the realm of next-generation sequencing (NGS), the success of a study, particularly in sensitive applications like pediatric cancer genomics, hinges on a clear understanding of two fundamental metrics: sequencing depth and coverage. While often used interchangeably, these terms describe distinct concepts that collectively determine the quality, reliability, and completeness of genomic data [1] [2]. For researchers using targeted panels like the AmpliSeq for Illumina Childhood Cancer Panel, grasping this distinction is not merely academic; it is a practical necessity for designing robust experiments, accurately interpreting variant calls, and ultimately, making confident clinical decisions [3]. This guide provides a detailed comparison of these pivotal metrics, framed within the context of optimizing performance for childhood cancer research.
Understanding the Core Concepts: Depth and Coverage
Sequencing Depth
Sequencing depth, also called read depth, refers to the average number of times a specific nucleotide in the genome is sequenced [1] [2]. It is a measure of data redundancy and confidence at a given base position.
- Definition: The number of times a particular nucleotide is read during the sequencing process [1].
- Calculation: It is calculated theoretically using the Lander/Waterman equation:
C = (L * N) / G, whereCis coverage,Lis read length,Nis the number of reads, andGis the haploid genome length [4] [5]. - Purpose: A higher depth directly increases confidence in base calling, which is crucial for detecting rare variants or sequencing heterogeneous samples like tumor tissues [1] [6].
Sequencing Coverage
Sequencing coverage (or breadth of coverage) describes the proportion of the target genome or region that has been sequenced at least once [1] [2]. It is a measure of how comprehensively the area of interest has been sampled.
- Definition: The percentage of the genome or targeted region that has been sequenced [1].
- Purpose: It ensures that the entirety, or as much as possible, of the target region—whether the whole genome, exome, or a specific panel—has been sequenced, thereby minimizing gaps in the data [1].
The following diagram illustrates the logical relationship between these two concepts in an NGS experiment:
Key Differences at a Glance
The following table summarizes the fundamental distinctions between sequencing depth and coverage.
| Aspect | Sequencing Depth | Sequencing Coverage |
|---|---|---|
| Definition | Average number of times a nucleotide is read [1] [2]. | Proportion of the target genome sequenced at least once [1] [2]. |
| Key Focus | Accuracy and confidence at specific base positions [2]. | Completeness of genomic representation [2]. |
| Primary Metric | Numerical multiple (e.g., 30x, 100x, 1000x) [1]. | Percentage (e.g., 95% coverage) [1]. |
| Role in Variant Calling | Critical for confidence in calling variants, especially rare ones [1] [6]. | Ensures variants are not missed due to unsequenced regions [1]. |
| Common Challenges | Higher cost for deep sequencing; managing data volume [2]. | Uneven representation of complex regions (e.g., high GC content) [1] [6]. |
Performance Metrics and Experimental Data from the AmpliSeq Childhood Cancer Panel
Validation studies for targeted NGS panels provide concrete data on how depth and coverage translate into assay performance. Research on the AmpliSeq for Illumina Childhood Cancer Panel offers a relevant case study.
Key Performance Metrics for a Targeted Panel
The table below summarizes the panel's specifications and reported performance metrics from a 2022 validation study [3].
| Metric | Specification / Reported Performance |
|---|---|
| Target Genes | 203 genes associated with pediatric cancer [3] [7]. |
| Input Quantity | 100 ng of DNA and RNA [3]. |
| Mean Read Depth Achieved | >1000x [3]. |
| Sensitivity (DNA, 5% VAF) | 98.5% [3]. |
| Specificity | 100% [3]. |
| Reproducibility (DNA) | 100% [3]. |
| Clinical Impact | 49% of mutations and 97% of fusions identified had clinical impact [3]. |
The Interplay of Metrics in Practice
Beyond raw depth, other metrics are critical for evaluating the efficiency of a targeted sequencing run [6]:
- On-target Rate: The percentage of sequencing reads that map to the intended target regions. A high rate indicates strong probe specificity and efficient enrichment [6].
- Coverage Uniformity: How evenly sequencing reads are distributed across the target regions. The Fold-80 base penalty metric quantifies this; a value of 1 indicates perfect uniformity, while higher values indicate uneven coverage [6].
- Duplicate Rate: The fraction of reads that are exact duplicates, which offer no additional information and are typically removed. High rates can indicate PCR over-amplification or low input material [6].
The following workflow diagrams the experimental protocol for the AmpliSeq Childhood Cancer Panel and how these key metrics are derived from the data:
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of the AmpliSeq Childhood Cancer Panel requires specific reagents and materials. The following table details key components of the workflow [3] [7].
| Item | Function in the Workflow |
|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | Ready-to-use primer pool targeting 203 genes associated with childhood cancer for PCR-based amplification [7]. |
| AmpliSeq Library PLUS for Illumina | Contains reagents for preparing the sequencing libraries, including enzymatic fragmentation and adapter ligation [7]. |
| AmpliSeq CD Indexes for Illumina | Unique barcode sequences used to label individual samples, enabling multiplexing in a single sequencing run [7]. |
| AmpliSeq cDNA Synthesis for Illumina | Converts input RNA to cDNA, a required step for detecting gene fusions via the RNA panel [3] [7]. |
| AmpliSeq Library Equalizer for Illumina | Beads and reagents for normalizing library concentrations before pooling, ensuring balanced representation of samples [7]. |
| Illumina MiSeq/NextSeq Systems | Sequencing instruments using SBS technology to generate the final sequence data [7]. |
| Seraseq Tumor Mutation DNA & Myeloid Fusion RNA Mix | Commercial positive controls containing known variants used for assay validation and determining sensitivity/LOD [3]. |
Selecting Optimal Depth and Coverage for Your Research
Choosing the appropriate depth and coverage is a critical step in experimental design, balancing data quality with cost.
- Define Study Objectives: The required depth is driven by the specific biological question. For example, detecting low-frequency somatic variants in a heterogeneous tumor sample demands much greater depth (500x–1000x) than calling germline variants (30x–50x) [1] [2].
- Consider Sample Characteristics: Low-quality or degraded samples (e.g., from FFPE tissue) may require higher coverage to ensure the target region is adequately represented [1] [7].
- Account for Genomic Context: Regions with high GC content, repetitive elements, or other complexities are often harder to sequence and may be underrepresented, requiring higher overall coverage to ensure they are captured [1] [6].
- Balance with Resources: Higher depth and coverage require more sequencing resources, which increases cost. The optimal design achieves sufficient statistical power while remaining cost-effective [1] [2].
Sequencing depth and coverage are distinct yet complementary pillars of a successful NGS experiment. Depth governs the confidence in your data at each base, while coverage ensures the comprehensiveness of your data across the entire target region. As demonstrated in the validation of the AmpliSeq Childhood Cancer Panel, a mean read depth >1000x enables high sensitivity for variant detection, but this must be considered alongside other metrics like on-target rate and coverage uniformity [3] [6]. A clear understanding of these metrics, combined with a rigorous experimental protocol and the appropriate reagents, empowers researchers to generate reliable, clinically actionable genomic insights in the fight against childhood cancer.
The Clinical Imperative for High Read Depth in Detecting Low-Frequency Pediatric Cancer Variants
The accurate genomic classification of pediatric cancers is a cornerstone of modern precision medicine, directly influencing diagnosis, risk stratification, and treatment selection. Unlike many adult cancers, pediatric malignancies are characterized by a relatively low mutational burden, but the alterations that are present are often clinically decisive. A significant diagnostic challenge emerges because many of these critical variants, including emerging resistance mutations or subclonal populations, can be present at very low allele frequencies. This article examines the performance metrics of targeted next-generation sequencing (NGS) panels, with a specific focus on the AmpliSeq for Illumina Childhood Cancer Panel, in addressing the clinical imperative for high read depth to reliably detect these low-frequency variants in pediatric cancer.
The Critical Need for Sensitivity in Pediatric Cancer Genomics
The landscape of pediatric acute leukemia (AL) demonstrates why sensitive detection is non-negotiable. AL is the most common pediatric neoplasm and the primary cause of cancer-related death in childhood. Its genetic landscape is defined by a low number of mutations, but these are generally highly clinically relevant [3]. Effective management relies on a complex combination of techniques like flow cytometry, FISH, and karyotype analysis to classify the disease into clinically relevant genomic subtypes [8]. A single-assay NGS approach can streamline this process, but its utility depends on its ability to match the sensitivity of these combined traditional methods.
The clinical impact of a sensitive NGS panel is significant. One validation study of the AmpliSeq Childhood Cancer Panel found that it identified clinically relevant results in 43% of pediatric AL patients. Furthermore, 49% of the mutations identified were considered targetable, and 41% helped refine the diagnosis, directly influencing potential treatment pathways [3].
The challenge of low-frequency variants is not theoretical. Broad analysis of clinical cancer samples reveals that a substantial fraction of clinically actionable variants have low variant allele frequencies (VAFs), frequently below 5%. This can be due to low tumor purity or treatment-induced mutations [9].
Table: Prevalence of Low VAF Mutations in Key Hotspot Genes
| Gene | Hotspot Example | Percentage of Mutations with VAF < 5% |
|---|---|---|
| EGFR | T790M | 24% |
| PIK3CA | E545 | 17% |
| EGFR | General | 16% |
| KRAS | G12 | 12% |
| BRAF | General | 10% |
Source: Adapted from [9]
These findings underscore that not detecting low-VAF variants means missing a significant number of clinically actionable alterations. For example, the EGFR T790M mutation is a known mechanism of resistance to tyrosine kinase inhibitor therapy, and its detection is critical for switching patients to a third-generation inhibitor like osimertinib [9]. Failing to detect it at a low VAF could therefore deprive a patient of an effective subsequent therapy.
Performance Benchmarking: AmpliSeq Childhood Cancer Panel and Alternatives
The AmpliSeq for Illumina Childhood Cancer Panel is a targeted NGS solution designed specifically for the genomic profile of childhood and young adult cancers. It investigates 203 genes associated with these malignancies, detecting single nucleotide variants (SNVs), insertions-deletions (indels), copy number variants (CNVs), and gene fusions from DNA and RNA [7]. A key performance metric for any panel is its achieved read depth, as this directly influences sensitivity.
Table: Performance Metrics of the AmpliSeq Childhood Cancer Panel in a Validation Study
| Performance Metric | DNA | RNA |
|---|---|---|
| Mean Read Depth | >1000x | Information Not Specified |
| Sensitivity | 98.5% (for variants at 5% VAF) | 94.4% |
| Specificity | 100% | Information Not Specified |
| Reproducibility | 100% | 89% |
Source: Adapted from [3]
The validation data shows that with a mean read depth exceeding 1000x, the panel achieves a high sensitivity of 98.5% for DNA variants at a 5% VAF, demonstrating its suitability for detecting low-frequency mutations [3]. The high specificity and reproducibility further confirm its reliability for clinical application.
Alternative sequencing approaches offer different trade-offs between breadth and depth. Whole-exome sequencing (WES), for instance, provides a broader view of the coding genome but typically at a lower read depth (often around 100-200x), which limits its sensitivity for low-VAF variants [9]. More recently, long-read whole-genome sequencing (WGS) with adaptive sampling, as demonstrated in a study of 57 pediatric acute leukemia cases, has emerged as a comprehensive single-assay alternative. This method can identify subtype-defining alterations, including karyotypic abnormalities and complex structural variants, within 48 hours. A key advantage is its ability to perform real-time analysis, identifying driving alterations in as little as 15 minutes for karyotype or up to 6 hours for complex fusions, all while maintaining the breadth to assess copy-number variation [8]. However, the sensitivity of this method for very low VAF single-nucleotide variants compared to deep-targeted panels like AmpliSeq requires further direct comparison.
Experimental Protocols for Validation and Detection
Protocol: Technical Validation of a Targeted NGS Panel
The following methodology was used to validate the AmpliSeq Childhood Cancer Panel, establishing its performance metrics [3]:
- Sample Selection: Use commercial controls (e.g., SeraSeq Tumor Mutation DNA Mix) with known mutations at specific VAFs to assess sensitivity, specificity, and limit of detection (LOD). Include patient samples from relevant cancer types (e.g., BCP-ALL, T-ALL, AML).
- Nucleic Acid Extraction: Extract DNA and RNA using standardized kits (e.g., QIAamp DNA Mini Kit, Direct-zol RNA MiniPrep). Assess purity (OD260/280 >1.8) and integrity, with quantification via fluorometry (e.g., Qubit Fluorometer).
- Library Preparation & Sequencing: Prepare libraries using the AmpliSeq for Illumina Childhood Cancer Panel kit according to manufacturer instructions. Use 100 ng of DNA and RNA input. For RNA, perform reverse transcription to cDNA first. Pool DNA and RNA libraries at a 5:1 ratio and sequence on an Illumina MiSeq sequencer.
- Data Analysis: Analyze sequencing data for the established metrics of mean read depth, sensitivity, specificity, and reproducibility against the known variants in the controls.
Protocol: Investigating Low VAF Variants in Clinical Samples
A large-scale study of clinical samples provides a framework for assessing low-VAF prevalence [9]:
- Sequencing: Profile DNA from tumor samples (e.g., FFPE or fresh-frozen) using a custom targeted panel (e.g., CancerSCAN, covering 381 genes) with a mean coverage of approximately 900x.
- Variant Calling: Identify somatic SNVs using a combination of callers like MuTect and LoFreq, the latter being designed for low-VAF variants. Apply a regression-based filter trained on low-VAF variants in normal samples to enhance accuracy.
- Germline Filtering: In the absence of a paired normal for every sample, use a panel of >400 unpaired normal samples to filter out common germline variants effectively.
- VAF Analysis & Validation: Characterize the VAF distribution of mutations, focusing on known hotspots. Correlate VAFs from sequencing with an orthogonal method, such as digital PCR (dPCR), for validation.
Visualizing the Clinical Impact of Detecting Low-Frequency Variants
The following diagram illustrates the clinical decision-making pathway that depends on the sensitive detection of low-VAF variants, a capability defined by sequencing read depth.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table: Key Research Reagent Solutions for Targeted NGS in Pediatric Cancer
| Item | Function | Example Product |
|---|---|---|
| Targeted NGS Panel | Simultaneously targets genes associated with pediatric cancers for variant detection. | AmpliSeq for Illumina Childhood Cancer Panel [7] |
| Library Prep Kit | Contains reagents for preparing sequencing libraries from the panel amplicons. | AmpliSeq Library PLUS for Illumina [7] |
| Index Adapters | Unique molecular barcodes added to each sample for multiplexing. | AmpliSeq CD Indexes for Illumina [7] |
| cDNA Synthesis Kit | Converts total RNA to cDNA for fusion gene detection from RNA input. | AmpliSeq cDNA Synthesis for Illumina [7] |
| DNA/RNA Extraction Kits | Isolates high-quality nucleic acids from various sample types (blood, bone marrow, FFPE). | QIAamp DNA Mini Kit, Direct-zol RNA MiniPrep [3] |
| Library Normalization Beads | Normalizes library concentrations for balanced sequencing throughput. | AmpliSeq Library Equalizer for Illumina [7] |
| Quality Control Instruments | Assesses nucleic acid purity, integrity, and concentration. | Qubit Fluorometer, TapeStation, Labchip [3] |
| Positive Control Materials | Validates assay sensitivity, specificity, and limit of detection. | SeraSeq Tumor Mutation DNA Mix, SeraSeq Myeloid Fusion RNA Mix [3] |
The move towards precision medicine in pediatric oncology is irrevocably linked to the sensitivity of our genomic tools. The high prevalence of low-VAF, clinically actionable variants makes a compelling case for the use of deep-targeted sequencing. The AmpliSeq Childhood Cancer Panel, with its demonstrated high sensitivity and specificity at a 5% VAF and mean read depth greater than 1000x, represents a robust solution for integrating comprehensive genomic profiling into clinical practice. While alternative methods like long-read WGS with adaptive sampling show great promise for rapid and comprehensive structural variant detection, deep-targeted panels currently provide the validated, high-sensitivity data required to guide critical treatment decisions for children with cancer.
Targeted next-generation sequencing (NGS) panels have revolutionized molecular diagnostics in pediatric oncology by enabling comprehensive genomic profiling from minimal input samples. The AmpliSeq for Illumina Childhood Cancer Panel represents a significant advancement as a targeted 203-gene solution specifically designed for childhood and young adult cancers. This review provides a systematic comparison of its performance metrics against alternative panels, including detailed analytical validation data, clinical utility assessments, and implementation protocols. By synthesizing evidence from multiple validation studies, we demonstrate how this panel integrates into clinical research workflows to refine diagnoses, inform prognostic stratification, and identify targetable alterations in pediatric malignancies, particularly acute leukemia.
Performance Metrics and Comparative Analysis
The AmpliSeq Childhood Cancer Panel interrogates 203 genes to detect multiple variant types, including single nucleotide variants (SNVs), insertions and deletions (InDels), copy number variants (CNVs), and gene fusions across diverse pediatric cancer types [3] [7]. The panel's design addresses the distinctive genetic landscape of pediatric cancers, which characteristically have lower mutational burden but higher clinical relevance compared to adult cancers [3].
Analytical Performance Comparison
The following table summarizes key analytical validation metrics for the AmpliSeq Childhood Cancer Panel alongside two alternative pediatric cancer panels:
Table 1: Comparative Analytical Performance of Pediatric Cancer NGS Panels
| Performance Parameter | AmpliSeq Childhood Cancer Panel | CANSeqKids Panel | OncoKids Panel |
|---|---|---|---|
| Number of Genes | 203 genes [3] | 203 unique genes [10] | 190 total targets (44 full gene, 82 hotspot, 24 CNV, 1421 fusions) [11] |
| Sensitivity (DNA) | 98.5% (variants at 5% VAF) [3] | >99% [10] | Robust performance demonstrated [11] |
| Sensitivity (RNA) | 94.4% (fusion detection) [3] | >99% [10] | Robust performance demonstrated [11] |
| Specificity | 100% (DNA), 100% (RNA) [3] | >99% [10] | Robust performance demonstrated [11] |
| Limit of Detection | 5% VAF for SNVs/InDels [3] | 5% allele fraction for SNVs/InDels, 5 copies for amplifications, 1,100 reads for fusions [10] | Low input amounts (20 ng DNA/RNA) [11] |
| Reproducibility | 100% (DNA), 89% (RNA) [3] | >99% [10] | Robust reproducibility demonstrated [11] |
| Mean Read Depth | >1000× [3] | Not specified | Not specified |
| Input Requirements | 10-100 ng DNA/RNA [3] [7] | 5 ng nucleic acid, 20% neoplastic content [10] | 20 ng DNA/RNA [11] |
Clinical Utility and Impact
The clinical impact of genomic findings is a critical metric for evaluating any diagnostic panel. The following table compares the clinical utility of the AmpliSeq Childhood Cancer Panel with broader precision medicine initiatives in pediatric oncology:
Table 2: Clinical Impact Assessment Across Pediatric Cancer Profiling Approaches
| Clinical Utility Metric | AmpliSeq Childhood Cancer Panel | Large-Scale Precision Medicine Programs |
|---|---|---|
| Patients with Clinically Relevant Findings | 43% of pediatric acute leukemia patients [3] | 69% (MAPPYACTS), 86% (GAIN) [12] |
| Diagnostic Refinement | 41% of mutations, 97% of fusions [3] | 5% (GAIN consortium) [12] |
| Therapeutically Targetable Alterations | 49% of mutations [3] | 70% (GAIN consortium) [12] |
| PGT Uptake | Not specified | 10-33% across major platforms [12] |
| Objective Response Rate with PGT | Not specified | 17-38% (depending on evidence level) [12] |
Experimental Protocols and Methodologies
Library Preparation and Sequencing Workflow
The AmpliSeq Childhood Cancer Panel employs a PCR-based amplification approach with optimized protocols for degraded samples and low nucleic acid input [3] [7]. The standardized workflow ensures consistency across different laboratory settings.
