UV Irradiation for PCR Workstation Decontamination: A Scientist's Guide to Contamination Control
This article provides a comprehensive guide for researchers and drug development professionals on utilizing UV irradiation to eliminate PCR contamination.
UV Irradiation for PCR Workstation Decontamination: A Scientist's Guide to Contamination Control
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on utilizing UV irradiation to eliminate PCR contamination. It covers the foundational science of how UV light inactivates DNA, details effective decontamination protocols and workstation features, offers advanced troubleshooting for persistent contamination, and validates methods through peer-reviewed evidence and comparative analysis with other techniques. The goal is to equip laboratories with the knowledge to establish robust, reliable workflows that prevent false positives and ensure data integrity in sensitive molecular biology applications.
The Science of UV Light: How It Inactivates DNA Contaminants
Frequently Asked Questions (FAQs)
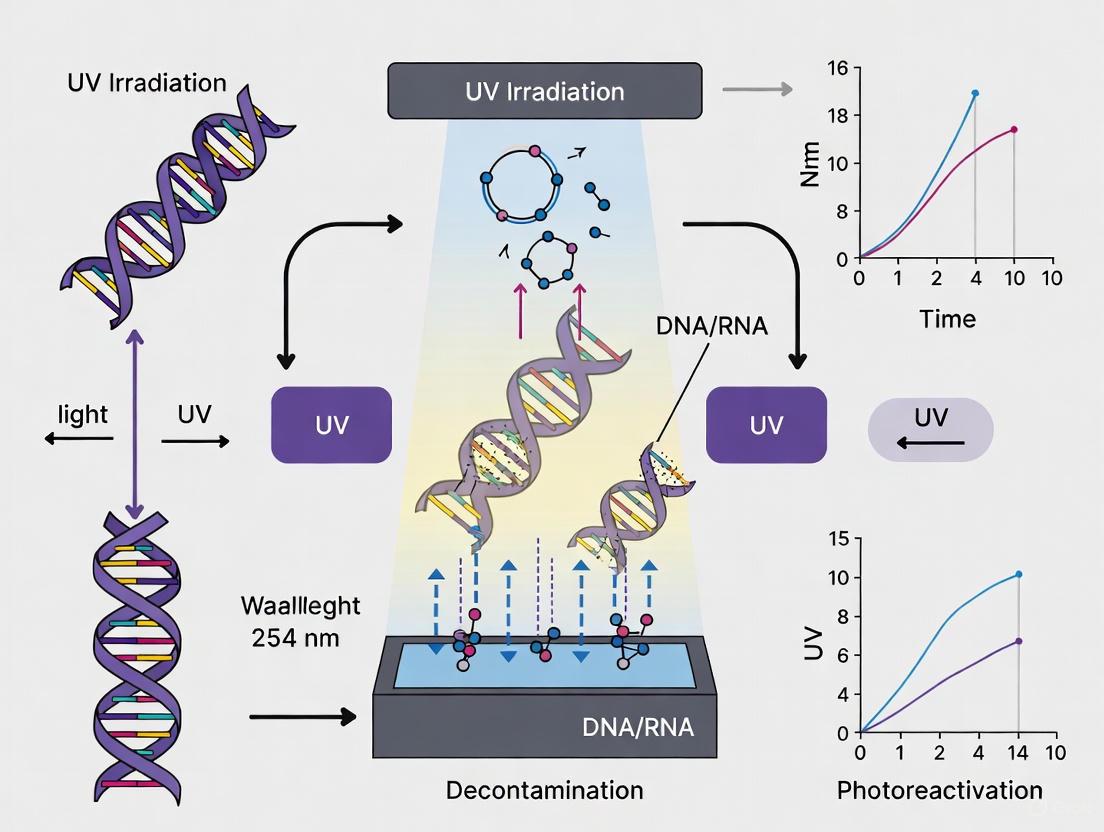
FAQ 1: What are pyrimidine dimers and how do they inactivate DNA? Pyrimidine dimers are a type of DNA lesion primarily formed when DNA absorbs ultraviolet (UV) radiation. The two main types are Cyclobutane Pyrimidine Dimers (CPDs) and Pyrimidine (6-4) Pyrimidone Photoproducts (6-4PPs) [1]. They form between adjacent pyrimidine bases (thymine or cytosine) on the same DNA strand [1]. This covalent linkage prevents the separation of DNA strands, which is essential for fundamental cellular processes like transcription and replication [2]. By blocking these processes, pyrimidine dimers effectively inactivate DNA, leading to cell death, which is the desired outcome for decontaminating PCR workstations.
FAQ 2: Why is UV-C light, specifically at 254 nm, used for decontamination? UV-C light (100-280 nm) is highly effective because its wavelength corresponds closely with the DNA absorption peak of approximately 260 nm [1]. This makes DNA a prime chromophore for UV-C photons. The action spectrum for DNA damage peaks around 260-265 nm and falls sharply at longer wavelengths [1] [3]. Therefore, 254 nm UV-C light, which is readily produced by germicidal lamps, induces maximum DNA damage with high efficiency, leading to the rapid formation of pyrimidine dimers and reliable inactivation of contaminating microorganisms [4] [5].
FAQ 3: What factors can reduce the efficacy of UV decontamination in my PCR workstation? Several factors can impact decontamination efficacy:
- UV Lamp Age and Output: UV lamps have a finite lifespan. Their efficiency begins to decline after approximately 5,000 hours of operation, with output decreasing by about 15% for every additional 1,000 hours [4]. Regular replacement is crucial.
- Dirty or Clogged Filters: Dust filters in the air recirculation module can become clogged, reducing airflow and the efficiency of UV air sterilization [4].
- Shadows and Surface Obstructions: UV light travels in a straight line. Items placed inside the cabinet can create shadows, shielding contaminants from direct exposure. Ensure all surfaces are directly exposed to the UV light during the decontamination cycle [5].
- Distance from the Light Source: UV intensity follows the inverse-square law, meaning it decreases dramatically with distance.
FAQ 4: How can I experimentally verify the formation of pyrimidine dimers in my lab? You can verify dimer formation and assess decontamination efficiency using several methods:
- Enzyme-Linked Immunosorbent Assay (ELISA): Utilizes damage-specific monoclonal antibodies to detect and quantify pyrimidine dimers [6].
- CPD-Specific Glycosylases: Enzymes like T4 pyrimidine dimer glycosylase (T4-pdg) can be used to cleave DNA at CPD sites. The cleavage products are then visualized through gel electrophoresis, providing a qualitative or semi-quantitative assessment [7] [3].
- Microbiological Assays: A practical method involves exposing a thin lawn of a non-pathogenic, UV-sensitive bacterium (e.g., E. coli) on an agar plate inside the workstation. After UV exposure, the plate is incubated. A successful decontamination cycle will result in no bacterial growth.
Troubleshooting Guides
Problem: Inconsistent Decontamination Results
- Potential Cause: Old or inefficient UV lamps.
- Solution: Track UV lamp usage hours. Replace lamps after 9,000 hours of operation or as recommended by the manufacturer, typically every 6-12 months depending on usage [4].
- Potential Cause: Surface contamination not directly exposed to UV light.
- Solution: Ensure the workstation is not overcrowded. Wipe down all interior surfaces with an appropriate decontaminant (e.g., 70% ethanol, diluted sodium hypochlorite) before initiating the UV cycle to remove dust and biofilms that might shield organisms. Always refer to the manufacturer's manual for compatible cleaning solutions to avoid damaging acrylic surfaces or protective coatings [4].
Problem: Positive Control Growth in Verification Assays
- Potential Cause: Insufficient UV dose or exposure time.
- Solution: Calibrate the UV exposure. Measure the UV intensity at the working surface using a UV-C radiometer. Ensure the dose (intensity × time) is sufficient to inactivate common contaminants. The required dose can be validated using the microbiological assay described above.
- Potential Cause: Malfunctioning UV lamp or air recirculation system.
- Solution: Perform routine maintenance. Check that the UV indicator on the air recirculating module is lit during operation [4]. Clean the dust filters monthly by rinsing with warm water and letting them air dry completely [4].
Quantitative Data on Pyrimidine Dimer Formation
Table 1: Relative Frequency of CPD Formation at Different Dipyrimidine Sites under UVB Light This table summarizes the distribution of CPDs formed by direct UV irradiation, which is relevant for understanding the mechanism of DNA inactivation [3].
| Dipyrimidine Site | Relative Frequency of CPD Formation (%) |
|---|---|
| TT | 36 ± 4 |
| TC | 32 ± 3 |
| CT | 20 ± 5 |
| CC | 13 ± 2 |
Table 2: Comparison of Common DNA Lesions Induced by UV Light This table differentiates between the two primary photolesions [6] [1].
| Feature | Cyclobutane Pyrimidine Dimer (CPD) | (6-4) Pyrimidine-Pyrimidone Photoproduct (6-4PP) |
|---|---|---|
| Chemical Structure | Cyclobutane ring between C5 and C6 atoms | Single covalent bond between C6 and C4 atoms |
| Relative Abundance | Higher (Most frequent) | Lower |
| Common Formation Sites | TpT, TpC, CpC | TpC, TpT, CpT, CpC |
| Absorption Peak | ~230 nm (UV-C) | ~310-330 nm (UV-A) |
Experimental Protocols for Verification
Protocol 1: Verification Using Immunoassay This method provides sensitive and quantitative detection of pyrimidine dimers.