Workflow for AmpliSeq Childhood Cancer Panel Analysis
Detailed Experimental Protocol
The validation study for the AmpliSeq Childhood Cancer Panel utilized 76 pediatric patients diagnosed with B-cell precursor ALL (n=51), T-ALL (n=11), and AML (n=14) [3]. The methodological approach included:
- Nucleic Acid Extraction: DNA extraction using Gentra Puregene kit (Qiagen), QIAamp DNA Mini Kit, or QIAamp DNA 2.7 Micro Kit (Qiagen). RNA extraction using guanidine thiocyanate-phenol-chloroform method (TriPure, Roche) or column-based methods (Direct-zol RNA MiniPrep, Zymo Research) [3].
- Quality Control: DNA/RNA purity assessment with OD260/280 ratio >1.8 using spectrophotometry; integrity evaluation by Labchip or TapeStation; concentration determination via fluorometric quantification with Qubit 4.0 Fluorimeter [3].
- Library Preparation: 100 ng each of DNA and RNA used as input. DNA generated 3,069 amplicons (average size: 114 bp); RNA generated 1,701 amplicons (average size: 122 bp) targeting fusion genes. RNA reverse transcribed to cDNA using AmpliSeq cDNA Synthesis kit. Libraries prepared with sample-specific barcodes [3].
- Sequencing: Libraries pooled at 5:1 ratio (DNA:RNA), diluted to 17-20 pM, and sequenced on MiSeq sequencer [3].
Similar validation methodologies were employed for the CANSeqTMKids panel, which utilized 65 samples including FFPE tissue, cell blocks, whole blood, bone marrow, and commercial controls [10]. The OncoKids panel validation employed 192 unique clinical samples across various pediatric tumor types [11].
Technical Implementation and Research Reagents
Essential Research Reagent Solutions
Table 3: Key Research Reagents for Panel Implementation
| Reagent / Kit | Manufacturer | Function in Workflow |
|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | Illumina | Core panel containing primers for amplifying 203 target genes |
| AmpliSeq Library PLUS | Illumina | Reagents for library preparation (24, 96, or 384 reactions) [7] |
| AmpliSeq CD Indexes | Illumina | Sample-specific barcodes for multiplexing (Sets A-D available) [7] |
| AmpliSeq cDNA Synthesis for Illumina | Illumina | Converts total RNA to cDNA for RNA fusion detection [7] |
| AmpliSeq Library Equalizer for Illumina | Illumina | Normalizes libraries for balanced sequencing [7] |
| SeraSeq Tumor Mutation DNA Mix | SeraCare | Positive control for DNA variant detection (22 genes) [3] |
| SeraSeq Myeloid Fusion RNA Mix | SeraCare | Positive control for RNA fusion detection [3] |
| Qubit dsDNA BR Assay Kit | Thermo Fisher Scientific | Fluorometric quantification of DNA concentration [3] |
| Qubit RNA BR Assay Kit | Thermo Fisher Scientific | Fluorometric quantification of RNA concentration [3] |
Platform Integration and Automation
The AmpliSeq Childhood Cancer Panel is compatible with multiple Illumina sequencing platforms, including MiSeq, NextSeq 550, NextSeq 1000/2000, and MiniSeq systems [7]. The library preparation process requires 5-6 hours with less than 1.5 hours of hands-on time, enabling rapid turnaround in clinical research settings [7]. Automation compatibility with liquid handling robots further enhances reproducibility and throughput [7].
The CANSeqTMKids panel, utilizing Thermo Fisher's Oncomine Childhood Cancer Research Assay, demonstrates alternative platform implementation with compatibility for Ion GeneStudio S5 Prime sequencing systems and automated library preparation on Ion Chef instruments [10].
Clinical Utility in Pediatric Cancer Research
Diagnostic and Therapeutic Impact
The decision pathway below illustrates how molecular findings from the AmpliSeq Childhood Cancer Panel translate to clinical applications:
Clinical Translation of Genomic Findings
The AmpliSeq panel demonstrated significant clinical impact in validation studies, with 49% of identified mutations and 97% of detected fusions having clinical relevance [3]. Specifically, 41% of mutations refined diagnostic classification, while 49% were considered therapeutically targetable [3]. For RNA analysis, fusion genes were particularly impactful, refining diagnosis in 97% of cases [3].
Integration with Precision Medicine Platforms
The AmpliSeq Childhood Cancer Panel aligns with major global precision medicine initiatives for pediatric oncology, including:
- MAPPYACTS: European trial demonstrating 69% actionable targets in relapsed/refractory pediatric cancers [12]
- GAIN/iCat2: US consortium reporting 86% of patients with clinically impactful alterations [12]
- INFORM: Multinational registry showing significant survival benefit with targeted therapy for specific mutations [12]
- ZERO Childhood Cancer: Australian program achieving 67% targeted therapy recommendation rate [12]
These programs collectively demonstrate that comprehensive molecular profiling can identify actionable targets in the majority of high-risk pediatric cancer patients, with objective response rates of 17-38% when matched therapies are administered based on high-level evidence [12].
The AmpliSeq for Illumina Childhood Cancer Panel represents a robust, analytically validated solution for molecular profiling of pediatric malignancies. Performance metrics demonstrate high sensitivity, specificity, and reproducibility across multiple sample types, with significant clinical utility in refining diagnosis and identifying targetable alterations. When compared to alternative panels, it offers competitive performance with the advantage of integration into Illumina sequencing workflows. As pediatric precision medicine continues to evolve, standardized targeted panels like AmpliSeq provide clinically actionable insights that can improve outcomes for children with cancer, particularly when integrated into multidisciplinary molecular tumor boards and precision oncology platforms.
How Read Depth Influences Variant Calling Accuracy and Confidence in Heterogeneous Samples
In the context of AmpliSeq Childhood Cancer Panel research, understanding the relationship between read depth and variant calling accuracy is paramount for reliable molecular characterization of pediatric cancers. Next-generation sequencing (NGS) has revolutionized genomic analysis in clinical and research settings, yet determining optimal sequencing parameters remains challenging, particularly for heterogeneous samples such as tumors. Sequencing depth (the average number of times a nucleotide is read) and coverage (the percentage of the target region sequenced) interact to determine variant detection confidence, especially for low-frequency variants in mixed cell populations [1].
This guide objectively compares the performance of the AmpliSeq Childhood Cancer Panel with alternative approaches, examining how read depth influences diagnostic sensitivity across various genomic alterations. We present experimental data from technical validations and benchmarking studies to inform researchers and drug development professionals about critical performance metrics for pediatric cancer genomics.
Fundamental Concepts: Depth, Coverage, and Heterogeneous Samples
Defining Key Metrics
Sequencing depth (or read depth) refers to the number of times a specific nucleotide is read during sequencing, expressed as an average multiple (e.g., 1000×) [1]. Coverage describes the percentage of the target genome or exome that has been sequenced at least once [1]. These metrics jointly determine variant calling accuracy, with higher depth increasing confidence in base calls and broader coverage ensuring comprehensive genomic assessment.
In heterogeneous samples like tumors, where genetic variants may be present in only a subset of cells, sufficient read depth becomes critical for detecting low-frequency variants. The variant allele frequency (VAF) - the percentage of sequencing reads containing a specific variant - must be distinguished from the overall sequencing depth, as low VAF variants require greater total depth for reliable detection [13].
Technical Considerations for the AmpliSeq Childhood Cancer Panel
The AmpliSeq for Illumina Childhood Cancer Panel is a targeted resequencing solution designed specifically for pediatric and young adult cancers [7]. This PCR-based panelinterrogates 203 genes associated with childhood cancers using only 10 ng of input DNA or RNA, making it suitable for precious clinical samples [7]. The panel detects multiple variant types including single nucleotide polymorphisms (SNPs), insertions-deletions (indels), copy number variants (CNVs), and gene fusions across diverse pediatric cancer types including leukemias, brain tumors, and sarcomas [7].
Table 1: AmpliSeq Childhood Cancer Panel Specifications
| Parameter | Specification |
|---|---|
| Target Genes | 203 |
| Input Requirement | 10 ng DNA or RNA |
| Hands-on Time | <1.5 hours |
| Assay Time | 5-6 hours (library prep only) |
| Variant Types Detected | SNPs, indels, CNVs, gene fusions |
| Compatible Systems | MiSeq, NextSeq 500/1000/2000, MiniSeq |
| Specialized Samples | Blood, bone marrow, FFPE tissue, low-input samples |
Experimental Data: Read Depth and Variant Calling Performance
Technical Validation of the AmpliSeq Childhood Cancer Panel
A comprehensive validation study demonstrated that the AmpliSeq Childhood Cancer Panel achieved a mean read depth greater than 1000× across targeted regions, providing sufficient coverage for reliable variant detection [13]. The panel showed a sensitivity of 98.5% for DNA variants with 5% variant allele frequency (VAF), indicating robust performance for detecting low-frequency variants in heterogeneous samples [13]. For RNA-based fusion detection, the panel achieved 94.4% sensitivity, demonstrating utility for identifying structural variants [13].
The validation established 100% specificity and reproducibility for DNA variants, while RNA fusion detection showed 89% reproducibility [13]. These metrics confirm that the panel provides reliable performance for comprehensive molecular characterization of pediatric acute leukemia, with 49% of identified mutations and 97% of detected fusions demonstrating clinical impact for diagnosis refinement or treatment selection [13].
Comparative Performance Across Sequencing Depths
Empirical evaluation using ultra-deep whole-genome sequencing (approximately 410×) examined how gradual depth reductions affect variant calling accuracy [14]. The study constructed simulation datasets with depths ranging from 0.05× to 410×, revealing that SNV concordance rates exceeded 95% at 17.6× depth when compared to ultra-deep sequencing data [14]. However, indel detection showed only 60% concordance at the same depth, indicating that accurate indel calling requires substantially higher read depths [14].
For clinical applications, the study found that >13.7× depth achieved >99% concordance with SNP microarray data, suggesting this as a minimum threshold for reliable SNV detection in diagnostic settings [14]. The transition/transversion (Ts/Tv) ratio, a quality metric for variant calls, remained above 2.0 at depths as low as 0.5× when using the HaplotypeCaller algorithm, though traditional callers like UnifiedGenotyper showed significantly poorer performance at low depths [14].
Table 2: Variant Calling Accuracy at Different Sequencing Depths
| Sequencing Depth | SNV Concordance | Indel Concordance | Recommended Application |
|---|---|---|---|
| <5× | <95% | <30% | Population-level studies only |
| 13.7× | >99% (vs. microarray) | N/A | Clinical SNV detection |
| 17.6× | >95% (vs. WGS) | ~60% | Research SNV detection |
| 30× | >99% | ~80% | Standard WGS |
| 100× | >99% | >90% | Indel-rich regions |
| 1000× | >99.5% | >98% | Low-frequency variant detection |
Impact of Bioinformatics Tools on Depth Requirements
Benchmarking studies reveal that bioinformatics tools significantly influence the relationship between read depth and variant calling accuracy. Deep learning-based variant callers such as Clair3 and DeepVariant demonstrate superior performance compared to traditional methods, particularly for Oxford Nanopore Technologies (ONT) long-read sequencing data [15]. These tools achieve SNP F1 scores of 99.99% and indel F1 scores of 99.53% when using high-accuracy basecalling models, outperforming Illumina short-read sequencing with standard variant callers [16].
Notably, advanced computational methods can compensate for lower sequencing depths. The HBimpute pipeline, which leverages haplotype blocks to artificially increase effective read depth, reduces imputation error rates by half compared to state-of-the-art software like BEAGLE and STITCH [17]. This approach increases the average read-depth from 0.5× to 83× through computational means, enabling accurate calling of copy number variations even from low-depth sequencing data [17].
Experimental Protocols for Technical Validation
Sample Preparation and Library Construction
The technical validation of the AmpliSeq Childhood Cancer Panel followed a rigorous protocol [13]. DNA and RNA were extracted from patient samples using column-based methods (QIAamp DNA Mini Kit, Direct-zol RNA MiniPrep) with quality assessment via spectrophotometry (OD260/280 ratio >1.8) and integrity analysis (Labchip or TapeStation) [13]. Library preparation utilized 100 ng of input DNA or RNA following manufacturer's instructions, generating 3,069 amplicons per DNA sample (average size 114 bp) and 1,701 amplicons per RNA sample (average size 122 bp) [13].
For comparative accuracy assessment across depths, the ultra-deep WGS study employed a random read sampling approach [14]. Researchers began with 410× WGS data and computationally subsampled reads to create 54 discrete depth levels ranging from 0.05× to 410× [14]. This enabled empirical measurement of variant calling accuracy across the depth spectrum while controlling for other variables.
Sequencing and Data Analysis
Sequencing was performed on Illumina platforms (MiSeq, NextSeq) with data processing primarily based on GATK Best Practices guidelines [14] [13]. The variant calling pipeline included duplicate read removal, base quality score recalibration, and variant filtration using either variant quality score recalibration (VQSR) or hard filtering (HF) approaches [14].
For the AmpliSeq panel validation, sensitivity and specificity were assessed using commercial controls: SeraSeq Tumor Mutation DNA Mix (for DNA variants) and SeraSeq Myeloid Fusion RNA Mix (for RNA fusions) [13]. The limit of detection (LOD) was established through serial dilution experiments, determining the minimum variant allele frequency detectable with 95% confidence [13].
Diagram 1: Experimental workflow for panel validation
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for AmpliSeq Panel Validation
| Reagent/Material | Function | Specifications |
|---|---|---|
| AmpliSeq Childhood Cancer Panel | Target enrichment | 203 genes, 24 reactions |
| AmpliSeq Library PLUS | Library preparation | 24, 96, or 384 reactions |
| AmpliSeq CD Indexes | Sample multiplexing | 96 indexes per set |
| SeraSeq Tumor Mutation DNA Mix | Positive control for DNA variants | 10% VAF for 22 genes |
| SeraSeq Myeloid Fusion RNA Mix | Positive control for RNA fusions | Includes ETV6::ABL1, BCR::ABL1, etc. |
| NA12878 DNA | Negative control for DNA variants | Coriell Institute |
| IVS-0035 RNA | Negative control for RNA fusions | Invivoscribe |
| QIAamp DNA Mini Kit | DNA extraction | Blood, tissue, cells |
| Direct-zol RNA MiniPrep | RNA extraction | All sample types including FFPE |
The relationship between read depth and variant calling accuracy demonstrates distinct patterns across different variant types and genomic contexts. For the AmpliSeq Childhood Cancer Panel, mean read depths exceeding 1000× provide sufficient sensitivity to detect variants with allele frequencies as low as 5%, which is crucial for heterogeneous tumor samples [13]. While SNVs can be reliably detected at depths as low as 13.7-17.6×, accurate indel calling requires substantially higher depths (>100×) due to the increased complexity of alignment and variant detection [14].
The integration of advanced bioinformatics tools, particularly deep learning-based variant callers, can partially compensate for lower sequencing depths by improving variant detection algorithms [15] [16]. Similarly, computational imputation methods like HBimpute demonstrate that strategic data analysis can effectively increase usable read depth, enabling accurate variant calling from initially low-coverage data [17].
For researchers utilizing the AmpliSeq Childhood Cancer Panel, these findings support the recommendation of high read depths (>500×) for comprehensive molecular characterization of pediatric cancers, particularly when assessing indels, copy number variations, and low-frequency variants in heterogeneous samples. The panel's validated performance at these depths provides clinicians and researchers with confidence in detecting clinically actionable variants that inform diagnosis, prognosis, and therapeutic strategies for childhood cancers.
Achieving Optimal Performance: Technical Validation and Workflow for the Childhood Cancer Panel
This guide provides a detailed comparison of the library preparation protocol for the AmpliSeq for Illumina Childhood Cancer Panel, placing its performance metrics—specifically input requirements and hands-on time—within the broader context of targeted next-generation sequencing (NGS) for pediatric cancer research.
Product Comparison at a Glance
The table below summarizes the core library preparation specifications for the AmpliSeq Childhood Cancer Panel and two other common pediatric cancer NGS panels.
| Parameter | AmpliSeq for Illumina Childhood Cancer Panel | OncoKids Panel | CANSeqTMKids Panel |
|---|---|---|---|
| DNA Input | 100 ng (high-quality) [3] | 20 ng [11] | 5 ng (minimum) [10] |
| RNA Input | 100 ng [3] | 20 ng [11] | 10 ng (minimum) [10] |
| Total Hands-On Time | < 1.5 hours [7] | Information Missing | Information Missing |
| Total Assay Time (Library Prep) | 5-6 hours [7] | Information Missing | Information Missing |
| Compatible Sample Types | Blood, Bone Marrow, FFPE Tissue [7] | FFPE Tissue, Bone Marrow, Peripheral Blood [11] | FFPE Tissue, Bone Marrow, Whole Blood, Cell Blocks [10] |
Detailed Protocol and Experimental Methodology
The following section details the experimental procedures and key metrics from a validation study of the AmpliSeq Childhood Cancer Panel, providing a practical framework for its implementation [3] [13].
Sample Preparation and Quality Control
In the referenced validation study, DNA and RNA were co-extracted from diagnostic samples of 76 pediatric acute leukemia patients [3] [13].
- Extraction Kits: DNA was extracted using Qiagen kits (Gentra Puregene, QIAamp DNA Mini Kit, or QIAamp DNA 2.7 Micro Kit). RNA was extracted via guanidine thiocyanate-phenol-chloroform (TriPure, Roche) or column-based methods (Direct-zol RNA MiniPrep, Zymo Research) [3].