- Sample Preparation: Place DNA samples (e.g., plasmid DNA or a DNA smear) on a clean surface inside the PCR workstation.
- UV Exposure: Subject the workstation to a standard decontamination cycle.
- DNA Extraction and Fixation: Retrieve the DNA samples and immobilize them on a nitrocellulose membrane.
- Antibody Incubation: Incubate the membrane with a primary antibody specific for CPDs or 6-4PPs.
- Detection: Use an enzyme-conjugated secondary antibody and a chemiluminescent substrate to detect the bound primary antibody. The signal intensity correlates with the number of pyrimidine dimers formed [6].
Protocol 2: Verification Using T4 Endonuclease V Assay This method is based on the enzymatic cleavage of DNA at CPD sites.
- Sample Preparation and UV Exposure: Follow steps 1 and 2 from Protocol 1.
- Enzymatic Digestion: Treat the exposed DNA with T4 pyrimidine dimer glycosylase (T4-pdg), which recognizes CPDs and cleaves the DNA backbone at these sites [7] [3].
- Analysis: Run the digested DNA samples on an agarose gel. A smear of DNA fragments indicates successful cleavage and confirms the presence of CPDs. A non-irradiated control sample should show intact, high-molecular-weight DNA.
Visualizing the Mechanism and Workflow
UV-Induced DNA Inactivation Path
Decontamination Verification Flow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Pyrimidine Dimer Research and Verification
| Item | Function / Explanation |
|---|---|
| CPD-specific Monoclonal Antibodies | Used in ELISA or immunofluorescence to specifically detect and quantify CPDs in DNA samples extracted after UV exposure [6]. |
| T4 Pyrimidine Dimer Glycosylase (T4-pdg) | A key enzyme that recognizes CPDs and cleaves the DNA glycosyl bond, used in assays to visualize CPD formation via gel electrophoresis [7] [3]. |
| UV-C Radiometer | A crucial instrument for calibrating and validating PCR workstations by measuring the intensity of UV-C light at the working surface to ensure a sufficient decontamination dose. |
| HEPA Filter | (For filtered workstations) Maintains a positive pressure, contaminant-free environment by removing particles from incoming air, preventing recontamination after UV cycling [5]. |
| DNA Substrate (e.g., Plasmid DNA) | A pure DNA sample used as a target in verification assays. UV-induced dimer formation can be quantified by changes in its electrophoretic mobility or through immunoassay [3]. |
Frequently Asked Questions (FAQs)
Q1: Why is 254 nm the most prevalent wavelength used for UV decontamination in PCR workstations? The wavelength of 254 nm is widely used because it is very close to the optimal germicidal wavelength, which is approximately 262 nm [8]. This wavelength is efficiently produced by conventional low-pressure mercury vapor lamps, making it a standard and cost-effective technology [9] [8]. At 254 nm, UV photons are absorbed by microbial DNA and RNA, causing the formation of thymine dimers and other photoproducts that inhibit replication and transcription, leading to microbial death [9] [10].
Q2: What are the primary factors that can reduce the efficacy of 254 nm UV-C decontamination? Several factors can significantly impact efficacy:
- Shadowing and Direct Line-of-Sight: Microorganisms located in shadows or on surfaces not directly exposed to the light will not be effectively inactivated [11] [8].
- Distance from Source: The inverse square law applies; doubling the distance from the UV source quarters the irradiance, drastically increasing the time required for effective disinfection [8].
- Surface Type and Porosity: Irregular, rough, or porous surfaces (like cloth gowns) can shield microorganisms and reduce UV penetration compared to smooth, non-porous surfaces (like stainless steel) [12] [10].
- Organic Matter: The presence of organic debris or biofilms can protect microbes from UV exposure [8] [10].
- Microbial Type and Load: Bacterial spores are far more resistant than vegetative cells and viruses, and a higher initial microbial load requires a higher UV dose for complete inactivation [9] [13].
Q3: Can 254 nm UV-C light inactivate resilient bacterial spores like those from Clostridioides difficile? Yes, but it requires a significantly higher dose compared to vegetative bacteria and viruses. One study demonstrated that a complete reduction of C. difficile spores on agar plates required a UV dose of 2208 mJ/cm², achieved after 20 minutes of exposure [9]. The study also noted that moist surfaces or liquids can enhance the efficacy of UV-C treatment against these resilient spores [9].