- Quality Control (QC): Nucleic acid purity was confirmed with a spectrophotometer (OD260/280 ratio >1.8). Integrity was assessed via Labchip (PerkinElmer) or TapeStation (Agilent). Concentration was determined by fluorometric quantification (Qubit 4.0 Fluorimeter, ThermoFisher) [3].
Library Preparation Workflow
The library preparation for the AmpliSeq Childhood Cancer Panel follows a PCR-based protocol. The workflow involves parallel processing of DNA and RNA, which are later pooled for sequencing [3] [18].
Key Performance Outcomes from Validation
The technical validation of the panel demonstrated robust performance, underpinning its reliability for clinical research [3] [13].
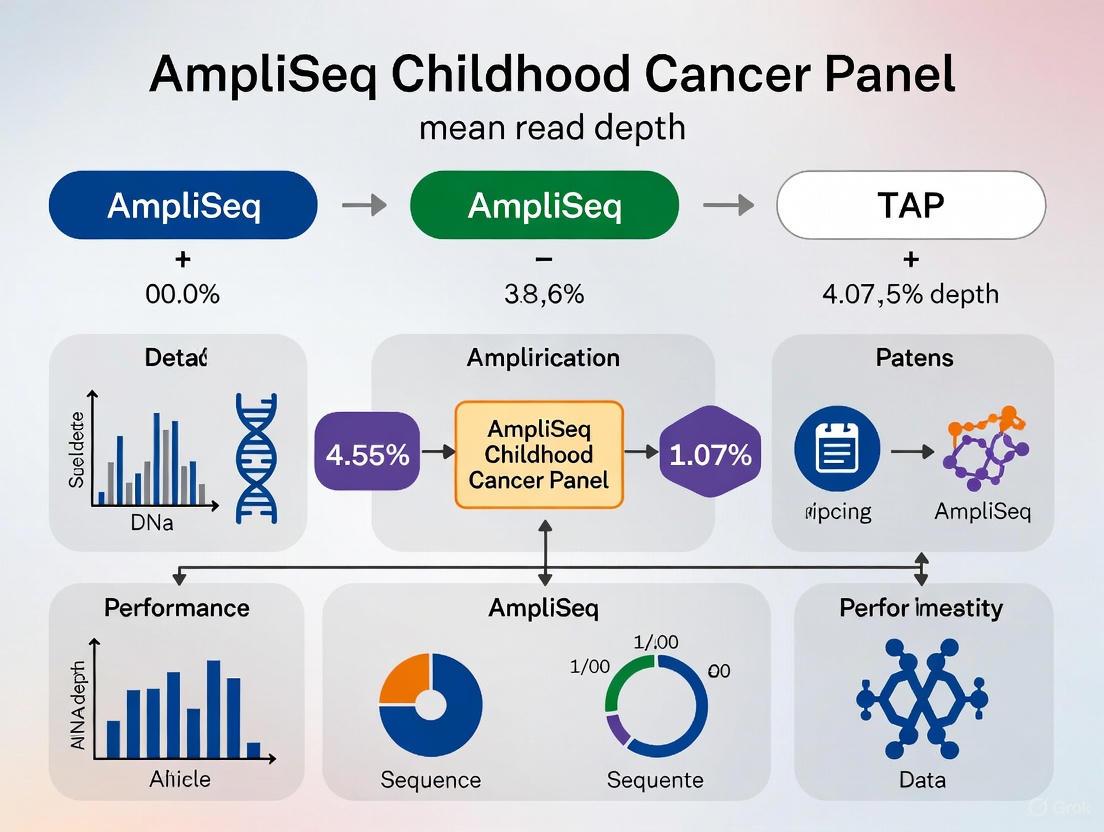
- Sequencing Metrics: The assay achieved a mean read depth greater than 1000x, ensuring accurate variant detection [3] [13].
- Sensitivity and Specificity: The panel showed high DNA sensitivity of 98.5% for variants at 5% variant allele frequency (VAF) and RNA sensitivity of 94.4% for fusion detection. Specificity and reproducibility for DNA were 100% [3] [13].
- Clinical Utility: The panel provided clinically impactful results, refining diagnosis or revealing targetable mutations in 43% of patients in the studied cohort [3] [13].
The Scientist's Toolkit: Essential Research Reagents
The table below lists the key consumables and instruments required to perform the library preparation and sequencing workflow for the AmpliSeq Childhood Cancer Panel, based on the validation study and manufacturer specifications [3] [7] [18].
| Item Category | Specific Product or Instrument | Function in the Workflow |
|---|---|---|
| Core Panel | AmpliSeq for Illumina Childhood Cancer Panel (20028446) | Contains primer pools to target 203 genes for DNA and RNA analysis [7] [18]. |
| Library Prep Kit | AmpliSeq Library PLUS for Illumina (24, 96, or 384 reactions) | Provides core reagents for PCR-based library construction [7] [18]. |
| Index Adapters | AmpliSeq CD Indexes (e.g., Set A, 96 indexes) | Adds unique barcodes to each sample for multiplexed sequencing [7] [18]. |
| cDNA Synthesis Kit | AmpliSeq cDNA Synthesis for Illumina (20022654) | Converts input RNA to cDNA for the RNA fusion component of the panel [7] [18]. |
| Quantification Instrument | Qubit 4.0 Fluorimeter with dsDNA BR and RNA BR Assay Kits | Precisely measures concentration of DNA, RNA, and final libraries [3]. |
| Sequencing System | MiSeq, NextSeq 500/550/1000/2000, or MiniSeq System | Performs the actual sequencing; choice depends on required throughput [7] [18]. |
Experimental Design and Analysis Workflow
The following diagram visualizes the comprehensive experimental design from sample collection to data analysis, illustrating the workflow used in the technical validation of the panel [3] [13].
A documented mean read depth greater than 1000x represents a critical benchmark for next-generation sequencing (NGS) panels, ensuring high sensitivity and reliability for detecting low-frequency variants in cancer research. This article objectively compares the performance of the AmpliSeq for Illumina Childhood Cancer Panel against other targeted sequencing approaches, focusing on this key metric and its implications for precision oncology.
In targeted NGS, read depth (or coverage) refers to the average number of times a specific nucleotide in the genome is sequenced. A higher read depth directly enhances the confidence of variant calling, particularly for detecting subclonal populations and low-frequency mutations that are common in cancer genomics. Targeted gene panels sequence key genes of interest to high depth, allowing identification of rare variants that might be missed by broader, shallower sequencing approaches [19]. For the AmpliSeq Childhood Cancer Panel, a mean read depth greater than 1000x was a pivotal achievement in its validation, facilitating a high sensitivity for DNA variants with a variant allele frequency (VAF) as low as 5% [3].
Performance Benchmarking: AmpliSeq Childhood Cancer Panel vs. Alternatives
The table below summarizes key performance metrics from analytical validation studies for the AmpliSeq Childhood Cancer Panel and a comparable pediatric cancer panel, CANSeqTMKids.
| Performance Metric | AmpliSeq Childhood Cancer Panel [3] | CANSeqTMKids Panel [10] | Significance in Performance Benchmarking |
|---|---|---|---|
| Documented Mean Read Depth | >1000x | Implied by sensitivity (Coverage not specified) | High depth enables detection of low-frequency variants and ensures data robustness. |
| Sensitivity (DNA SNVs/Indels) | 98.5% (for variants at 5% VAF) | >99% (at 5% allele fraction) | Near-perfect sensitivity is crucial for not missing clinically actionable mutations. |
| Sensitivity (RNA Fusions) | 94.4% | Not explicitly stated (Fusion detection validated) | Critical for accurately identifying fusion genes, which are common drivers in pediatric cancer. |
| Limit of Detection (VAF) | 5% VAF | 5% allele fraction | Defines the lowest variant frequency an assay can reliably detect. |
| Reproducibility | 100% (DNA), 89% (RNA) | >99% | Measures the assay's consistency across repeated runs. |
| Specificity | 100% (DNA), 100% (RNA) | >99% | High specificity minimizes false positive results. |
Experimental Protocols for Key Validation Studies
Protocol: Validation of the AmpliSeq Childhood Cancer Panel
The following workflow and detailed methodology were used to establish the performance metrics for the AmpliSeq panel [3].
Sample Selection and Nucleic Acid Extraction:
- The study used a cohort of 76 pediatric patients diagnosed with acute leukemia (B-ALL, T-ALL, AML) and commercial reference standards (e.g., SeraSeq Tumor Mutation DNA Mix) [3].
- DNA was extracted using Gentra Puregene or QIAamp kits, and RNA was extracted via guanidine thiocyanate-phenol-chloroform or column-based methods. Quality and quantity were assessed using spectrophotometry (OD260/280 >1.8), fluorometry (Qubit), and integrity analyzers (Labchip, TapeStation) [3].
Library Preparation and Sequencing:
- Libraries were prepared per the manufacturer's instructions using 100 ng of DNA and 100 ng of RNA (converted to cDNA). The panel generates 3,069 DNA amplicons and 1,701 RNA amplicons.
- Libraries were pooled at a 5:1 DNA-to-RNA ratio and sequenced on an Illumina MiSeq sequencer, achieving the documented mean read depth of >1000x [3].
Data Analysis and Validation:
- Performance metrics were calculated by comparing NGS results against known variants in commercial controls and results from conventional techniques (qRT-PCR, Sanger sequencing). Sensitivity and specificity were determined from true positive, false positive, and false negative variant calls [3].
Protocol: Analytical Validation of the CANSeqTMKids Panel
The CANSeqTMKids panel validation followed a similar rigorous framework, as outlined below [10].
- Sample Cohort: The study utilized 65 samples, including FFPE tissue, cell blocks, blood, bone marrow, cell lines, and commercial controls (e.g., Seraseq mutation mixes and Coriell HapMap samples) [10].
- Library Preparation and Sequencing: Libraries were prepared manually or automated (Ion Chef) using the Oncomine Childhood Cancer Research Assay (OCCRA). Templating was performed on Ion 540 chips, and sequencing was run on the Ion GeneStudio S5 Prime system [10].
- Data Analysis: Raw data was processed and aligned to hg19 via the Ion Torrent Suite. Variant calling and fusion detection were performed using the Ion Reporter software with the OCCRA workflow. Positive percent agreement (PPA, sensitivity) and positive predictive value (PPV) were calculated for all variant types [10].
The Scientist's Toolkit: Essential Research Reagents and Materials
The table below lists key reagents and materials essential for conducting robust targeted sequencing experiments, as featured in the validation studies.
| Item Name | Function / Application | Example Products / Kits |
|---|---|---|
| Commercial Reference Standards | Acts as a positive control for assessing sensitivity, specificity, and limit of detection. Contains known variants at defined allele frequencies. | SeraSeq Tumor Mutation DNA Mix, SeraSeq Myeloid Fusion RNA Mix, AcroMetrix Oncology Hotspot Control [3] [10] |
| Nucleic Acid Extraction Kits | Isolates high-quality DNA and RNA from various sample types, which is critical for successful library preparation. | QIAamp DNA Mini Kit, Gentra Puregene Kit (Qiagen), Direct-zol RNA MiniPrep (Zymo Research) [3] |
| Targeted Sequencing Panel | The core reagent containing primers to amplify the genes of interest. Defines the content of the assay. | AmpliSeq for Illumina Childhood Cancer Panel, Oncomine Childhood Cancer Research Assay [3] [10] |
| Library Preparation Kit | Converts the extracted nucleic acids into a sequencing-ready library by adding platform-specific adapters and barcodes. | AmpliSeq Library PLUS for Illumina, Oncomine Childhood Cancer Chef-Ready Kit [3] [7] [10] |
| NGS Instrument & Flow Cell | The platform that performs the sequencing by synthesis. Different systems offer varying levels of throughput and read length. | Illumina MiSeq, NextSeq 500/1000/2000 systems; Ion GeneStudio S5 Prime System with Ion 540 chip [3] [7] [10] |
The benchmark of a mean read depth exceeding 1000x, as demonstrated by the AmpliSeq Childhood Cancer Panel, is a cornerstone of high-performance targeted sequencing in oncology. This metric, coupled with high sensitivity and specificity, ensures that researchers and clinicians can reliably detect the low-frequency variants that are essential for refining diagnoses, prognoses, and treatment strategies in pediatric cancers. When selecting a targeted panel, verifying these independently validated performance metrics is paramount for generating clinically actionable genomic data.
Targeted next-generation sequencing (NGS) panels have become indispensable tools in clinical oncology research, enabling comprehensive molecular profiling from minimal nucleic acid inputs. Validation of these assays requires rigorous demonstration of sensitivity, specificity, and reproducibility using well-characterized controls and clinical samples. This guide objectively compares the performance of the AmpliSeq for Illumina Childhood Cancer Panel with other commercially available targeted sequencing panels for pediatric malignancies, focusing on key analytical metrics established through standardized validation frameworks.
Performance Metrics Comparison of Pediatric Cancer NGS Panels
The table below summarizes key performance characteristics across multiple targeted NGS panels designed for pediatric cancer profiling.
Table 1: Comparative Analytical Performance of Pediatric Cancer NGS Panels
| Panel Name | DNA Sensitivity | RNA Sensitivity | Specificity | Limit of Detection | Target Genes | Sample Input |
|---|---|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel [3] | 98.5% (5% VAF) | 94.4% | 100% (DNA), 89% (RNA reproducibility) | 5% VAF (DNA) | 203 genes | 100 ng DNA/RNA |
| CANSeqTMKids [10] | >99% | >99% | >99% | 5% VAF (SNVs/INDELs), 5 copies (CNVs), 1100 reads (fusions) | 203 genes | 5 ng (20% neoplastic) |
| OncoKids [11] | Robust (exact values not specified) | Robust (exact values not specified) | Robust (exact values not specified) | Not specified | 150 genes (44 full coding, 82 hotspots, 24 CNVs) + 1421 fusions | 20 ng DNA/RNA |
Experimental Protocols and Methodologies
AmpliSeq Childhood Cancer Panel Validation
The validation of the AmpliSeq Childhood Cancer Panel followed a comprehensive protocol to establish key performance metrics [3].
Sample Selection and Controls: The study utilized 76 pediatric patients diagnosed with B-cell precursor ALL (n=51), T-ALL (n=11), and AML (n=14). Commercial controls included SeraSeq Tumor Mutation DNA Mix (v2 AF10 HC) as positive control for DNA analyses and SeraSeq Myeloid Fusion RNA Mix for RNA analyses. Negative controls consisted of NA12878 (DNA) and IVS-0035 (RNA) [3].
Library Preparation and Sequencing: Libraries were prepared using 100 ng each of DNA and RNA according to the manufacturer's instructions. The DNA component generated 3069 amplicons per sample, while RNA was reverse transcribed to cDNA before generating 1701 amplicons targeting gene fusions. Barcoded libraries were pooled at a 5:1 ratio (DNA:RNA) and sequenced on a MiSeq sequencer, achieving a mean read depth >1000× [3].
Data Analysis: Sensitivity and specificity calculations were based on variant calling performance against known variants in control materials. The panel demonstrated 98.5% sensitivity for DNA variants at 5% variant allele frequency (VAF) and 94.4% sensitivity for RNA fusions, with 100% specificity and reproducibility for DNA and 89% reproducibility for RNA [3].
CANSeqTMKids Validation Methodology
The CANSeqTMKids assay was validated across 65 samples including FFPE tissue, cell blocks, blood, bone marrow, cell lines, and commercial controls [10].
Library Preparation: Libraries were prepared using both manual and automated (Ion Chef) processes with the Oncomine Childhood Cancer Research Assay. The automated process required 15 µL at 0.7 ng/µL for DNA and 10 µL at 1 ng/µL for RNA. DNA and RNA libraries were combined at an 80:20 ratio and templated on Ion 540 chips [10].
Sequencing and Analysis: Sequencing was performed on the Ion GeneStudio S5 Prime Sequencer. Data was processed using Ion Torrent Suite Software (versions 5.12 and 5.14) aligned to hg19. Variant calling used Ion Reporter (versions 5.14 and 5.16) with the OCCRA - w2.5 - IR workflow. The validation established >99% accuracy, sensitivity, repeatability, and reproducibility with a limit of detection of 5% allele fraction for SNVs and INDELs [10].
Workflow and Performance Relationships
The following diagram illustrates the key steps and performance relationships in the validation workflow for targeted NGS panels like the AmpliSeq Childhood Cancer Panel.
NGS Panel Validation Workflow and Key Metrics
The Scientist's Toolkit: Essential Research Reagents
The table below catalogues essential reagents and materials used in validation studies for targeted NGS panels, along with their specific functions in the experimental workflow.
Table 2: Essential Research Reagents for NGS Panel Validation
| Reagent/Material | Function | Example Products |
|---|---|---|
| Reference DNA Controls | Benchmark variant calling performance; establish sensitivity/specificity | SeraSeq Tumor Mutation DNA Mix, AcroMetrix Oncology Hotspot Control, NA12878 [3] [10] |
| Reference RNA Controls | Validate fusion detection sensitivity | SeraSeq Myeloid Fusion RNA Mix, Seraseq Fusion RNA Mix v4 [3] [10] |
| Nucleic Acid Extraction Kits | Isolate high-quality DNA/RNA from various sample types | QIAamp DNA Mini Kit, TriPure Isolation Reagent, column-based methods [3] |
| Library Preparation Kits | Prepare sequencing libraries from DNA/RNA inputs | AmpliSeq for Illumina Childhood Cancer Panel, Oncomine Childhood Cancer Research Assay [3] [10] |
| Target Enrichment Systems | Capture genomic regions of interest | AmpliSeq PCR-based enrichment, SureSelect hybridization-based capture [20] [21] |
| Sequencing Platforms | Generate sequence data from prepared libraries | Illumina MiSeq, Ion GeneStudio S5 Prime, Ion Torrent [3] [10] |
| Analysis Software | Process sequence data, call variants, generate reports | Ion Reporter, Torrent Suite Server, GO Pathology Workbench [10] |
Comparative Analysis of Panel Content and Applications
The AmpliSeq Childhood Cancer Panel targets 203 genes relevant to pediatric cancers, providing coverage of single nucleotide variants (SNVs), insertions/deletions (InDels), copy number variants (CNVs), and gene fusions in a single assay [3]. In validation studies, this panel demonstrated high clinical utility, with 49% of mutations and 97% of fusions identified having clinical impact, refining diagnosis in 41% of mutations and 97% of fusion genes [3].
The CANSeqTMKids panel covers a similar scope of 203 genes but has been optimized for lower input requirements (5 ng at 20% neoplastic content) and demonstrates >99% sensitivity across variant types [10]. Both panels utilize overlapping amplicon designs specifically tailored for pediatric cancers, which differ from adult-focused panels in their gene content and variant prioritization.
The OncoKids panel employs a different strategy, focusing on full coding regions of 44 cancer predisposition genes, hotspots in 82 genes, and 24 amplification targets, plus an extensive fusion repertoire of 1,421 targets [11]. This design reflects alternative approaches to capturing clinically relevant variation in pediatric malignancies.
Validation data demonstrates that the AmpliSeq Childhood Cancer Panel achieves robust performance metrics, particularly in DNA sensitivity (98.5%) and specificity (100%), with slightly lower but still strong RNA sensitivity (94.4%). Comparative analysis shows that alternative panels like CANSeqTMKids report higher overall sensitivity (>99%) but may utilize different validation conditions and input requirements. The selection of an appropriate pediatric cancer panel depends on specific research needs, including sample type, input requirements, and the particular variant types of interest. All validated panels show significant clinical utility for molecular profiling of childhood malignancies, enabling refined diagnosis and potential targeted treatment strategies.
Establishing Limit of Detection (LOD) for Variants at 5% Variant Allele Frequency (VAF)
Performance Comparison of NGS Panels for 5% VAF Detection
Table 1: Performance Metrics of Targeted NGS Panels for 5% VAF Detection
| Panel / Method | Mean Read Depth | Reported Sensitivity at 5% VAF | Specificity | Reproducibility | Key Validation Findings |
|---|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel [3] [13] | >1000× | DNA: 98.5%RNA: 94.4% | 100% (DNA)100% (RNA) | 100% (DNA)89% (RNA) | Validated for pediatric AL diagnostics; 49% of mutations and 97% of fusions had clinical impact |
| Oncomine Myeloid Panel [22] | Not specified | 5% for SNVs10% for indels | High (exact % not specified) | High (exact % not specified) | 48% of variants facilitated/clarified diagnoses; 29% affected prognoses |
| WES with 15 Gbp data [23] | 189× | LOD between 5-10% | Not specified | Varies with coverage | LOD estimated at 8.7% VAF with 15 Gbp sequencing data |
| Custom TP53 Testing [24] | ≥1000× | Reliable detection down to 5% | High with confirmation | 100% for 5-10% VAF range | All TP53 variants in 5-10% VAF range confirmed with second NGS panel |
Experimental Protocols for LOD Validation
AmpliSeq Childhood Cancer Panel Validation Protocol
Sample Preparation and Controls [3] [13]
- Positive Controls: SeraSeq Tumor Mutation DNA Mix (average VAF 10%) containing 22 clinically relevant genes including FLT3, NPM1, TP53, and KRAS
- RNA Fusion Controls: SeraSeq Myeloid Fusion RNA Mix with ETV6::ABL1, TCF3::PBX1, BCR::ABL1, RUNX1::RUNX1T1, and PML::RARA fusions
- Negative Controls: NA12878 (DNA) and IVS-0035 (RNA)
- Input Requirements: 100 ng DNA and 100 ng RNA per sample
- Patient Cohort: 76 pediatric patients with BCP-ALL (n=51), T-ALL (n=11), and AML (n=14)
Library Preparation and Sequencing [3] [13]
- DNA Analysis: 3069 amplicons per sample, average size 114 bp
- RNA Analysis: 1701 amplicons targeting gene fusions, average size 122 bp
- Library Pooling: DNA and RNA libraries pooled at 5:1 ratio
- Sequencing Platform: MiSeq Sequencer with minimum target read depth >1000×
- Data Analysis: Variant calling with Dragen somatic pipeline using "panel of normal" to remove technical artifacts
Orthogonal Confirmation Methods for Low VAF Variants
Blocker Displacement Amplification with Sanger Sequencing [25]
- Principle: BDA technology enables preferential amplification of low-level variants over wildtype sequence
- Application: Confirmation of WES findings at ≤5% VAF
- Performance: Can detect variants down to 0.1% VAF, with 52% of putative WES variants at 0.5-5% VAF being disconfirmed as false positives
- Validation: 82% disconfirmation rate for cancer-related variants in the 0.5-5% VAF range
Digital Droplet PCR Validation [24]
- Application: Orthogonal confirmation of TP53 variants detected at 5-10% VAF by NGS
- Requirements: Coverage ≥1000× with minimum 50 reads for mutant allele and background noise level ≤0.5%
- Outcome: 100% concordance for TP53 variants in 5-10% VAF range when validated with second NGS panel and ddPCR
Coverage Depth and Error Rate Considerations
Table 2: Coverage Depth Requirements for Reliable 5% VAF Detection
| Sequencing Type | Minimum Recommended Depth | Variant Supporting Reads | False Negative Risk | Applications |
|---|---|---|---|---|
| Targeted Panels (AmpliSeq) [3] [13] | >1000× | Not specified | <1.5% for DNA variants | Clinical diagnostics |
| Theoretical Calculation [26] | 1,650× | ≥30 mutated reads | Minimal with proper thresholds | Research settings |
| WES [23] | 189× (15 Gbp data) | Not specified | LOD ~8.7% | Comprehensive mutation screening |
| Clinical TP53 Testing [24] | ≥1000× | ≥50 mutant reads | Minimal with confirmation | Hematologic malignancies |
Error Rate Management [26]
- Sequencing Errors: Conventional intrinsic NGS error rates range between 0.1-1% (Phred quality score 20-30)
- Assay-Specific Errors: Include errors from DNA processing, library preparation, and amplification steps
- Binomial Distribution Calculations: For 5% VAF detection with 1% error rate, coverage of 1,650× with threshold of 30 mutated reads minimizes false positives
- Coverage Calculator: Available tool for determining minimum coverage parameters based on intended LOD and error tolerance
Clinical Utility and Impact
Diagnostic and Therapeutic Implications [3] [13]
- Diagnostic Refinement: 41% of mutations refined diagnosis in pediatric acute leukemia
- Targetable Alterations: 49% of detected mutations were considered targetable
- Fusion Genes: 97% of identified fusion genes had diagnostic impact
- Overall Clinical Impact: 43% of patients tested had clinically relevant findings
Subclonal Mutation Significance [24]
- TP53 in CLL: 23% of pathogenic TP53 variants detected in 5-10% VAF range
- Clinical Reporting: Validated algorithm allows safe reporting of TP53 variants with VAF ≥5%
- Prognostic Impact: Detection of even single variants predictive of overall survival independent of age, sex, or diagnosis
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for 5% VAF Validation
| Reagent / Kit | Function | Application in LOD Studies |
|---|---|---|
| SeraSeq Tumor Mutation DNA Mix [3] [13] | Multiplex biosynthetic positive control | Contains 22 clinically relevant variants at ~10% VAF for sensitivity determination |
| SeraSeq Myeloid Fusion RNA Mix [3] [13] | RNA fusion positive control | Validates fusion detection sensitivity with known fusion transcripts |
| AmpliSeq Library PLUS [7] | Library preparation reagents | Provides consistent library prep for 24, 96, or 384 samples |
| AmpliSeq CD Indexes [7] | Sample multiplexing | Enables barcoding of 96 samples per set for pooled sequencing |
| AmpliSeq cDNA Synthesis for Illumina [7] | RNA to cDNA conversion | Essential for RNA fusion analysis in the Childhood Cancer Panel |
| QIAamp DNA Mini Kit [3] | DNA extraction | High-quality DNA extraction with OD260/280 >1.8 |
| NEBNext FFPE DNA Repair Mix [25] | DNA repair | Critical for FFPE samples with potential DNA damage |
| PowerUp SYBR Green Master Mix [25] | qPCR detection | Used in BDA validation experiments for low VAF confirmation |
The molecular characterization of pediatric acute leukemia (AL) is crucial for refining diagnosis, prognosis, and treatment strategies. While next-generation sequencing (NGS) has revolutionized cancer genomics, its clinical application in pediatric cancers has been challenged by the predominance of panels designed for adult malignancies. The AmpliSeq for Illumina Childhood Cancer Panel was developed specifically to address this gap. This targeted NGS panel encompasses the distinct genetic landscape of childhood cancers, enabling simultaneous analysis of multiple variant types from minimal input nucleic acids. A 2022 validation study demonstrated that this panel identified clinically relevant genetic alterations in 43% of pediatric AL patients, significantly refining their diagnostic classification and opening avenues for personalized therapeutic interventions [3] [13].
Performance Metrics: How the Panel Compares
The technical performance of a sequencing panel determines its reliability in detecting clinically actionable variants. The following table summarizes the key analytical validation data for the AmpliSeq Childhood Cancer Panel compared to conventional techniques.
Table 1: Key Analytical Performance Metrics of the AmpliSeq Childhood Cancer Panel
| Performance Parameter | DNA Analysis | RNA Analysis (Fusions) | Comparison to Conventional Methods |
|---|---|---|---|
| Mean Read Depth | >1000x [3] | Not Specified | Surpasses the depth typically achieved by broader NGS approaches [27]. |
| Sensitivity | 98.5% (for variants at 5% VAF) [3] | 94.4% [3] | Higher sensitivity for fusion detection than some conventional RT-PCR methods [3]. |
| Specificity | 100% [3] | Not Explicitly Stated | 100% concordance with validated methods for DNA variants [3]. |
| Reproducibility | 100% [3] | 89% [3] | Shows high consistency, though RNA assay reproducibility is slightly lower [3]. |
| Input Requirement | 100 ng (used in validation) / 10 ng (manufacturer's spec) [3] [7] | 100 ng (used in validation) / 10 ng (manufacturer's spec) [3] [7] | Enables work with low-input and precious samples like bone marrow [7]. |
| Variant Types Detected | SNVs, InDels, CNVs [7] | Gene Fusions [7] | Consolidates multiple standalone tests (Sanger sequencing, FISH, karyotyping) into one [3]. |
The panel's ability to reliably detect variants with a 98.5% sensitivity at a 5% variant allele frequency (VAF) for DNA and 94.4% sensitivity for RNA fusions makes it a robust tool for identifying low-burden mutations, which are common in pediatric leukemias [3]. Its comprehensive design covers 203 genes, including 97 gene fusions and 82 DNA variants relevant to pediatric cancers, providing a more tailored solution than adult-focused panels [3] [7].
Experimental Protocol: Validation Methodology
The validation study provides a rigorous framework for assessing the panel's clinical utility. The methodology can be broken down into key stages, from sample selection through to data analysis.
Diagram 1: Experimental validation workflow for the AmpliSeq Childhood Cancer Panel.
Sample Selection and Controls
The study utilized 76 pediatric patients diagnosed with B-cell precursor ALL (BCP-ALL, n=51), T-ALL (n=11), and AML (n=14) [3] [13]. Samples were selected based on high DNA/RNA quality and clinical criteria that prioritized patients whose genetic results were not definitive using conventional diagnostics. To accurately assess performance, the study employed commercial controls:
- Positive DNA Control: SeraSeq Tumor Mutation DNA Mix, a multiplex biosynthetic mixture of known DNA variants at ~10% VAF [3] [13].
- Positive RNA Control: SeraSeq Myeloid Fusion RNA Mix, containing synthetic RNA fusions (ETV6::ABL1, TCF3::PBX1, BCR::ABL1, RUNX1::RUNX1T1, PML::RARA) [3] [13].
- Negative Controls: NA12878 (DNA) and IVS-0035 (RNA) to establish baseline specificity [3] [13].
Library Preparation and Sequencing
Library preparation followed the manufacturer's protocol with specific parameters [3] [13]:
- Input Nucleic Acids: 100 ng of DNA and 100 ng of RNA per sample. The panel can function with as little as 10 ng of high-quality DNA or RNA, making it suitable for precious biobank samples [7].
- Amplicon Generation: The DNA panel generated 3,069 amplicons (average size 114 bp), while the RNA panel generated 1,701 amplicons (average size 122 bp) targeting fusion genes.
- RNA Conversion: Total RNA was reverse transcribed to cDNA using the AmpliSeq cDNA Synthesis kit prior to library preparation [3] [7].
- Sequencing: Barcoded libraries were pooled at a 5:1 DNA:RNA ratio and sequenced on a MiSeq sequencer (Illumina) [3]. The panel is also compatible with other Illumina platforms like the NextSeq 550 and NextSeq 2000 systems [7].
Orthogonal Validation
All significant variants identified by the NGS panel were confirmed using established, orthogonal molecular techniques, a critical step for clinical validation [3]:
- FLT3-ITD and NPM1 mutations: Labeled-PCR amplification.
- FLT3 tyrosine kinase domain, cKIT, and GATA1 mutations: Sanger sequencing.
- Fusion genes (e.g., RUNX1::RUNX1T1, BCR::ABL1, ETV6::RUNX1): Quantitative RT-PCR using Europe Against Cancer Program guidelines.
Clinical Impact and Comparison to Alternatives
The ultimate test of a diagnostic panel is its ability to influence patient management. The clinical utility of the AmpliSeq Childhood Cancer Panel was demonstrated by its significant impact on diagnostic refinement and targetable mutation identification.
Table 2: Clinical Impact of Genetic Findings in Pediatric Acute Leukemia
| Clinical Impact Category | DNA Mutations | RNA Fusion Genes | Overall Patient Impact |
|---|---|---|---|
| Refined Diagnosis | 41% of mutations [3] | 97% of fusions [3] | N/A |
| Therapeutically Targetable | 49% of mutations [3] | Not Specified | N/A |
| Total Clinical Impact | 49% of mutations had clinical impact [3] | 97% of fusions had clinical impact [3] | 43% of patients had clinically relevant findings [3] |
The data reveals that fusion genes identified via RNA sequencing had a more pronounced effect on diagnostic refinement (97%) compared to DNA mutations (41%) [3]. This underscores the critical importance of incorporating RNA analysis in pediatric AL diagnostics, as many leukemia subtypes are defined by specific chromosomal rearrangements. Furthermore, the high proportion of targetable mutations (49%) highlights the panel's role in advancing precision medicine by identifying potential avenues for targeted therapies [3].
The Broader Diagnostic Context
The transition from conventional methods to integrated NGS testing represents a paradigm shift in pediatric oncology. The workflow below contrasts these approaches.
Diagram 2: Comparison of conventional versus integrated NGS diagnostic workflows.
- Conventional Techniques: Traditional diagnostic pathways rely on multiple, sequential tests—karyotyping, FISH, RT-PCR, and Sanger sequencing—which are labor-intensive, time-consuming, and may miss cryptic or novel alterations [3] [28].
- Targeted NGS Panels: The AmpliSeq panel consolidates these tests into a single, high-throughput assay. This integrated approach is particularly valuable for pediatric AL, which has a relatively low mutational burden but where the alterations present are generally clinically relevant [3] [13]. The panel's design specifically includes genes relevant to pediatric cancers, unlike many adult-focused panels, ensuring comprehensive coverage of childhood-specific alterations [3].
Essential Research Reagent Solutions
Successful implementation of the AmpliSeq Childhood Cancer Panel in a research setting requires several key reagents and accessories. The following table details the essential components.
Table 3: Key Research Reagent Solutions for the AmpliSeq Workflow
| Reagent / Accessory | Catalog Number Example | Function in the Workflow |
|---|---|---|
| AmpliSeq Childhood Cancer Panel | 20028446 [7] | Core primer pool for targeting 203 genes associated with childhood cancer. |
| AmpliSeq Library PLUS | 20019101 (24 reactions) [7] | Master mix and enzymes for library preparation. |
| AmpliSeq CD Indexes | Set A-D available [7] | Unique barcode adapters for multiplexing samples in a single sequencing run. |
| AmpliSeq cDNA Synthesis for Illumina | 20022654 [7] | Converts total RNA to cDNA, a required step for RNA (fusion gene) analysis. |
| AmpliSeq Library Equalizer | 20019171 [7] | Bead-based reagent for normalizing libraries prior to pooling, ensuring balanced sequencing. |
| AmpliSeq for Illumina Direct FFPE DNA | 20023378 [7] | Enables DNA preparation directly from FFPE tissues without deparaffinization or purification. |
The AmpliSeq for Illumina Childhood Cancer Panel represents a significant advancement in the molecular diagnosis of pediatric acute leukemia. Technical validation data confirms its high sensitivity, specificity, and robustness with a mean read depth >1000x [3]. Most importantly, its clinical application demonstrates a direct impact on patient management, delivering clinically relevant findings for 43% of tested patients and refining diagnosis in a substantial proportion of cases through the detection of key fusions and mutations [3].
For researchers and clinicians, this panel offers a streamlined, consolidated workflow that replaces multiple conventional tests, saving both time and precious sample material. Its focused design on pediatric cancer genes makes it a more appropriate tool than many adult-oriented panels. As the field of pediatric oncology continues to move towards precision medicine, the integration of such targeted NGS panels into routine diagnostic practice is not just beneficial—it is becoming indispensable for optimizing patient stratification and treatment strategies.
Troubleshooting Sequencing Runs: Strategies to Overcome Common Challenges and Optimize Metrics
The implementation of next-generation sequencing (NGS) in pediatric cancer diagnostics represents a significant advancement for precision medicine. However, researchers and clinicians often face technical challenges in achieving reliable results. This guide objectively compares the performance of the AmpliSeq for Illumina Childhood Cancer Panel against other available panels—OncoKids, CANSeqKids, and ALLseq—by examining how each addresses common pitfalls in sample quality, library preparation, and coverage uniformity. Performance data, drawn from recent validation studies, are synthesized to provide a clear comparison for professionals in research and drug development.
Sample Quality and Input Requirements
The quality and quantity of input nucleic acids are critical for successful NGS. The table below compares the minimum requirements and sample type flexibility of different pediatric cancer panels.
Table 1: Sample Quality and Input Requirements Comparison
| Panel Name | Minimum DNA Input | Minimum RNA Input | Supported Sample Types | Low-Input Performance |
|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | 10 ng [7] | 10 ng [7] | Blood, bone marrow, FFPE [7] [18] | Not specified in search results |
| OncoKids | 20 ng [29] | 20 ng [29] | Fresh, frozen, FFPE tissue [29] | Validated for low-input samples [29] |
| CANSeqKids | 5 ng (optimized) [10] | 10 ng (optimized) [10] | FFPE, bone marrow, whole blood, cell blocks [10] | Optimized for 5 ng DNA input; validated with 20% neoplastic content [10] |
| ALLseq | 200 ng (total library pool) [30] | Included in library pool | Not specified | Not specified |
The AmpliSeq panel demonstrates flexibility, supporting blood, bone marrow, and FFPE samples with a low 10 ng input requirement for both DNA and RNA [7]. The CANSeqKids panel shows robust performance with the lowest validated DNA input of just 5 ng and reliable results with samples containing as low as 20% neoplastic content, which is crucial for heterogeneous tumor samples [10].
Library Preparation and Workflow Efficiency
Library preparation complexity and hands-on time significantly impact laboratory throughput and potential for human error.