Q4: What safety precautions are mandatory when operating UV-C cabinets in a laboratory? UV-C radiation poses risks to skin and eyes. Strict safety protocols must be followed [8]:
- Room Evacuation: UV-C disinfection cycles must only be run in unoccupied rooms.
- Safety Interlocks: Use cabinets with automatic shutoff features that deactivate UV lamps when the front panel is opened [14] [15].
- Warning Signage: Clear signs must be posted to indicate when UV-C is in operation.
- Protective Equipment: Wear appropriate personal protective equipment if exposure is possible.
- Ozone Ventilation: Ensure adequate ventilation if using lamps that emit wavelengths below 240 nm, which can generate ozone [8].
Troubleshooting Common Experimental Issues
Problem: Inconsistent Decontamination Results Across Replicates
- Potential Cause: Inconsistent or unverified UV irradiance and dose.
- Solution: Use a calibrated radiometer to regularly measure the UV irradiance (mW/cm²) at the sample plane. Calculate the dose (mJ/cm²) as irradiance × exposure time (seconds). Ensure the UV lamps have been warmed up for the manufacturer-specified time (e.g., 30 minutes) before use to stabilize output [9] [8].
Problem: Persistent PCR Contamination Despite UV Treatment
- Potential Cause 1: Shadowing effects leaving areas untreated.
- Solution: Ensure all items in the cabinet are positioned for maximal light exposure. Consider using a cabinet with an omnidirectional design and highly reflective interior walls to minimize shadows and ensure uniform light distribution [11] [12].
- Potential Cause 2: The UV exposure time or dose is insufficient for the contaminating organism.
- Solution: Validate the decontamination cycle against a resilient challenge organism (e.g., bacterial spores). Increase the exposure time to deliver a higher dose, as spores may require doses thousands of times higher than viruses [9] [15].
Problem: Rapid Degradation of Plastic Materials Inside the UV Cabinet
- Potential Cause: Prolonged and cumulative UV exposure damages certain polymers.
- Solution: Cover sensitive plastic components with UV-opaque materials during the decontamination cycle or use UV-resistant materials for items permanently housed within the cabinet [8].
Quantitative Efficacy Data
The following tables summarize the doses of 254 nm UV-C light required to inactivate various microorganisms, as reported in the literature. This data is essential for validating and designing decontamination protocols.
Table 1: UV-C Dose (254 nm) for Microbial Inactivation on Surfaces
| Microorganism | Surface/Medium | Target Reduction | Required UV Dose (mJ/cm²) | Citation |
|---|---|---|---|---|
| SARS-CoV-2 (Alpha strain) | Stainless Steel (non-porous) | Complete inactivation (in cell culture) | 2.5 - 3.4 | [12] |
| SARS-CoV-2 (Alpha strain) | Plastic Apron (non-porous) | Complete inactivation (in cell culture) | 2.9 | [12] |
| SARS-CoV-2 (Alpha strain) | Disposable Gown (porous) | Complete inactivation (in cell culture) | 3.4 | [12] |
| Clostridioides difficile spores | BHI Agar Plates | Complete reduction (≥5 log₁₀) | 2208 | [9] |
| Geobacillus spores | Spore Strip | Effective Decontamination | ~1000 (1 J/cm²) | [16] |
Table 2: UV-C Dose (254 nm) for Microbial Inactivation in Water
| Microorganism | Water Type | Target Reduction | Required UV Dose (mJ/cm²) | Citation |
|---|---|---|---|---|
| Escherichia coli | Filtered Water | 1-log (90%) | ~5 | [13] |
| MS2 Bacteriophage | Challenge Water | 1-log (90%) | ~30 | [13] |
| Cryptosporidium parvum oocysts | Laboratory Water | 1-log (90%) | 485.3 | [13] |
Detailed Experimental Protocols
Protocol 1: Validating UV-C Cabinet Efficacy Against Bacterial Spores This protocol is adapted from studies on C. difficile and Geobacillus spores [9] [16].
Spore Preparation and Purification:
- Prepare spores of a suitable test organism (e.g., Bacillus subtilis or Geobacillus steareothermophilus) on sporulation agar.
- Harvest the spore lawn and purify it through repeated centrifugation and washing in phosphate-buffered saline (PBS).
- Heat-treat the suspension (e.g., 70°C for 20 min) to eliminate remaining vegetative cells.
- Enumerate the viable spores by serial dilution in PBS and plating on BHI agar with 0.1% sodium taurocholate to promote germination. Confirm spore purity via phase-contrast microscopy [9].
Surface Inoculation:
UV-C Exposure:
- Turn on the UV cabinet lamps and allow them to warm up for at least 30 minutes to stabilize output [9].