Table 2: Library Preparation and Workflow Comparison
| Panel Name | Library Prep Technology | Hands-On Time | Total Assay Time | Automation Compatibility |
|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | PCR-based amplicon [3] [7] | < 1.5 hours [7] | 5-6 hours (library prep only) [7] | Liquid handling robots [7] |
| OncoKids | Ion AmpliSeq (Amplicon) [29] | Not specified | Not specified | Not specified |
| CANSeqKids | Amplicon (Oncomine Childhood Cancer Research Assay) [10] | Not specified | Not specified | Automated Ion Chef process available [10] |
| ALLseq | Ampliseq-based custom design [30] | Not specified | Not specified | Not specified |
The AmpliSeq panel offers a streamlined process with less than 1.5 hours of hands-on time and compatibility with liquid handling robots, enabling laboratories to scale their operations efficiently [7]. The CANSeqKids validation study demonstrated that automation could successfully improve assay efficiency without compromising data quality [10].
Coverage Uniformity and Sequencing Metrics
Coverage uniformity ensures consistent detection of variants across all targeted regions, which is vital for confident variant calling.
Table 3: Sequencing Performance and Coverage Metrics
| Panel Name | Mean Read Depth | Coverage Uniformity | Genes Targeted | Variant Types Detected |
|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | >1000× [3] | Not specified | 203 genes [3] [7] | SNVs, Indels, CNVs, fusions [7] |
| OncoKids | Not specified | Not specified | Full spectrum for pediatric cancers [29] | Mutations, amplifications, fusions [29] |
| CANSeqKids | Not specified | Not specified | 203 unique genes [10] | SNVs, INDELs, CNVs, fusions [10] |
| ALLseq | 1903× [30] | >95% [30] | 54 genes (22 hotspot, 32 whole coding) [30] | SNVs, Indels, CNVs, fusions, expression [30] |
The AmpliSeq panel achieves a mean read depth greater than 1000×, providing a solid foundation for sensitive variant detection [3]. The ALLseq panel demonstrates exceptional performance with a mean read depth of 1903× and coverage uniformity greater than 95%, resulting in less than 1% of amplicons with poor coverage [30]. This high uniformity is critical for comprehensive assessment of all targeted regions.
Research Reagent Solutions
Successful implementation of these panels requires specific reagent kits and components. The following table details essential solutions for the AmpliSeq for Illumina Childhood Cancer Panel workflow.
Table 4: Key Research Reagent Solutions for AmpliSeq Childhood Cancer Panel
| Reagent Solution | Catalog Number Example | Function in Workflow |
|---|---|---|
| Library Preparation Kit | AmpliSeq Library PLUS for Illumina (20019101) [7] | Provides core reagents for preparing sequencing libraries; required for all samples |
| Index Adapters | AmpliSeq CD Indexes Set A-D (20031676) [7] | Enables sample multiplexing by adding unique barcode sequences to each library |
| cDNA Synthesis Kit | AmpliSeq cDNA Synthesis for Illumina (20022654) [7] | Converts RNA to cDNA for fusion detection; essential for RNA component of panel |
| Library Normalization | AmpliSeq Library Equalizer for Illumina (20019171) [7] | Simplifies library pooling by normalizing concentrations before sequencing |
| Direct FFPE DNA Prep | AmpliSeq for Illumina Direct FFPE DNA (20023378) [7] | Enables library construction from FFPE tissues without deparaffinization or DNA purification |
| Sample Identification | AmpliSeq for Illumina Sample ID Panel (20019162) [7] | Provides SNP-based genotyping for sample tracking and identification |
Experimental Protocols and Validation Data
Sensitivity and Limit of Detection
The AmpliSeq panel demonstrated a 98.5% sensitivity for DNA variants at 5% variant allele frequency (VAF) and 94.4% sensitivity for RNA fusions in a rigorous validation study [3]. The CANSeqKids panel established its limit of detection at 5% allele fraction for SNVs and INDELs, and 1,100 reads for fusion detection [10]. The ALLseq panel showed exceptional sensitivity with a limit of detection established at 2% VAF for SNVs and indels and 0.5 copy number ratio for CNVs [30].
Analytical Validation Methodologies
Validation studies followed established guidelines from the Association for Molecular Pathology (AMP) and College of American Pathologists [10]. These typically employ:
- Commercial controls with known variants at different allele frequencies [3] [10]
- Coriell HapMap samples for specificity determination [10]
- Retrospective clinical specimens with previous molecular characterization [10] [30]
- Reproducibility testing through multiple sequencing runs [3]
For the AmpliSeq panel, validation used commercial controls including SeraSeq Tumor Mutation DNA Mix and SeraSeq Myeloid Fusion RNA Mix, achieving 100% specificity and reproducibility for DNA and 89% reproducibility for RNA [3].
Workflow and Logical Relationships
This workflow diagram illustrates the logical progression from sample to result in pediatric cancer NGS testing, highlighting where common pitfalls occur and the strategic approaches different panels use to address them. The AmpliSeq panel addresses preparation inefficiency through its streamlined protocol, while CANSeqKids tackles sample quality issues with low input requirements, and ALLseq mitigates coverage dropouts with exceptional uniformity.
Each pediatric cancer NGS panel offers distinct advantages for addressing common technical challenges. The AmpliSeq for Illumina Childhood Cancer Panel provides a balanced solution with strong sensitivity, relatively fast workflow, and comprehensive gene coverage. The CANSeqKids panel excels in low-input scenarios and has demonstrated robust automation capabilities. The ALLseq panel offers exceptional coverage uniformity and sensitivity for low-frequency variants, though with a more focused gene set. Researchers should select panels based on their specific sample types, variant detection needs, and laboratory workflow requirements to optimize performance in pediatric cancer genomic studies.
The integration of next-generation sequencing (NGS) into clinical and research oncology has revolutionized diagnostic precision, particularly for pediatric cancers. Targeted panels like the AmpliSeq for Illumina Childhood Cancer Panel provide comprehensive genomic profiling by evaluating multiple variant types—including single nucleotide variants (SNVs), insertions/deletions (indels), copy number variants (CNVs), and gene fusions—across 203 genes associated with childhood and young adult cancers [7]. However, the performance of such panels is fundamentally dependent on the quantity and quality of input nucleic acids, especially when derived from challenging sources like formalin-fixed paraffin-embedded (FFPE) tissues [31].
FFPE specimens represent one of the most abundant resources in biomedical research, with over a billion samples archived worldwide in hospitals and tissue banks [31]. While offering unparalleled access to retrospective clinical material, the FFPE process introduces significant challenges for molecular analysis. Formalin fixation induces chemical cross-linking between proteins and nucleic acids, leading to DNA fragmentation and RNA damage through oxidation and other modifications [31] [32]. These artifacts can substantially impact NGS metrics including mean read depth, sensitivity, and specificity—critical performance parameters for reliable clinical assessment [3] [33]. Understanding and optimizing extraction methodologies is therefore essential for maximizing the utility of these valuable but compromised sample types in AmpliSeq Childhood Cancer Panel applications.
Comparative Analysis of FFPE Nucleic Acid Extraction Methods
Systematic Evaluation of RNA Extraction Kits
Recovery of high-quality RNA from FFPE samples remains particularly challenging due to RNA's inherent instability compared to DNA. A systematic comparison of seven commercially available FFPE RNA extraction kits revealed significant disparities in both quantity and quality of recovered RNA [31]. The study utilized nine FFPE tissue samples from three tissue types (tonsil, appendix, and B-cell lymphoma lymph node), with each extraction performed in triplicate for statistical robustness (total n=189 extractions) [31].
Table 1: Performance Comparison of Select FFPE RNA Extraction Kits
| Kit Manufacturer | Relative RNA Quantity | RNA Quality Score (RQS) | DV200 Value | Best For |
|---|---|---|---|---|
| Promega (ReliaPrep FFPE Total RNA Miniprep) | Highest yield [31] | High [31] | High [31] | Maximum recovery across most tissue types |
| Roche | Moderate [31] | Superior quality [31] | Superior [31] | Applications requiring optimal RNA integrity |
| Thermo Fisher | High (for appendix tissue) [31] | Not specified | Not specified | Tissue-specific optimization |
The evaluation identified the Promega ReliaPrep FFPE Total RNA Miniprep system as providing the best balance of quantity and quality, yielding the maximum RNA recovery for all tonsil and lymph node samples and one of the three appendix samples [31]. Notably, the Roche kit demonstrated superior qualitative performance in terms of RNA Quality Score (RQS) and DV200 values, though with lower overall yield [31]. The RQS parameter assesses RNA integrity on a scale of 1-10 (with 10 representing intact RNA), while DV200 represents the percentage of RNA fragments larger than 200 nucleotides [31]. Both metrics are crucial for predicting successful downstream sequencing applications.
DNA Extraction and Protocol Optimization
Similar challenges exist for DNA extraction from FFPE tissues. Formalin-induced fragmentation and cross-linking can dramatically reduce DNA integrity, complicating amplification-based methods like the AmpliSeq Childhood Cancer Panel [32]. A recent study evaluating the Maxwell RSC Xcelerate DNA FFPE Kit (Promega) demonstrated effective recovery of DNA with low degradation indices, yet even with favorable quantitative metrics, generation of complete short tandem repeat (STR) profiles remained challenging due to persistent fragmentation [32].
Protocol optimization can substantially improve DNA yield and integrity. Research implementing modified protocols with Qiagen QIAamp DNA FFPE kits achieved an 82% increase in DNA yield and significantly improved DNA Integrity Number (DIN) from 3.2 to 7.2 compared to manufacturer's standard protocols [34]. This enhancement involved adjustments to deparaffinization, digestion, and purification steps, demonstrating that customized approaches can dramatically improve nucleic acid recovery from suboptimal FFPE samples [34].
Table 2: Impact of Protocol Optimization on DNA Extraction from FFPE Tissues
| Parameter | Standard Protocol | Optimized Protocol | Improvement |
|---|---|---|---|
| DNA Yield | Baseline | +82% [34] | Significant |
| DNA Integrity Number (DIN) | 3.2 [34] | 7.2 [34] | Substantial |
| Suitability for NGS | Limited | High [34] | Enabled |
Specialized Solutions for FFPE Samples
The AmpliSeq ecosystem includes AmpliSeq for Illumina Direct FFPE DNA, a specialized solution designed to prepare DNA from FFPE tissues without requiring deparaffinization or DNA purification [7]. This approach streamlines the workflow and minimizes sample loss, potentially enhancing recovery from limited specimens. While comprehensive performance data relative to other extraction methods wasn't available in the search results, integration within the AmpliSeq workflow offers potential advantages for processing challenging FFPE samples.
Technical Validation of the AmpliSeq Childhood Cancer Panel with FFPE Samples
Performance Metrics with Suboptimal Samples
The AmpliSeq Childhood Cancer Panel is designed to work with minimal input material (as little as 10 ng of DNA or RNA) and is compatible with FFPE tissues, blood, bone marrow, and other low-input samples [7]. A comprehensive validation study demonstrated robust performance even with suboptimal samples, achieving a mean read depth greater than 1000×—exceeding the threshold for reliable variant detection [3] [33].
The panel exhibited high sensitivity for both DNA (98.5% for variants with 5% variant allele frequency) and RNA (94.4%), with 100% specificity and reproducibility for DNA and 89% reproducibility for RNA [3] [33]. These metrics confirm that despite the challenges of FFPE-derived nucleic acids, the panel maintains analytical performance suitable for clinical application.
Workflow and Experimental Protocol
The validated experimental workflow for the AmpliSeq Childhood Cancer Panel involves several critical steps to ensure optimal performance with FFPE-derived nucleic acids [3]:
Nucleic Acid Extraction and Quantification: DNA and RNA are extracted separately using optimized methods. The purity is assessed via spectrophotometry (OD260/280 ratio >1.8), followed by fluorometric quantification using Qubit dsDNA BR Assay Kit for DNA and RNA BR Assay Kit for RNA [3].
Quality Assessment: Integrity is evaluated using fragment analyzers such as Labchip (PerkinElmer) or TapeStation (Agilent) [3]. For FFPE-derived RNA, the DV200 metric (percentage of fragments >200 nucleotides) is particularly valuable for predicting success in downstream sequencing [31].
Library Preparation: A total of 100 ng of DNA and 100 ng of RNA (converted to cDNA using AmpliSeq cDNA Synthesis Kit) are used as input. The process employs the AmpliSeq for Illumina Childhood Cancer Panel kit with specific barcodes for each sample [3].
Sequencing: Normalized libraries are pooled at a 5:1 DNA:RNA ratio and sequenced on MiSeq or NextSeq platforms [3] [7].
This workflow diagram illustrates the optimized process for handling FFPE samples with the AmpliSeq Childhood Cancer Panel:
Impact of Input Quality on Clinical Utility
The clinical impact of molecular profiling with the AmpliSeq Childhood Cancer Panel is substantial, with 49% of mutations and 97% of fusion identifications demonstrating direct clinical relevance [3] [33]. These findings refined diagnoses in 41% of mutations and identified potentially targetable alterations in 49% of mutations [3] [33]. The high clinical impact of fusion detection (97%) underscores the critical importance of optimizing RNA quality from FFPE samples, as fusion identification typically requires intact RNA for reliable detection [3].
The Scientist's Toolkit: Essential Reagents and Methods
Successful nucleic acid extraction from FFPE samples requires specific reagents and methodologies to overcome preservation-induced damage. The following table summarizes key solutions used in the studies cited throughout this guide:
Table 3: Research Reagent Solutions for FFPE Nucleic Acid Extraction
| Reagent/Method | Function | Example Products/Protocols |
|---|---|---|
| Specialized Lysis Buffers | Degrade formalin-induced cross-links using specific enzymes and buffers [31] | Proteinase K, Heat-Induced Epitope Retrieval (HIER) buffers [31] |
| Deparaffinization Solutions | Remove paraffin embedding while preserving nucleic acids | Xylene, proprietary deparaffinization oils [31] |
| DNA Repair Enzymes | Reverse formalin-induced DNA damage and fragmentation | Enzymatic repair mixes (not specified in studies) [32] |
| Fragment Size Selection | Enrich for appropriately sized fragments for NGS | AMPure XP beads, specialized column purification [3] |
| Quality Assessment Tools | Evaluate nucleic acid integrity and suitability for NGS | Bioanalyzer, TapeStation, Qubit Fluorometer [31] [3] [34] |
Optimizing input DNA and RNA quality from FFPE tissues requires a multifaceted approach that begins with pre-analytical factors including fixation time, formalin pH (buffered formalin is superior), and storage conditions [32]. Selection of appropriate extraction methodologies significantly impacts downstream NGS performance, with the Promega ReliaPrep system showing advantages for RNA recovery and specialized FFPE DNA kits like Maxwell RSC Xcelerate providing enhanced DNA integrity [31] [32].
Protocol optimization can dramatically improve yield and quality, as demonstrated by the 82% increase in DNA recovery and improved DIN scores through modification of standard Qiagen protocols [34]. Finally, the AmpliSeq Childhood Cancer Panel's robust performance with FFPE-derived nucleic acids—achieving mean read depths >1000× and high sensitivity—confirms that with appropriate extraction and quality control, even challenging samples can yield clinically actionable results [3] [33].
As NGS continues to transform pediatric cancer diagnostics, systematic approaches to nucleic acid extraction from FFPE samples will remain essential for unlocking the full potential of archival specimens in both research and clinical settings.
Balancing Sequencing Depth, Panel Size, and Cost for Efficient Resource Allocation
In the realm of pediatric cancer genomics, effective resource allocation hinges on optimizing three interdependent parameters: sequencing depth, panel size, and cost. Targeted next-generation sequencing (NGS) panels, such as the AmpliSeq for Illumina Childhood Cancer Panel, have become indispensable tools for clinical research, enabling comprehensive evaluation of somatic variants across hundreds of genes simultaneously [7]. The strategic balance of these factors directly impacts the detection sensitivity for critical variants, the scope of genomic interrogation, and the economic feasibility of implementing NGS in research settings.
The AmpliSeq Childhood Cancer Panel represents a specialized tool designed specifically for investigating childhood and young adult cancers, covering 203 genes associated with pediatric malignancies including leukemias, brain tumors, and sarcomas [7]. This panel utilizes a PCR-based amplicon sequencing approach that requires minimal hands-on time (<1.5 hours) and low input DNA or RNA (10 ng), making it particularly suitable for precious pediatric samples [7]. Understanding how to maximize the performance of this panel while maintaining cost efficiency is paramount for researchers and drug development professionals seeking to advance precision oncology for childhood cancers.
Key Metrics: Defining Depth, Coverage, and Panel Content
Sequencing Depth vs. Coverage
Although often used interchangeably, sequencing depth and coverage represent distinct quality metrics that collectively determine data reliability:
- Sequencing Depth: Refers to the number of times a specific nucleotide is read during sequencing, expressed as an average multiple (e.g., 1000×). Deeper sequencing increases confidence in variant calling, especially for detecting low-frequency variants in heterogeneous tumor samples [1].
- Sequencing Coverage: Describes the percentage of the target region sequenced at least once, ensuring comprehensive genomic assessment. Inadequate coverage creates gaps where variants may be missed entirely [1].
High depth without sufficient coverage yields confident calls on a limited subset of targets, while broad coverage with insufficient depth provides incomplete variant information across the entire panel. The AmpliSeq Childhood Cancer Panel has demonstrated robust performance with mean read depths exceeding 1000× in validation studies, enabling high sensitivity for variant detection [3].
Panel Size and Content Considerations
The AmpliSeq Childhood Cancer Panelinterrogates 203 genes specifically selected for their relevance to pediatric malignancies, incorporating multiple variant types including single nucleotide variants (SNVs), insertions-deletions (indels), gene fusions, and copy number variants (CNVs) [7]. This targeted approach contrasts with whole-genome or whole-exome sequencing by focusing computational resources and sequencing capacity on clinically actionable targets, thereby reducing per-gene costs while maximizing relevant information yield.
The panel's design includes 97 gene fusions, 82 DNA variants, 44 genes with full exon coverage, and 24 genes for CNV analysis, creating a comprehensive profile specifically tailored to pediatric cancer genomics [3]. This strategic gene selection exemplifies how appropriate panel sizing balances comprehensiveness with practical sequencing constraints.
Table 1: Technical Specifications of the AmpliSeq Childhood Cancer Panel
| Parameter | Specification | Performance Implications |
|---|---|---|
| Number of Genes | 203 genes | Balanced scope for pediatric cancers |
| Variant Types | SNVs, indels, fusions, CNVs | Comprehensive variant profiling |
| Input Requirements | 10 ng DNA or RNA | Suitable for limited pediatric samples |
| Hands-on Time | <1.5 hours | Streamlined workflow efficiency |
| Assay Time | 5-6 hours (library prep) | Rapid turnaround for clinical research |
| Mean Read Depth | >1000× | High sensitivity for variant detection |
Experimental Approaches for Performance Benchmarking
Reference Materials and Validation Protocols
Robust performance assessment of targeted sequencing panels requires well-characterized reference materials and standardized benchmarking approaches. The National Institute of Standards and Technology (NIST) has developed Genome in a Bottle (GIAB) reference materials from extensively characterized human genomes, which provide high-confidence "truth sets" for variant calling accuracy evaluation [35].
Experimental validation of the AmpliSeq Childhood Cancer Panel should incorporate:
- DNA Controls: Commercially available reference standards like SeraSeq Tumor Mutation DNA Mix containing known variants at predetermined allele frequencies enable sensitivity and limit of detection determinations [3].
- RNA Controls: Synthetic RNA fusion mixes (e.g., SeraSeq Myeloid Fusion RNA Mix) validate fusion detection capabilities for clinically relevant translocations [3].
- Negative Controls: Established cell lines like NA12878 provide baseline specificity measurements by establishing false positive rates [3].
The following workflow diagram illustrates the key stages in performance benchmarking:
Performance Metrics Calculation
Standardized performance metrics enable objective comparison across different sequencing approaches and panels. The Global Alliance for Genomics and Health (GA4GH) Benchmarking Team has established sophisticated variant comparison tools and standardized metrics that include:
- Sensitivity: Calculated as TP/(TP+FN), where TP represents true positives and FN represents false negatives [35].
- Precision: Determined as TP/(TP+FP), measuring the proportion of identified variants that are true positives.
- Coverage Uniformity: Assessment of how evenly sequencing reads are distributed across target regions.
- Limit of Detection (LOD): The lowest variant allele frequency (VAF) reliably detected by the assay.
For the AmpliSeq Childhood Cancer Panel, validation studies have demonstrated 98.5% sensitivity for DNA variants with 5% VAF and 94.4% sensitivity for RNA fusions, with 100% specificity and reproducibility for DNA variants [3]. These metrics provide crucial benchmarks for evaluating the panel's performance relative to alternative approaches.
Comparative Performance Analysis
Depth and Sensitivity Relationships
Sequencing depth directly impacts detection sensitivity, particularly for low-frequency variants present in heterogeneous tumor samples. The AmpliSeq Childhood Cancer Panel's ability to achieve mean read depths exceeding 1000× enables reliable detection of variants at low allele frequencies (5% VAF) with 98.5% sensitivity [3]. This performance characteristic is critical for identifying subclonal populations in pediatric tumors that may have therapeutic implications.
Different sequencing applications require distinct depth thresholds for optimal performance:
- Rare Variant Detection: Studies seeking low-frequency variants in circulating tumor DNA or heterogeneous tumors require higher depths (>1000×) to distinguish true variants from sequencing artifacts [1].
- Common Variant Detection: Germline variant detection in homogeneous samples may achieve sufficient sensitivity at moderate depths (200-500×).
- Structural Variants: Fusion detection requires adequate junction-spanning reads, with the AmpliSeq panel demonstrating 94.4% sensitivity for RNA fusions [3].
Alternative NGS Approaches
The AmpliSeq Childhood Cancer Panel represents one approach within a broader ecosystem of NGS technologies. Alternative strategies include:
- Hybrid Capture Panels: Techniques like the TruSight Rapid Capture kit use oligo probes to capture regions of interest, typically requiring more input DNA and longer preparation times but offering flexible target regions [35].
- Comprehensive Panels: Assays like OncoKids provide broader coverage of pediatric malignancies with DNA content covering 44 cancer predisposition loci, mutation hotspots in 82 genes, and amplification events in 24 genes, plus 1421 targeted gene fusions in RNA [11].
- Customizable Platforms: Ion AmpliSeq On-Demand panels enable researchers to design custom panels from over 5,000 pretested genes, offering flexibility for specific research questions in germline analysis [36].
Table 2: Comparative Analysis of Targeted NGS Approaches for Pediatric Cancers
| Parameter | AmpliSeq Childhood Cancer Panel | OncoKids Panel | Ion AmpliSeq On-Demand |
|---|---|---|---|
| Technology | PCR-based amplicon sequencing | Amplification-based NGS | Ultrahigh multiplex PCR |
| Target Content | 203 genes | 44 full genes + 82 hotspots + 24 CNV + 1421 fusions | Customizable (1-500 genes) |
| Input Requirements | 10 ng DNA or RNA | 20 ng DNA and RNA | Varies by panel size |
| Hands-on Time | <1.5 hours | Not specified | Streamlined workflow |
| Variant Types | SNVs, indels, fusions, CNVs | SNVs, indels, fusions, CNVs | SNPs, indels, CNVs, fusions |
| Clinical Utility in AL | 43% of patients with clinically relevant findings | Not specified | Germline applications only |
Cost-Benefit Analysis and Resource Allocation
Economic Considerations in NGS Testing
The economic evaluation of NGS technologies involves multiple methodological approaches, with significant variability in cost calculations based on included components. Comprehensive cost assessment should incorporate:
- Direct Testing Costs: Reagents, consumables, and sequencing reagents specifically required for the assay.
- Instrumentation and Infrastructure: Equipment amortization, maintenance, and facility operations.
- Personnel Requirements: Technical time for library preparation, sequencing, and data analysis.
- Downstream Analyses: Bioinformatics pipeline implementation, data storage, and interpretation.
Recent systematic reviews indicate that targeted panel sequencing (2-52 genes) demonstrates cost-effectiveness when assessing four or more genes, while larger panels (hundreds of genes) may not consistently show favorable cost-benefit ratios across all applications [37]. The AmpliSeq Childhood Cancer Panel's focused 203-gene content positions it strategically within this economic landscape, offering substantial genomic content while remaining more targeted than comprehensive whole-exome or whole-genome approaches.
Strategic Resource Allocation Framework
Optimal resource allocation requires balancing informational yield with practical constraints. The following decision framework illustrates key considerations:
Strategic decisions should prioritize:
- Clinical Actionability: Focusing on genes with established diagnostic, prognostic, or therapeutic implications maximizes clinical utility. For the AmpliSeq Childhood Cancer Panel, 49% of identified mutations and 97% of fusions demonstrated clinical impact in validation studies [3].
- Sample Limitations: Pediatric samples often have quantity limitations, favoring approaches with low input requirements like the AmpliSeq panel (10 ng) [7].
- Operational Efficiency: Hands-on time and total assay time significantly impact laboratory workflow and staffing requirements.
Essential Research Reagent Solutions
Successful implementation of targeted NGS requires specific reagents and materials to ensure robust performance. The following table outlines key components for the AmpliSeq Childhood Cancer Panel workflow:
Table 3: Essential Research Reagents for AmpliSeq Childhood Cancer Panel Implementation
| Reagent Category | Specific Product Examples | Function in Workflow |
|---|---|---|
| Library Preparation | AmpliSeq Library PLUS | Provides reagents for preparing sequencing libraries |
| Index Adapters | AmpliSeq CD Indexes Sets A-D | Enables sample multiplexing with unique barcodes |
| RNA Conversion | AmpliSeq cDNA Synthesis for Illumina | Converts RNA to cDNA for fusion detection |
| Library Normalization | AmpliSeq Library Equalizer for Illumina | Normalizes libraries for balanced sequencing |
| FFPE Optimization | AmpliSeq for Illumina Direct FFPE DNA | Enables DNA preparation from FFPE tissues without purification |
| Quality Control | Bioanalyzer High Sensitivity DNA Chip, Qubit Assays | Assesses library quality and quantity before sequencing |
| Sequencing Controls | SeraSeq Tumor Mutation DNA Mix, Myeloid Fusion RNA Mix | Validates assay performance and establishes sensitivity |
Balancing sequencing depth, panel size, and cost requires careful consideration of research objectives, sample characteristics, and clinical applications. The AmpliSeq Childhood Cancer Panel represents a optimized solution for pediatric cancer genomics, offering comprehensive coverage of relevant targets with demonstrated sensitivity and specificity. Validation studies confirm its utility in clinical research settings, with 43% of pediatric acute leukemia patients showing clinically relevant findings that refined diagnosis, prognosis, or treatment approaches [3].
For researchers implementing this technology, key recommendations include:
- Depth Optimization: Target mean read depths >1000× for reliable detection of variants at 5% VAF in heterogeneous samples.
- Quality Assurance: Incorporate standardized reference materials and control samples to continuously monitor assay performance.
- Economic Evaluation: Consider holistic testing costs beyond reagent expenses, including personnel time, data analysis, and clinical interpretation.
- Clinical Utility Focus: Prioritize genes and variants with established impact on patient management to maximize the translational potential of sequencing findings.
As NGS technologies continue to evolve, ongoing assessment of the balance between sequencing parameters and costs will enable increasingly efficient resource allocation in pediatric cancer research.
Next-generation sequencing (NGS) has redefined diagnostic and prognostic strategies for pediatric cancers, with targeted panels like the AmpliSeq for Illumina Childhood Cancer Panel being integrated into clinical practice. However, the journey from sample to sequence is fraught with potential pitfalls, notably low coverage regions and sample failure, which can compromise data integrity. Within the broader thesis on performance metrics for AmpliSeq Childhood Cancer Panel mean read depth research, this guide objectively compares the panel's performance in managing these QC challenges, providing a detailed framework for validation, troubleshooting, and data interpretation to ensure reliable results in a clinical research setting.
Performance Metrics and Experimental Validation of the AmpliSeq Childhood Cancer Panel
Rigorous experimental validation is fundamental for establishing the reliability of any NGS panel. A 2022 study provides critical performance data for the AmpliSeq Childhood Cancer Panel, which targets 203 genes and includes DNA analysis for SNVs, Indels, and CNVs, plus RNA analysis for fusion genes [38].
Key Experimental Protocols from Validation Studies
The following table summarizes the core methodologies employed in the validation of the panel:
Table 1: Experimental Protocols for Panel Validation
| Validation Aspect | Detailed Methodology | Source/Materials Used |
|---|---|---|
| Sample Selection | 76 pediatric AL patients (51 BCP-ALL, 11 T-ALL, 14 AML); non-consecutive samples prioritized for those with non-defining conventional genetics [38]. | Hospital Sant Joan de Déu and collaborating centers [38]. |
| Nucleic Acid Extraction | DNA: Gentra Puregene kit, QIAamp DNA Mini/Micro Kit (Qiagen). RNA: Guanidine thiocyanate-phenol-chloroform (TriPure, Roche) or column-based (Direct-zol, Zymo Research) [38]. | Various commercial kits [38]. |
| Library Preparation & Sequencing | 100 ng each of DNA and RNA (converted to cDNA). Libraries prepared with panel-specific reagents, pooled at a 5:1 DNA:RNA ratio, sequenced on MiSeq [38]. | AmpliSeq for Illumina Childhood Cancer Panel Kit; MiSeq Sequencer [38]. |
| Sensitivity & LOD Assessment | DNA: SeraSeq Tumor Mutation DNA Mix (AF10%). RNA: SeraSeq Myeloid Fusion RNA Mix. Specificity assessed with Coriell NA12878 (DNA) and Invivoscribe IVS-0035 (RNA) [38]. | Commercial reference standards (SeraCare, Coriell, Invivoscribe) [38]. |
| Data Analysis | Variant calling via DNA Amplicon App; fusion calling via RNA Amplicon App; CNV analysis with OncoCNV caller in BaseSpace Sequence Hub or Local Run Manager [39]. | BaseSpace Sequence Hub, Local Run Manager [39]. |
Quantitative Performance Data
The validation study yielded the following key performance metrics, which set the benchmark for expected performance and aid in interpreting QC flags.
Table 2: Key Performance Metrics from Validation Studies
| Metric | Reported Performance | Experimental Context |
|---|---|---|
| Mean Read Depth | >1000x [38]. | Achieved across the panel during validation [38]. |
| DNA Sensitivity | 98.5% for variants at 5% VAF [38]. | Assessed using commercial positive controls [38]. |
| RNA Sensitivity | 94.4% [38]. | Assessed for fusion detection using RNA controls [38]. |
| Specificity | 100% for DNA and RNA [38]. | No false positives detected in negative controls [38]. |
| Reproducibility | 100% for DNA, 89% for RNA [38]. | Measured through replicate experiments [38]. |
| Input Quantity | 10 ng high-quality DNA or RNA [7]. | Manufacturer's minimum specification [7]. |
| On-Target Bases | Metric reflects amplicon design success and mapping to target regions [39]. | Key QC metric reported in analysis software [39]. |
A Framework for Interpreting QC Flags and Managing Low Coverage
Quality control is a continuous process throughout the NGS workflow. The Next-Generation Sequencing Quality Initiative (NGS QI) provides a structured approach, emphasizing Quality System Essentials (QSE) for personnel, equipment, and process management [40]. Adhering to this framework is key to proactively managing QC issues.
The following diagram illustrates a systematic workflow for investigating and resolving common QC flags related to sample failure and low coverage.
Troubleshooting Low Coverage Regions
Low coverage in specific regions can lead to missed variants. The AmpliSeq panel's amplicon-based design is generally robust, but issues can arise.
- Amplicon-Specific Dropouts: Primer-binding sites can be affected by known or novel SNPs, leading to inefficient amplification. Consult panel design files and cross-reference with population variant databases.
- Suboptimal Sample Quality: While the panel works with 10 ng of input [7], degraded DNA or RNA can cause uneven coverage. Use fluorometric quantification (e.g., Qubit) and integrity assessment (e.g., TapeStation) to ensure input quality surpasses the minimum requirements [38].
- Sequencing Depth and Sample Multiplexing: If overall mean depth is low, consider reducing the number of samples pooled per run. You can manipulate coverage by "increasing sequencing throughput... or reducing the number of samples pooled per run" [39].
Comparison with Alternative Pediatric Cancer NGS Panels
While the AmpliSeq Childhood Cancer Panel is a key tool, other panels like the OncoKids panel offer alternatives. The following table provides a high-level comparison based on published validation data.
Table 3: Comparison of Pediatric Cancer NGS Panels
| Feature | AmpliSeq for Illumina Childhood Cancer Panel | OncoKids NGS Panel |
|---|---|---|
| Variant Types | SNPs, Indels, CNVs, Gene Fusions [38] [7] | SNVs, Indels, CNVs, Gene Fusions [11] |
| DNA Targets | 82 genes (hotspots), 44 genes (full exon), 24 genes (CNVs) [38] | 44 genes (full coding), 82 genes (hotspots), 24 genes (amplification) [11] |
| RNA Targets | 97 gene fusions [38] | 1421 targeted gene fusions [11] |
| Input Requirements | 10 ng DNA and RNA [7] | 20 ng DNA and RNA [11] |
| Reported Sensitivity (DNA) | 98.5% (5% VAF) [38] | Robust (specific % not stated) [11] |
| Reported Mean Read Depth | >1000x [38] | Not explicitly stated |
| Sample Compatibility | Blood, Bone Marrow, FFPE [7] | FFPE, Frozen Tissue, Bone Marrow, Blood [11] |
| Key Differentiator | Integrated Illumina workflow with BaseSpace analysis [7] [39] | Designed for a broad spectrum of pediatric solid tumors and leukemias [11] |
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation and troubleshooting of the AmpliSeq panel depend on several key reagents and tools.
Table 4: Essential Research Reagents and Tools
| Item | Function | Example Product/Citation |
|---|---|---|
| Commercial Reference Standards | Assess assay sensitivity, specificity, and LOD using known variants [38]. | SeraSeq Tumor Mutation DNA Mix; SeraSeq Myeloid Fusion RNA Mix [38]. |
| Negative Control Materials | Establish baseline specificity and detect contamination [38]. | NA12878 (Coriell) for DNA; IVS-0035 (Invivoscribe) for RNA [38]. |
| Nucleic Acid Quantification Kits | Fluorometric quantification for accurate input measurement [38]. | Qubit dsDNA BR Assay Kit; Qubit RNA BR Assay Kit [38]. |
| Library Prep Reagents | Generate amplicon libraries with sample-specific barcodes [7]. | AmpliSeq Library PLUS for Illumina; AmpliSeq CD Indexes [7]. |
| cDNA Synthesis Kit | Converts input RNA to cDNA for fusion gene analysis [7]. | AmpliSeq cDNA Synthesis for Illumina [7]. |
| Bioinformatic QC Tools | Assess raw data quality, adapter contamination, and base quality [41]. | FastQC, Trimmomatic, Cutadapt [41]. |
| Analysis Suites | Perform alignment, variant calling, fusion calling, and CNV analysis [39]. | BaseSpace Sequence Hub (DNA/RNA Amplicon Apps), Local Run Manager [39]. |
Best Practices for QC and Data Analysis
Consistent application of best practices is the most effective strategy for preventing and managing QC issues.
- Implement Longitudinal Tracking: Use tools like the NGS QI's "Identifying and Monitoring NGS Key Performance Indicators SOP" to track metrics like mean read depth and on-target rate over time, enabling early detection of workflow drift [40].
- Conduct QC at Every Stage: Perform QC checks after nucleic acid extraction, library preparation, and sequencing. Using multiple QC tools (e.g., FastQC, Trimmomatic) increases the sensitivity of problem identification [41].
- Validate and Lock Down Workflows: Once a workflow is validated against performance benchmarks, it should be locked down. Any change, from reagent lot to software version, requires re-validation to ensure consistent performance [40].
Interpreting QC flags for the AmpliSeq Childhood Cancer Panel requires a deep understanding of its validated performance metrics and a systematic approach to troubleshooting. The panel demonstrates high sensitivity, specificity, and robust mean read depth (>1000x), providing a reliable foundation for pediatric cancer research [38]. When compared to alternatives like OncoKids, the choice often hinges on specific gene content and institutional workflow preferences. Ultimately, by adhering to rigorous quality management systems, employing recommended reagent solutions, and implementing longitudinal performance tracking, researchers can confidently manage low coverage regions and failed samples, ensuring the generation of clinically actionable genomic data.
Comparative Analysis and Clinical Validation: How the Panel Stacks Up Against Other Methods
Next-generation sequencing (NGS) has revolutionized genomic research, and targeted enrichment strategies are crucial for focusing sequencing efforts on regions of interest. Two primary methods—amplicon-based and hybridization-capture—dominate the field, each with distinct technical advantages and limitations. Understanding their performance characteristics is essential for researchers, particularly those utilizing panels like the AmpliSeq Childhood Cancer Panel for pediatric cancer research. This guide provides an objective comparison of these methodologies, supported by experimental data and performance metrics relevant to scientific and drug development professionals.
Performance Metrics Comparison
Extensive evaluations reveal consistent differences in performance metrics between amplicon-based and hybridization-capture methods. The table below summarizes key quantitative findings from published concordance studies.