- Measure the baseline irradiance at the sample plane using a calibrated radiometer.
- Expose the inoculated coupons to UV-C for predetermined times (e.g., 0, 5, 10, 20, 30 min). Include non-irradiated controls.
- Calculate the delivered dose for each time point: Dose (mJ/cm²) = Irradiance (mW/cm²) × Time (s).
Post-Exposure Analysis:
- After exposure, transfer each coupon to a container with a neutralization buffer or PBS.
- Vortex vigorously to resuspend the spores.
- Perform serial dilutions and plate on appropriate agar media.
- Incubate anaerobically/aerobically as required and count the resulting colonies after 24-48 hours.
- Calculate the log₁₀ reduction compared to the non-irradiated control.
Protocol 2: Testing Viral Inactivation on Personal Protective Equipment (PPE) This protocol is based on studies with SARS-CoV-2 and H1N1 on masks and gowns [12] [17].
Viral Stock and Surface Preparation:
- Use a validated viral stock (e.g., SARS-CoV-2, H1N1, or a safer surrogate like MS2 bacteriophage) at a known titer (e.g., 10⁵ TCID₅₀/mL).
- Cut PPE materials (surgical mask, N95 respirator fabric, gown swatch) into standardized coupons.
Inoculation and Drying:
- Inoculate a small, defined area (e.g., 1 cm²) of each coupon with a precise volume of viral suspension (e.g., 100 µL).
- Allow the inoculum to dry for a fixed period (e.g., 20 min) in a biosafety cabinet.
UV-C Treatment:
Virus Recovery and Titration:
- Recover the virus from the coupons by immersing them in cell culture medium and vortexing.
- Determine the remaining infectious viral titer using a suitable cell culture assay (e.g., TCID₅₀ on Vero E6 cells for SARS-CoV-2) and observe for cytopathic effects [12].
- Use RT-qPCR to quantify viral genomic material, noting that PCR may detect non-viable virus and overestimate survival [12].
Workflow and Safety Diagrams
UV Decontamination Workflow
UV Safety Protocol
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for UV-C Decontamination Research
| Item | Function/Application | Example from Literature |
|---|---|---|
| Low-Pressure Mercury Lamps | Standard source for 254 nm UV-C radiation. | Used in a DESAG Lightbox UVIS for C. difficile spore inactivation [9]. |
| Calibrated Radiometer | Essential for measuring UV irradiance (mW/cm²) to calculate accurate dose. | Thorlabs PM100D power meter used in spore studies [9]. |
| Chemical Actinometry Solutions | Chemical method for validating and calibrating UV dose in liquid or reactor systems. | Used to establish UV dose in water disinfection studies [13]. |
| Phase-Contrast Microscope | Used to confirm spore purity and morphology in suspension preparation. | Axioscope microscope used for C. difficile spore validation [9]. |
| HEPA-Filtered Biosafety Cabinet | Provides a sterile, contained environment for preparing spore/viral inoculations pre- and post-UV exposure. | Essential for aseptic technique in sample preparation [15]. |
| Sporulation Agar | Specialized growth medium to induce and support high-yield spore formation. | A 70:30 mixture of sporulation medium and BHI used for C. difficile [9]. |
| Sodium Taurocholate | Bile salt used in culture media to stimulate the germination of C. difficile spores for viable counting. | Added to BHI agar at 0.1% (w/v) for colony counts [9]. |
| Phosphate-Buffered Saline (PBS) | A neutral, isotonic buffer for preparing spore and viral suspensions, and for serial dilutions. | Used as a suspension and dilution medium for spores [9] [12]. |
FAQs on PCR Contamination
The most common sources of contamination are:
- Amplicon Carryover: PCR products from previous amplification reactions are the most prevalent source. The enormous number of molecules (up to 10^12) generated can easily contaminate new reactions [18].
- Genomic DNA (gDNA): A significant concern when performing reverse transcription PCR (RT-PCR) to detect RNA expression. Contaminating gDNA in an RNA sample can lead to false positive results [19].
- Cross-Contamination: Sample-to-sample contamination can occur, especially with samples requiring extensive processing before PCR [18].
- Contaminated Reagents and Equipment: Enzymes, water, buffers, and pipettes can become contaminated with exogenous DNA from the laboratory environment or previously handled cloned DNA [18].
Q2: How can I prevent genomic DNA contamination in RT-PCR experiments?
- DNase Treatment: Treat your RNA sample with DNase before the reverse transcription step to degrade any contaminating gDNA, then heat-inactivate the DNase [19].
- Exon-Spanning Assays: Design your PCR assays so that the primers bind to sequences in two different exons. Any amplification from gDNA will contain a large intron and may not be efficiently amplified, whereas amplification from cDNA will be the correct, shorter product [19].