Table 1: Comparative Performance Metrics of Amplicon-Based and Hybridization-Capture NGS Methods
| Performance Characteristic | Amplicon-Based Methods | Hybridization-Capture Methods | Supporting Evidence |
|---|---|---|---|
| On-Target Rate | Higher (due to primer specificity) [42] [43] [44] | Variable, generally lower [42] [44] | Evaluation of whole-exome sequencing approaches [42] |
| Coverage Uniformity | Lower (due to PCR bias) [42] [44] | Superior [42] [45] [44] | Whole-exome and lymphoma panel studies [42] [45] |
| Variant Calling Sensitivity | Generally high, but may miss some variants [42] [3] | High, with superior sensitivity for low-frequency variants [45] | Childhood cancer panel validation; lymphoma study [3] [45] |
| False Positive/Variant Discordance | Higher risk due to amplification errors [42] [46] | Lower noise and fewer false positives [43] [45] | Exome comparison; SARS-CoV-2 sequencing [42] [46] |
| Workflow & Hands-On Time | Simpler, fewer steps, faster (<1.5 hours for AmpliSeq) [43] [7] [44] | More complex, multiple steps, longer [43] [44] | Technical comparisons and panel specifications [43] [7] [44] |
| DNA Input Requirement | Lower (10 ng for AmpliSeq Childhood Cancer Panel) [3] [7] | Higher (often >50 ng) [44] | Panel validation and technical guides [3] [7] [44] |
| Scalability | Limited for large panels (primer design complexity) [43] [44] | Highly scalable (virtually unlimited targets) [43] [44] | Technical and application guides [43] [44] |
Experimental Data and Concordance Analysis
Whole-Exome Sequencing Concordance
A comprehensive 2015 study directly compared two amplicon-based (HaloPlex, Ion AmpliSeq) and two hybridization-capture-based (SureSelect, SeqCap) whole-exome sequencing methods. While all methods identified many of the same single-nucleotide variants (SNVs), amplicon-based methods missed variants detected by the other three methods and reported additional variants discordant with all other technologies [42]. Many false positives/negatives resulted from limited coverage, low variant frequency, or vicinity to read starts/ends. The study concluded that while amplicon methods had higher raw on-target rates, hybridization capture demonstrated better uniformity and reliability [42].
Targeted Panel Performance in Oncology
Childhood Cancer Panel Validation
A 2022 validation of the AmpliSeq for Illumina Childhood Cancer Panel (an amplicon-based method) reported excellent performance metrics. The panel achieved a mean read depth greater than 1000×, with 98.5% sensitivity for DNA variants at 5% variant allele frequency (VAF) and 94.4% sensitivity for RNA fusions. The method demonstrated 100% specificity and reproducibility for DNA and 89% reproducibility for RNA. The study successfully implemented the panel for clinical utility, finding clinically relevant results in 43% of pediatric acute leukemia patients [3].
Table 2: Key Reagent Solutions for Targeted NGS Validation
| Research Reagent | Function in Validation | Example Product/Source |
|---|---|---|
| Reference DNA Materials | Provides benchmark for specificity/sensitivity | Coriell HapMap samples (NA12878); SeraSeq Tumor Mutation DNA Mix [35] [3] [10] |
| RNA Fusion Controls | Validates fusion detection sensitivity | SeraSeq Myeloid Fusion RNA Mix [3] |
| Library Preparation Kits | Converts nucleic acids to sequence-ready libraries | AmpliSeq Library PLUS for Illumina; Illumina DNA Prep Kit [3] [46] |
| Target Enrichment Panels | Selects genomic regions of interest | AmpliSeq Childhood Cancer Panel; TruSight Rapid Capture kit [35] [3] [7] |
| Sequence Adapters & Indexes | Enables sample multiplexing and sequencing | AmpliSeq CD Indexes for Illumina [7] |
| Nucleic Acid Quantification Kits | Ensures accurate input measurement | Qubit dsDNA HS Assay Kit; TapeStation System [42] [3] |
Lymphoma Panel Comparison
A 2017 study compared capture hybridization and amplicon sequencing using a 32-gene panel for lymphoma. Researchers found hybridization capture superior to amplicon sequencing by providing deeper, more uniform coverage and yielding higher sensitivity for variant calling. The study established that hybridization capture provided better performance for detecting actionable mutations in formalin-fixed, paraffin-embedded (FFPE) tumor samples [45].
Methodological Workflows
The fundamental difference between these methods lies in their approach to target enrichment. The diagram below illustrates the key steps in each workflow.
Both amplicon-based and hybridization-capture methods offer distinct advantages for targeted NGS. Amplicon-based approaches like the AmpliSeq Childhood Cancer Panel provide an excellent balance of speed, simplicity, and low input requirements, making them ideal for focused panels where high sensitivity for known targets is paramount [3] [7]. Hybridization-capture methods offer superior coverage uniformity, scalability for larger genomic regions, and fewer false positives, making them better suited for comprehensive profiling studies [42] [45] [44].
The choice between these methodologies ultimately depends on research objectives, target size, sample quality, and resource constraints. For pediatric cancer research using the AmpliSeq Childhood Cancer Panel, the demonstrated high sensitivity, reproducibility, and clinical utility support its use for targeted molecular profiling, while researchers requiring broader genomic coverage may benefit from hybridization-capture alternatives.
The molecular characterization of pediatric acute leukemia (AL) is fundamental for refining diagnosis, prognosis, and treatment strategies. Next-generation sequencing (NGS) has revolutionized this process by enabling the parallel analysis of numerous genetic alterations. The AmpliSeq for Illumina Childhood Cancer Panel is a targeted NGS solution designed specifically for the genomic landscape of childhood and young adult cancers. This panel analyzes 203 genes, covering multiple variant types including gene fusions, single nucleotide variants (SNVs), insertions/deletions (InDels), and copy number variants (CNVs) [3] [7]. A pivotal validation study demonstrated its profound clinical utility, showing that 41% of identified mutations refined diagnostic classification and 49% were considered targetable, indicating potential for directed therapies [3] [33] [13]. This guide objectively compares the performance of this panel within the context of pediatric AL diagnostics and research.
Performance Metrics and Clinical Utility Data
The clinical utility of a diagnostic test is defined as the likelihood that its results will inform and support clinical decisions that improve patient outcomes [47]. For the AmpliSeq Childhood Cancer Panel, this was quantitatively assessed in a cohort of 76 pediatric patients with AL.
The table below summarizes the key performance and clinical utility metrics from the validation study:
| Metric Category | Specific Parameter | Performance / Impact Data |
|---|---|---|
| Sequencing Performance | Mean Read Depth | > 1000x [3] [13] |
| DNA Sensitivity (5% VAF) | 98.5% [3] [33] | |
| RNA Sensitivity | 94.4% [3] [33] | |
| Specificity & Reproducibility (DNA) | 100% [3] [33] | |
| Overall Clinical Utility | Patients with Clinically Relevant Findings | 43% [3] [33] [13] |
| Impact of DNA Mutations | Mutations Refining Diagnosis | 41% [3] [33] [13] |
| Mutations with Targetable Findings | 49% [3] [33] [13] | |
| Impact of RNA Fusions | Fusion Genes Refining Diagnosis | 97% [3] [33] [13] |
Technical Comparison with Alternative Panels
The following table compares the technical specifications of the AmpliSeq Childhood Cancer Panel with another pediatric-focused panel, OncoKids.
| Specification | AmpliSeq for Illumina Childhood Cancer Panel | OncoKids Panel |
|---|---|---|
| Total Genes | 203 genes [3] [7] | 206 genes (44 full exon, 82 hotspots, 24 CNVs) [11] |
| Variant Types | SNVs, InDels, CNVs, Gene Fusions [3] [7] | SNVs, InDels, CNVs, Gene Fusions [11] |
| DNA Input | 100 ng (library prep) [3] / 10 ng [7] | 20 ng [11] |
| RNA Input | 100 ng (library prep) [3] / 10 ng [7] | 20 ng [11] |
| Fusion Coverage | 97 gene fusions [3] | 1421 targeted gene fusions [11] |
| Sample Compatibility | Blood, Bone Marrow, FFPE [7] | FFPE, Frozen Tissue, Bone Marrow, Blood [11] |
Experimental Protocols and Methodologies
The validation of the AmpliSeq Childhood Cancer Panel followed a rigorous protocol to establish its analytical and clinical utility in a pediatric AL cohort [3] [13].
Sample Selection and Conventional Techniques
- Cohort: 76 pediatric patients diagnosed with B-cell precursor ALL (n=51), T-ALL (n=11), and AML (n=14) were selected from multiple Spanish centers (2016-2020). Selection prioritized patients with non-defining genetic results from conventional methods [3] [13].
- Control Materials: Commercial controls were used for validation. SeraSeq Tumor Mutation DNA Mix was the DNA positive control, and SeraSeq Myeloid Fusion RNA Mix was the RNA positive control [3].
- Conventional Methods: The mutational status of genes like
FLT3andNPM1was assessed by labeled-PCR amplification, whileCKITandGATA1mutations were tested by Sanger sequencing. Fusion genes were studied by quantitative RT-PCR using Europe Against Cancer Program guidelines [3] [13]. These conventional results served as a benchmark for the NGS panel.
Library Preparation and Sequencing Workflow
The integrated experimental workflow for the panel is summarized in the diagram below.
- Nucleic Acid Extraction: DNA was extracted using Qiagen kits (e.g., Gentra Puregene, QIAamp DNA Mini Kit). RNA was extracted via guanidine thiocyanate-phenol-chloroform or column-based methods. Purity (OD260/280 >1.8) and integrity were rigorously checked [3] [13].
- Library Preparation: For DNA, 100 ng was used to generate 3069 amplicons. For RNA, 100 ng was reverse-transcribed to cDNA and then used to generate 1701 amplicons targeting gene fusions. Libraries were prepared with sample-specific barcodes [3].
- Sequencing: DNA and RNA libraries were pooled at a 5:1 ratio, diluted to 17–20 pM, and sequenced on an Illumina MiSeq sequencer [3].
Signaling Pathways and Clinical Decision Impact
The genetic alterations detected by the panel directly influence clinical decision-making through defined biological pathways and clinical actionability. The pathway from genetic variant to clinical impact is illustrated below.
The panel's high clinical utility stems from its design, which focuses on genes with established roles in oncogenesis and treatment response. The identified variants can be categorized based on their clinical impact:
- Diagnostic Refinement: The panel identified genetic alterations that directly refine leukemia classification. For instance, fusion genes like
BCR::ABL1,RUNX1::RUNX1T1, andPML::RARAare defining lesions for specific leukemia subtypes and were detected with high sensitivity [3]. This is critical as accurate classification directly influences the choice of therapy protocols. - Targetable Findings: Nearly half of the mutations identified were considered "targetable," meaning they point to potential therapies. Examples include mutations in genes like
FLT3, which may indicate the use of FLT3 inhibitors, or other kinase pathway mutations that have available targeted drugs [3] [33]. This directly enables a precision medicine approach.
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details essential materials and reagents used in the featured validation study [3] [7] that are crucial for researchers seeking to replicate or implement this workflow.
| Research Reagent / Kit | Function in the Workflow |
|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel | Core panel containing primers to amplify targets across 203 genes for DNA and RNA analysis [3] [7]. |
| AmpliSeq Library PLUS for Illumina | Reagents for preparing sequencing libraries from the amplified PCR products [7]. |
| AmpliSeq CD Indexes for Illumina | Unique barcode sequences used to label individual samples, enabling multiplexed sequencing [7]. |
| AmpliSeq cDNA Synthesis for Illumina | Enzyme mix to convert total RNA to cDNA, a required step before RNA library preparation [7]. |
| SeraSeq Tumor Mutation DNA Mix | Multiplexed biosynthetic positive control containing known DNA variants at specific allele frequencies for assay validation and QC [3]. |
| SeraSeq Myeloid Fusion RNA Mix | Synthetic RNA positive control containing specific fusion transcripts relevant to leukemia for RNA assay validation [3]. |
| QIAamp DNA Blood Mini Kit (Qiagen) | Used for the extraction of high-quality DNA from patient blood or bone marrow samples [3]. |
| Direct-zol RNA MiniPrep (Zymo Research) | A column-based method for the extraction of high-integrity RNA from patient samples [3]. |
The AmpliSeq for Illumina Childhood Cancer Panel represents a significant advancement in the molecular characterization of pediatric acute leukemia. The experimental data confirms that it is a technically robust assay with high sensitivity and specificity. More importantly, its demonstrated clinical utility is high, with a substantial proportion of findings leading to refined diagnosis (41% of mutations) and identification of targetable alterations (49% of mutations) [3] [33] [13]. For researchers and clinicians, this panel offers a streamlined, comprehensive alternative to multiple standalone tests, effectively integrating into the daily routine of pediatric hematology practice to support precision medicine initiatives.
The integration of next-generation sequencing (NGS) into clinical oncology requires rigorous validation of targeted gene panels to ensure reliable molecular diagnostics. For pediatric cancers, which often have low mutational burdens but clinically significant alterations, the reproducibility and precision of an assay are critical for accurate diagnosis, prognosis, and treatment planning. This guide objectively compares the performance of the AmpliSeq for Illumina Childhood Cancer Panel with other available NGS panels, focusing on key performance metrics established in validation studies. The data is presented within the broader context of performance metrics for AmpliSeq Childhood Cancer Panel mean read depth research, providing researchers and drug development professionals with a clear comparison of analytical validity.
Performance Metrics Comparison of Pediatric Cancer NGS Panels
The table below summarizes key performance metrics from analytical validation studies for several pediatric cancer NGS panels.
| Panel Name | Target Genes | Reported Reproducibility | Reported Sensitivity | Limit of Detection | Mean Read Depth |
|---|---|---|---|---|---|
| AmpliSeq for Illumina Childhood Cancer Panel [3] | 203 genes (97 fusions, 82 DNA variants, 44 full exons, 24 CNVs) [3] | DNA: 100%RNA: 89% [3] | DNA: 98.5% (at 5% VAF)RNA: 94.4% [3] | 5% VAF for DNA variants [3] | >1000× [3] |
| CANSeqTMKids (Oncomine Childhood Cancer Assay) [10] | 203 unique genes (130 for DNA, 91 for fusions) [10] | >99% (Overall Assay) [10] | >99% (Overall Assay) [10] | 5% AF for SNVs/Indels, 1,100 reads for fusions [10] | Not Specified |
| OncoKids [11] | 44 full genes, 82 hotspots, 24 CNV genes, 1421 fusions [11] | Robust performance reported [11] | Robust performance reported [11] | Not Specified | Not Specified |
Experimental Protocols for Key Validation Studies
AmpliSeq for Illumina Childhood Cancer Panel Validation
A 2022 study aimed to validate the AmpliSeq for Illumina Childhood Cancer Panel for pediatric acute leukemia (AL) diagnostics [3].
- Sample Cohort: The validation used 76 pediatric patients diagnosed with BCP-ALL (n=51), T-ALL (n=11), and AML (n=14). Samples were selected based on age (<25 years), availability of high-quality DNA/RNA, and non-defining genetic results from conventional diagnostics [3].
- Control Materials:
- DNA Sensitivity/Specificity: SeraSeq Tumor Mutation DNA Mix (v2 AF10 HC) was used as a positive control, and NA12878 (Coriell Institute) as a negative DNA control [3].
- RNA Sensitivity/Specificity: SeraSeq Myeloid Fusion RNA Mix was used as a positive control, and IVS-0035 (Invivoscribe) as a negative RNA control [3].
- Library Preparation & Sequencing: Libraries were prepared per manufacturer's instructions using 100 ng of DNA and 100 ng of RNA per sample. RNA was reverse transcribed to cDNA. DNA and RNA libraries were pooled at a 5:1 ratio and sequenced on a MiSeq sequencer [3].
- Data Analysis: The panel demonstrated 100% reproducibility for DNA and 89% reproducibility for RNA. It also showed high sensitivity (98.5% for DNA at 5% VAF and 94.4% for RNA) and 100% specificity for DNA [3].
CANSeqTMKids Panel Validation
The CANSeqTMKids assay, which uses the Oncomine Childhood Cancer Research Assay (OCCRA), was validated in 2023 for profiling childhood malignancies [10].
- Sample Cohort: The study used 65 samples including FFPE tissue (n=32), cell blocks (n=2), whole blood (n=8), bone marrow (n=4), cell lines (n=7), and commercial controls (n=12) [10].
- Library Preparation: Libraries were prepared both manually and automated on the Ion Chef system. The automated process required 15 µL of DNA at 0.7 ng/µL and 10 µL of RNA at 1 ng/µL. DNA and RNA libraries were combined at an 80:20 ratio and sequenced on the Ion GeneStudio S5 Prime system [10].
- Data Analysis: The assay determined >99% accuracy, sensitivity, and reproducibility. The limit of detection was established at 5% allele fraction for SNVs/Indels and 1,100 reads for fusion detection [10].
Workflow and Logical Relationships in Panel Validation
The following diagram illustrates the general workflow and logical relationships common to the validation of targeted NGS panels for childhood cancer.
Research Reagent Solutions for NGS Panel Validation
The table below details key reagents and materials essential for conducting NGS panel validation studies in pediatric cancer research.
| Item | Function in Validation | Specific Examples |
|---|---|---|
| Commercial Reference Standards | Validates assay sensitivity, specificity, and limit of detection using known variants at defined allele frequencies. | SeraSeq Tumor Mutation DNA Mix [3], SeraSeq Myeloid Fusion RNA Mix [3], AcroMetrix Oncology Hotspot Control [10] [48], Seraseq Fusion RNA Mix [10] |
| Library Preparation Kit | Contains reagents for amplifying target regions and attaching index adapters for sample multiplexing. | AmpliSeq Library PLUS for Illumina [7] [18], Oncomine Childhood Cancer Research Assay Chef-Ready Kit [10] |
| Index Adapters | Unique molecular barcodes added to each sample library to enable pooling and multiplexed sequencing. | AmpliSeq CD Indexes for Illumina [7] [18], IonCode Barcode Adapters [10] |
| cDNA Synthesis Kit | Converts input RNA into complementary DNA (cDNA) for subsequent library preparation of RNA targets. | AmpliSeq cDNA Synthesis for Illumina [7] [18] |
| Nucleic Acid Extraction Kits | Iserts high-quality DNA and RNA from various sample types, including FFPE tissue, blood, and bone marrow. | QIAamp DNA Mini Kit [3] [49], Direct-zol RNA MiniPrep [3], Gentra Puregene kit [3] |
| Library Normalization Beads | Streamlines the process of normalizing library concentrations before pooling, improving workflow consistency. | AmpliSeq Library Equalizer for Illumina [7], Equalizer Kit (Thermo Scientific) [10] |
Next-Generation Sequencing (NGS) has revolutionized molecular diagnostics, particularly in pediatric oncology where comprehensive genomic profiling is essential for accurate diagnosis and treatment. The AmpliSeq Childhood Cancer Panel (Illumina) represents a targeted approach designed specifically for investigating 203 genes associated with cancer in children and young adults [7]. Public health laboratories and research institutions implementing this technology face a critical decision: establishing in-house NGS capabilities versus outsourcing to specialized service providers. This analysis examines both approaches within the context of a broader thesis on performance metrics for AmpliSeq Childhood Cancer Panel mean read depth research, providing a framework for decision-making based on technical validation, operational efficiency, and economic considerations.
The choice between in-house and outsourced NGS has significant implications for research continuity, data security, cost structure, and ultimately, the advancement of personalized medicine in pediatric cancers. As validation studies demonstrate, the AmpliSeq Childhood Cancer Panel achieves a mean read depth greater than 1000× with high sensitivity (98.5% for variants with 5% variant allele frequency) and specificity (100%) when implemented under controlled conditions [3]. These performance metrics serve as crucial benchmarks for evaluating implementation success regardless of the chosen operational model.
Technical Performance of the AmpliSeq Childhood Cancer Panel
Analytical Validation Metrics
The AmpliSeq Childhood Cancer Panel has undergone rigorous technical validation in clinical research settings. A 2022 study evaluating the panel's performance established key quality metrics that define its analytical capabilities. The validation assessed multiple parameters using commercial controls and patient samples, with a focus on its application for pediatric acute leukemia diagnostics [3].
Table 1: Technical Performance Metrics of AmpliSeq Childhood Cancer Panel
| Performance Parameter | DNA Analysis | RNA Analysis |
|---|---|---|
| Mean Read Depth | >1000× | Not specified |
| Sensitivity | 98.5% (at 5% VAF) | 94.4% |
| Specificity | 100% | Not specified |
| Reproducibility | 100% | 89% |
| Limit of Detection | 5% VAF | Not specified |
The panel demonstrated substantial clinical impact in validation studies, with 49% of mutations and 97% of the identified fusions showing clinical relevance. Specifically, 41% of mutations refined diagnosis, while 49% were considered targetable for therapeutic interventions [3]. These performance characteristics establish the benchmark for implementation regardless of operational model.
Sequencing and Coverage Considerations
When implementing the AmpliSeq panel, understanding key sequencing metrics is crucial for both in-house and outsourced workflows. Sequencing depth (number of times a specific base is read) and coverage (proportion of the target region sequenced) are distinct but interrelated parameters that significantly impact variant detection capability [1].
For the AmpliSeq Childhood Cancer Panel, the achieved mean read depth of >1000× provides high confidence in variant calling, particularly important for detecting low-frequency variants in heterogeneous samples. Higher sequencing depth reduces errors and enables reliable detection of variants with lower variant allele frequencies, while comprehensive coverage ensures no critical genomic regions are missed in analysis [1].
In-House NGS Implementation
Infrastructure and Workflow Requirements
Establishing in-house NGS capabilities for the AmpliSeq Childhood Cancer Panel requires significant infrastructure investment. The core workflow involves nucleic acid extraction, library preparation using the panel-specific reagents, sequencing on compatible Illumina platforms, and bioinformatic analysis [3] [7].
Table 2: In-House Implementation Requirements for AmpliSeq Childhood Cancer Panel
| Component | Specification |
|---|---|
| Input Material | 10 ng high-quality DNA or RNA [7] |
| Library Prep Time | 5-6 hours (hands-on time <1.5 hours) [7] |
| Compatible Instruments | MiSeq, NextSeq 550, NextSeq 2000, NextSeq 1000, MiniSeq systems [7] |
| Variant Types Detected | SNPs, gene fusions, indels, copy number variants [7] |
| Specialized Sample Types | Blood, bone marrow, FFPE tissue [7] |
The library preparation employs a PCR-based protocol generating 3069 amplicons per DNA sample and 1701 amplicons per RNA sample, with average sizes of 114bp and 122bp respectively [3]. This targeted amplicon sequencing approach enables efficient coverage of coding regions across the 203 genes included in the panel.
Experimental Protocols and Validation
Implementing the AmpliSeq Childhood Cancer Panel in-house requires extensive validation following established guidelines. The technical validation study provides a methodological framework:
Sample Selection and Quality Control: The validation used commercial controls including SeraSeq Tumor Mutation DNA Mix and SeraSeq Myeloid Fusion RNA Mix, alongside clinical samples from pediatric patients with BCP-ALL (n=51), T-ALL (n=11), and AML (n=14). DNA quality was verified by spectrophotometry (OD260/280 ratio >1.8) and fluorometric quantification [3].
Library Preparation and Sequencing: Libraries were prepared following manufacturer's instructions with 100ng of DNA and RNA input. RNA was reverse transcribed to cDNA using the AmpliSeq cDNA Synthesis kit. Barcoded libraries were pooled at a 5:1 DNA:RNA ratio and sequenced on a MiSeq Sequencer [3].
Data Analysis: Bioinformatic processing involved alignment to reference genomes, variant calling with specific thresholds, and annotation. The validation established sensitivity and specificity metrics through comparison with known controls and orthogonal validation methods [3].
Outsourced NGS Sequencing Services
Service Provider Models and Capabilities
Outsourcing NGS for the AmpliSeq Childhood Cancer Panel to specialized service providers offers an alternative pathway for public health laboratories lacking infrastructure or expertise. These services typically fall into two categories: full-service providers that handle the entire workflow from sample to report, and limited-service providers that perform specific steps such as sequencing or data analysis [50] [51].
The primary advantages of outsourcing include access to expert skills and specialized equipment without capital investment, potentially faster turnaround times for individual projects, and reduced need for specialized staff training [50]. Service providers like Source Genomics highlight benefits such as fast turnaround times (as quick as 12 hours for some services), high-quality data from ISO-accredited laboratories, and comprehensive genomic profiling capabilities [51].
Considerations for Outsourcing the AmpliSeq Panel
When considering outsourcing for the AmpliSeq Childhood Cancer Panel, several factors require careful evaluation:
Technical Compatibility: Ensure the service provider has established workflows and validation data specifically for the AmpliSeq Childhood Cancer Panel, not just general NGS capabilities.
Data Quality Metrics: Request performance data including mean read depth, sensitivity for variant detection, and coverage uniformity comparable to published validations [3].
Turnaround Time: Assess the complete timeline from sample shipment to final report, accounting for shipping and potential delays.
Data Security and Ownership: Establish clear agreements regarding data storage, access, and intellectual property rights, particularly important for sensitive patient data [50] [52].
Cost Structure: Evaluate both per-sample costs and potential hidden expenses for additional analyses or data storage [50].
Comparative Analysis: In-House vs. Outsourced NGS
Operational and Economic Considerations
The decision between in-house and outsourced NGS implementation involves balancing multiple operational and economic factors that impact the total cost of ownership and research output.
Table 3: Cost-Benefit Analysis of In-House vs. Outsourced NGS
| Factor | In-House NGS | Outsourced NGS |
|---|---|---|
| Initial Investment | High (equipment, setup, validation) [50] | Low or none [50] |
| Per-Sample Cost | Lower for high volumes [50] | Potentially higher, but no equipment maintenance [50] |
| Data Control | Complete control over data and processes [50] | Limited control, potential security concerns [50] |
| Turnaround Time | Potentially faster for routine batches [50] | Possibly faster for one-off projects [50] |
| Expertise Requirements | Requires specialized staff and training [50] | Access to provider's expertise [50] [51] |
| Scalability | Limited by existing capacity | Easily scalable for large projects [50] |
| Quality Control | Direct oversight of all processes | Dependent on provider's standards [51] |
The economic analysis must account for both direct costs (reagents, equipment, service fees) and indirect costs (personnel time, data storage, facility overhead). For laboratories processing high sample volumes consistently, in-house implementation typically offers lower long-term costs, while outsourcing provides financial predictability and avoids capital depreciation [50].
Implementation Decision Framework
The choice between in-house and outsourced NGS for the AmpliSeq Childhood Cancer Panel should be guided by a structured decision framework that aligns with the institution's strategic goals and operational capabilities. The following workflow diagram illustrates the key decision points:
Figure 1: Decision workflow for implementing AmpliSeq Childhood Cancer Panel sequencing.
For public health contexts with ongoing surveillance needs or large research cohorts, a hybrid approach may be optimal: establishing core in-house capabilities for routine analyses while outsourcing specialized or peak-load projects. This model balances control and flexibility while managing operational risks [50].
Essential Research Reagent Solutions
Successful implementation of the AmpliSeq Childhood Cancer Panel, whether in-house or through a service provider, requires specific reagents and accessories to ensure optimal performance and data quality.
Table 4: Essential Research Reagents for AmpliSeq Childhood Cancer Panel
| Reagent Solution | Function | Implementation Context |
|---|---|---|
| AmpliSeq Library PLUS | Provides reagents for library preparation (24, 96, or 384 reactions) [7] | Required for both in-house and outsourced workflows |
| AmpliSeq CD Indexes | Unique barcodes for sample multiplexing [7] | Essential for in-house; managed by service providers |
| AmpliSeq cDNA Synthesis Kit | Converts total RNA to cDNA for RNA fusion detection [7] | Critical for RNA component analysis |
| AmpliSeq Library Equalizer | Normalizes libraries before sequencing [7] | Important for in-house workflow optimization |
| AmpliSeq for Illumina Direct FFPE | Enables library construction from FFPE tissues without DNA purification [7] | Valuable for retrospective sample analysis |
| Quality Control Kits | Assess nucleic acid quality and quantity (e.g., Qubit, TapeStation) [3] | Essential pre-sequencing step for both models |
These reagent solutions form the foundation of reliable AmpliSeq Childhood Cancer Panel implementation. For in-house workflows, maintaining adequate inventory of these reagents while managing shelf-life constraints requires careful supply chain management [53]. When outsourcing, verifying that the service provider uses validated, quality-assured reagents is essential for result comparability.
The decision between in-house implementation and outsourcing of AmpliSeq Childhood Cancer Panel sequencing represents a strategic choice that should be guided by institutional priorities, resources, and research objectives. For public health laboratories with sustained sample flow, specialized personnel, and data security requirements, in-house implementation offers greater control, lower long-term costs, and operational independence. For institutions with fluctuating project needs, limited capital resources, or specialized analysis requirements, outsourcing provides flexibility, access to expertise, and faster implementation.
Technical validation data demonstrates that the AmpliSeq Childhood Cancer Panel can achieve excellent performance metrics in both operational models when properly implemented [3]. The mean read depth of >1000×, high sensitivity and specificity, and demonstrated clinical utility establish this targeted panel as a valuable tool for pediatric cancer genomics. Ultimately, the optimal implementation model maximizes both scientific impact and operational efficiency within the specific constraints of the public health context, advancing the goal of precision medicine for childhood cancers.
Conclusion
The robust performance of the AmpliSeq Childhood Cancer Panel, characterized by a mean read depth exceeding 1000x and high sensitivity, firmly establishes its reliability for integrating molecular characterization into pediatric hematology and oncology practice. The validation data demonstrates that this targeted NGS approach successfully refines diagnosis and identifies actionable targets in a significant proportion of patients, moving the field closer to personalized treatment. Future directions should focus on the widespread implementation of such validated, cost-effective panels within public health systems, the continued expansion of pediatric-specific genomic knowledge, and the development of automated bioinformatics pipelines to further accelerate the delivery of precision medicine to young cancer patients globally.
References
- 1. Sequencing Depth vs. Coverage: Key Metrics in NGS ... [https://3billion.io/blog/sequencing-depth-vs-coverage]
- 2. Decoding Sequencing Depth and Coverage [https://www.cd-genomics.com/resource-sequencing-depth-and-coverage.html]
- 3. Technical Validation and Clinical Utility of an NGS ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9021638/]
- 4. Sequencing Coverage for NGS Experiments [https://www.illumina.com/science/technology/next-generation-sequencing/plan-experiments/coverage.html]
- 5. Sequencing depth and coverage: key considerations in ... [https://www.nature.com/articles/nrg3642]
- 6. Making the Most of Your NGS Data: Understanding Metrics ... [https://sequencing.roche.com/us/en/article-listing/metrics-for-target-enriched-ngs.html]
- 7. AmpliSeq for Illumina Childhood Cancer Panel [https://www.illumina.com/products/by-type/sequencing-kits/library-prep-kits/ampliseq-childhood-cancer-panel.html]
- 8. Real-time genomic characterization of pediatric acute ... [https://www.nature.com/articles/s41375-025-02565-y]
- 9. Prevalence and detection of low-allele-fraction variants in ... [https://www.nature.com/articles/s41467-017-01470-y]
- 10. Analytical validation and implementation of a pan cancer ... [https://www.frontiersin.org/journals/genetics/articles/10.3389/fgene.2023.1067457/full]
- 11. OncoKids: A Comprehensive Next-Generation Sequencing ... [https://www.sciencedirect.com/science/article/pii/S1525157818301028]
- 12. The Growing Role of Precision Medicine in Paediatric ... [https://link.springer.com/article/10.1007/s11523-025-01178-w]
- 13. Technical Validation and Clinical Utility of an NGS ... [https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2022.854098/full]
- 14. Empirical evaluation of variant calling accuracy using ultra ... [https://www.nature.com/articles/s41598-018-38346-0]
- 15. Benchmarking reveals superiority of deep learning variant ... [https://elifesciences.org/reviewed-preprints/98300v2]
- 16. Benchmarking reveals superiority of deep learning variant ... [https://elifesciences.org/articles/98300]
- 17. Increasing calling accuracy, coverage, and read-depth in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8699914/]
- 18. AmpliSeq for Illumina Childhood Cancer Panel [https://support.illumina.com/sequencing/sequencing_kits/ampliseq-for-illumina-childhood-cancer/product-compatibility.html]
- 19. Targeted Gene Sequencing Panels [https://www.illumina.com/techniques/sequencing/dna-sequencing/targeted-resequencing/targeted-panels.html]
- 20. Evaluation of the Performance of AmpliSeq and SureSelect ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6774276/]
- 21. Complete Guide to Ion Ampliseq Technology [https://www.thermofisher.com/blog/behindthebench/complete-guide-to-ion-ampliseq-technology-how-does-ion-ampliseq-technology-work/]
- 22. Validation, Implementation, and Clinical Impact of the ... [https://www.sciencedirect.com/science/article/pii/S1525157821002348]
- 23. A simple method to estimate the in-house limit of detection for ... [https://bmcgenomdata.biomedcentral.com/articles/10.1186/s12863-020-00956-x]
- 24. Five Percent Variant Allele Frequency Is a Reliable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9348859/]
- 25. Confirming putative variants at ≤ 5% allele frequency ... [https://www.nature.com/articles/s41598-021-91142-1]
- 26. Standardization of Sequencing Coverage Depth in NGS [https://pmc.ncbi.nlm.nih.gov/articles/PMC6738196/]
- 27. Practical guide for the comparison of two next-generation ... [https://brd.nci.nih.gov/brd/paper/j-clin-pathol/2019/practical-guide-for-the-comparison-of-two-next-generation-sequencing/129090]
- 28. From Conventional Chemotherapy to Precision Medicine - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12566757/]
- 29. CHLA Launches OncoKids – a Comprehensive DNA and ... [https://www.rna-seqblog.com/chla-launches-oncokids-a-comprehensive-dna-and-rna-pediatric-cancer-panel/]
- 30. Design and Validation of a Custom Next-Generation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10002321/]
- 31. Systematic comparison of quantity and quality of RNA ... [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-024-05890-5]
- 32. Forensic DNA Recovery from FFPE Tissue Using the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12469798/]
- 33. Technical Validation and Clinical Utility of an NGS ... [https://pubmed.ncbi.nlm.nih.gov/35463953/]
- 34. Maximizing DNA Yield and Integrity from Limited FFPE ... [https://scholarworks.utrgv.edu/somrs/2025/posters/150/]
- 35. Determining Performance Metrics for Targeted Next ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6172655/]
- 36. Ion AmpliSeq On-Demand Panels for Targeted Sequencing [https://www.thermofisher.com/order/catalog/product/ar/en/A38629]
- 37. Assessing the Cost-Effectiveness of Next-Generation ... [https://pubmed.ncbi.nlm.nih.gov/38729563/]
- 38. Technical Validation and Clinical Utility of an NGS Targeted Panel to Improve Molecular Characterization of Pediatric Acute Leukemia - PMC [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9021638/]
- 39. AmpliSeq for Illumina Childhood Cancer Panel Support [https://support.illumina.com/sequencing/sequencing_kits/ampliseq-for-illumina-childhood-cancer/questions1.html]
- 40. The Next-Generation Sequencing Quality Initiative and ... [https://wwwnc.cdc.gov/eid/article/31/13/24-1175_article]
- 41. Best Practices for Quality Control of NGS Data [https://www.basepairtech.com/blog/best-practices-for-quality-control-and-pre-processing-of-ngs-data-a-guide-for-academic-researchers/]
- 42. Evaluation of Hybridization Capture Versus Amplicon‐ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4832303/]
- 43. Hybridization capture vs amplicon sequencing | IDT [https://www.idtdna.com/page/support-and-education/decoded-plus/targeted-sequencing-hybridization-capture-or-amplicon-sequencing/]
- 44. Understanding the Differences Between Amplicon-Based ... [https://diagnostech.co.za/understanding-the-differences-between-amplicon-based-and-hybrid-capture-based-ngs-target-enrichment/]
- 45. Assessment of Capture and Amplicon-Based Approaches ... [https://www.sciencedirect.com/science/article/pii/S1525157817304257]
- 46. Performance of amplicon and capture based next-generation ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0289188]
- 47. The concept of clinical utility for diagnostic tests [https://www.thermofisher.com/blog/clinical-conversations/the-concept-of-clinical-utility-for-diagnostic-tests/]
- 48. Development and validation of a targeted gene ... [https://www.nature.com/articles/s41598-019-52000-3]
- 49. Genetic characterization of Lynch syndrome germline ... [https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2025.1589765/full]
- 50. In-House NGS vs. Outsourced Sequencing Services [https://seqwell.com/in-house-ngs-vs-outsourced-sequencing-services-which-option-makes-the-most-sense-for-your-lab/]
- 51. how-can-you-benefit-from-outsourcing-sequencing-services [https://sourcebioscience.com/how-can-you-benefit-from-outsourcing-sequencing-services/]
- 52. NGS Data Analysis: Choosing the Right Approach for Your ... [https://dromicsedu.com/blog-details/ngs-data-analysis-choosing-the-right-approach-for-your-research-and-when-to-outsource]
- 53. Managing health supply chain risks for the successful ... [https://pfscm.org/managing-health-supply-chain-risks-for-the-successful-implementation-of-genomic-surveillance-using-ngs/]