- No-RT Control: Always include a control where the reverse transcriptase enzyme is omitted from the reaction. Amplification in this control indicates the presence of gDNA contamination [19].
Q3: What laboratory practices are most effective for preventing amplicon carryover?
- Unidirectional Workflow: Establish physically separate areas for pre-PCR (reaction setup) and post-PCR (amplification and analysis) activities. Never bring reagents, equipment, or supplies from the post-PCR area back into the pre-PCR area [19] [18].
- Dedicated Equipment and Supplies: Use separate sets of pipettes, pipette tips with aerosol filters, lab coats, and waste containers for the pre-PCR and post-PCR areas [18].
- Aliquoting Reagents: Store all reagents, including primers, in single-experiment aliquots to minimize the risk of contaminating stock solutions [19] [18].
Q4: My PCR shows a smear on the gel. Is this contamination?
A smear can have multiple causes. First, run a negative control (no template DNA).
- If the negative control is clean, the smear is likely due to suboptimal PCR conditions (e.g., too much template, low annealing temperature, excessive cycle number) or poorly designed primers [18].
- If the negative control is also smeared, this indicates contamination, likely from amplicons. In this case, you must decontaminate your workspace and replace your reagents [18].
Q5: How effective is UV irradiation for decontaminating my PCR workstation?
UV irradiation is a recommended method for decontaminating equipment. It works by damaging residual DNA, primarily through the formation of thymidine dimers, which prevents its amplification [18].
- Efficacy: The efficacy of UV decontamination is highly dependent on the device's dosage (energy output), with different devices showing enormous variability [20]. It is most effective on clean, non-porous surfaces and equipment like pipettes, tubes, racks, and laminar flow cabinets [19] [21].
- Limitations: Its utility is limited on porous materials like cardboard, and its effectiveness can be reduced if surfaces are shadowed or soiled with organic debris that can shield contaminants [20].
Troubleshooting Common PCR Contamination Problems
| Problem & Symptoms | Possible Source of Contamination | Recommended Solution |
|---|---|---|
| Unexpected amplification in negative control; false positives. | Amplicon carryover from previous PCRs or contaminated reagents [19] [18]. | 1. Replace all reagents with fresh aliquots [19]. 2. Decontaminate pipettes and work surfaces with 10% bleach or UV irradiation [19] [18]. 3. Use a uracil-DNA-glycosylase (UDG) system to degrade carryover amplicons from previous reactions [22]. |
| Unexpected bands in no-RT control during RT-PCR. | Genomic DNA contamination in the RNA sample [19]. | 1. Treat RNA samples with DNase [19]. 2. Redesign primers to span an exon-exon junction [19]. 3. Always include a no-RT control to monitor for this issue [19]. |
| Smearing in both test samples and negative control on a gel. | Widespread amplicon contamination in the laboratory environment [18]. | 1. Move your pre-PCR setup to a new, decontaminated location [18]. 2. Use a new set of primers with different sequences that do not interact with the accumulated contaminants [23]. 3. Implement strict unidirectional workflow practices to prevent recurrence [21]. |
Experimental Protocols for Decontamination and Validation
Protocol 1: UV Decontamination of PCR Workstations and Equipment
This protocol outlines the use of UV irradiation to decontaminate surfaces and equipment in a laminar flow cabinet dedicated to pre-PCR setup [21] [18].
Materials:
- Laminar flow cabinet equipped with a UV lamp [21] [18].
- 70% ethanol or 5-10% bleach solution [19] [18].
- Pipettes, tube racks, and other non-porous equipment.
Method:
- Pre-Cleaning: Before UV exposure, wipe down all surfaces and equipment with 70% ethanol or a 5-10% bleach solution to remove any physical debris. A 5% bleach solution is particularly effective at degrading DNA [19].
- UV Exposure: Place pipettes, racks, and other small equipment inside the cabinet. Ensure all surfaces that will contact PCR reagents are facing the UV lamp. Close the cabinet sash and irradiate for a minimum of 15-30 minutes. For extensive contamination, leaving equipment under UV light overnight is effective [18].
- Post-Procedure: After UV treatment, the workstation and equipment are ready for setting up PCR reactions.
Technical Note: UV-C light in the range of 250-270 nm is most effective for germicidal purposes [24]. The dosage (mJ/cm²) is critical, and efficacy can vary between devices and based on distance from the source [20].
Protocol 2: Establishing a Contamination-Free PCR Workflow
This methodology describes the physical separation of laboratory spaces to minimize the risk of contamination [19] [21] [18].
Materials:
- Two physically separated rooms or dedicated bench spaces.
- Dedicated pipettes and aerosol-resistant filter tips for each area.
- Separate lab coats, waste containers, and supplies.
Method:
- Define Areas:
- Pre-PCR Area (Template-Free): A clean room or hood used exclusively for preparing the PCR master mix and setting up reactions. No template DNA, amplicons, or purified DNA should ever be introduced here [19] [18].
- Post-PCR Area: A separate room for adding template DNA, running the PCR thermocycler, and analyzing PCR products (e.g., gel electrophoresis) [19].
- Unidirectional Workflow: The experimental workflow must only move from the pre-PCR area to the post-PCR area. Reagents, equipment, or personnel should never move from the post-PCR area back to the pre-PCR area [18].
- Regular Decontamination: The pre-PCR area and its equipment should be routinely decontaminated with bleach or UV light [19] [18].
Research Reagent Solutions for Contamination Control
The following table details key reagents and materials used to prevent and control PCR contamination.
| Item | Function in Contamination Control |
|---|---|
| Aerosol-Resistant Filter Pipette Tips | Creates a barrier between the pipette and the liquid, preventing aerosols from contaminating the pipette shaft and subsequent samples [19] [21]. |
| Uracil-DNA-Glycosylase (UDG) | An enzyme used in a pre-PCR incubation step to cleave uracil-containing DNA from previous PCR reactions (where dUTP was used), preventing their amplification [22]. |
| DNase I, RNase-free | Degrades contaminating genomic DNA in RNA samples prior to reverse transcription for RT-PCR [19]. |
| Hot-Start DNA Polymerase | Remains inactive at room temperature, preventing non-specific amplification and primer-dimer formation during reaction setup, which reduces background and increases specificity [25] [23]. |
| Dedicated Laminar Flow Cabinet | Provides a HEPA-filtered, contaminant-free environment for setting up PCR reactions. Models with built-in UV lamps allow for easy surface decontamination [21]. |
| Bleach Solution (5-10%) | An effective and common laboratory chemical for degrading DNA on non-porous surfaces like benchtops and pipettes [19] [18]. |
Quantitative Data on UV Decontamination Efficacy
UV decontamination efficacy is highly variable and depends on the device and the surface being treated. The table below summarizes key data from studies on UV inactivation of viruses, which is relevant for understanding its potential for destroying DNA contaminants.
| UV Device / Parameter | Key Efficacy Findings & Quantitative Data |
|---|---|
| General UVGI Efficacy | UV-C light (200-280 nm) inactivates microbes by preventing transcription and replication. The 250-270 nm range is most effective [24]. |
| Handheld & Room Devices | A study of 14 UV technologies showed enormous variability in dosage (0.01 to 729 mJ cm⁻²) and antiviral efficacy, ranging from no decontamination to nearly achieving sterilization [20]. |
| Inactivation of SARS-CoV-2 Surrogate | On non-porous surfaces (e.g., stainless steel), sufficient UV dosage can achieve significant viral reduction. Porous materials like cardboard require a far greater dosage for the same effect [20]. |
| Clinical Surface Disinfection | One study reported complete clearance of viral RNA from surfaces after 15 minutes of UVC irradiation at 254 nm [24]. |
FAQs on Contamination and Data Integrity
Q1: What are the common sources of contamination in a PCR laboratory? Contamination can originate from multiple sources, including amplicon carryover from previous PCR reactions, foreign DNA templates, laboratory surfaces, reagents, and the researchers themselves (e.g., skin cells, aerosols) [25] [26]. In low-biomass samples, even minuscule amounts of contaminating DNA can lead to spurious results, as the target DNA 'signal' can be dwarfed by the contaminant 'noise' [26].
Q2: How does contamination specifically impact data integrity and reproducibility? Contamination compromises data integrity by introducing false positives, masking true signals, and generating misleading or non-reproducible data [26]. This can distort ecological patterns, cause false attribution of pathogen exposure, and lead to inaccurate conclusions about the presence of microbes in a sample [26]. The failure to reproduce published biomedical research is often linked to such undetected contamination issues [27].
Q3: Why is UV irradiation an effective method for decontamination in PCR workstations? Ultraviolet Germicidal Irradiation (UVGI), particularly at a wavelength of 254 nm, damages the genetic material (DNA or RNA) in biological cells and viruses, thereby inhibiting their ability to replicate [8]. This makes it a powerful, non-chemical "no-touch" method for decontaminating air and surfaces within enclosed workstations, significantly reducing surface and airborne contaminants [8] [28].
Q4: What are the critical factors that affect the efficacy of UV decontamination? The germicidal effectiveness of UV-C light is influenced by several factors [8]:
- Exposure Time: Longer dwell times result in higher microbial inactivation.
- Distance: The intensity of UV light follows the inverse square law; doubling the distance from the source quarters the energy received, significantly increasing the time required for disinfection [8].
- Intensity & Wavelength: Optimal germicidal action occurs between 250-270 nm [8].
- Environmental Conditions: Humidity and temperature can impact efficacy [8].
- Line of Sight: UV light cannot reach microbes hidden in shadows, folds, or crevices [8].
Q5: What are the essential safety precautions when using UV decontamination systems? UV-C light is harmful to humans and must be used with strict safety protocols [8] [28]:
- No Occupancy: UV-C disinfection must only occur in unoccupied rooms or enclosed workstations.
- Safety Interlocks: Use systems with safety shut-off switches that automatically deactivate UV lamps when a door is opened [28].
- Shielding: Workstations should use protective panels (e.g., Makrolon) that block UV light [28].
- Signage: Post warning signs to indicate UV hazard [8].
Troubleshooting Guide for UV-PCR Workstation Decontamination
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| PCR Contamination (False Positives) | Ineffective surface decontamination inside the workstation. | Ensure all interior surfaces are in the direct line of sight of the UV lamps. Clean surfaces with a DNA-degrading solution (e.g., dilute bleach) before UV irradiation to remove nucleic acids that may be shielded in organic matter [26]. |
| Inconsistent Decontamination Efficacy | Variable exposure times or lamp degradation. | Standardize decontamination cycles using the workstation's timer (e.g., 30 minutes) [28]. Regularly replace UV lamps as per manufacturer guidelines, as output diminishes over time [28]. |
| Airborne Contamination Post-Decontamination | Contaminants are reintroduced from the lab environment or the internal air is not purified. | Use a PCR workstation model with a HEPA filter, which provides three layers of UV irradiation and air filtration/circulation to ensure maximum decontamination of both surfaces and air [28]. |
| Persistent Background in Low-Biomass Samples | Contamination from reagents or cross-contamination between samples. | Incorporate comprehensive negative controls (e.g., sterile swabs, empty collection vessels, aliquots of preservation solution) throughout the entire workflow to identify contamination sources [26]. Use single-use, DNA-free plasticware where possible [26]. |
Quantitative Data on UVGI Decontamination
Table 1: Factors Influencing UVGI Efficacy [8]
| Factor | Impact on Efficacy | Optimal Range / Consideration |
|---|---|---|
| Wavelength | Determines the photon energy and microbial absorption. | 250–270 nm (most lethal), with 254 nm being standard for low-pressure mercury lamps [8]. |
| Exposure Time (Dwell Time) | Directly proportional to the total germicidal energy (fluence) delivered. | Must be calibrated based on intensity, distance, and target organism. |
| Distance from Source | Governed by the inverse-square law; intensity decreases rapidly with distance. | Minimize distance to the target surface for maximum efficiency [8]. |
| Relative Humidity | High humidity can reduce efficacy by creating a protective barrier for microbes. | Generally, lower humidity (<60%) is more favorable for UVGI. |
| Microbial Susceptibility | Different organisms have varying resistance to UV damage. | Enveloped viruses are more susceptible than non-enveloped ones; bacterial spores are more resistant than vegetative cells [29]. |
Table 2: Key Contamination Sources and Prevention Strategies in Low-Biomass Studies [26]
| Contamination Source | Prevention Strategy | Control Method |
|---|---|---|
| Human Operator | Use full personal protective equipment (PPE): gloves, mask, clean suit, and shoe covers. | Swab PPE and exposed skin to create control samples [26]. |
| Sampling Equipment & Reagents | Use single-use, DNA-free equipment. Decontaminate reusable tools with 80% ethanol followed by a DNA-degrading solution (e.g., bleach). | Include "blank" controls (e.g., an empty collection vessel, an aliquot of preservation solution) [26]. |
| Laboratory Environment | Pre-treat plasticware/glassware with autoclaving and/or UV-C light sterilization. Use PCR workstations with HEPA filtration and UV decontamination [28] [26]. | Swab workstation surfaces and use air samplers to monitor the background bioburden. |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Materials for UV Decontamination and Contamination Control
| Item | Function / Explanation |
|---|---|
| UV-C Lamp (254 nm) | The core source of germicidal radiation that inactivates microorganisms by damaging their DNA/RNA, preventing replication [8] [28]. |
| HEPA Filter | Used in advanced workstations to remove airborne particles and microbes, preventing contamination of samples from aerosols [28]. |
| DNA-Decontamination Solution (e.g., Bleach) | Used to chemically degrade contaminating DNA on surfaces and equipment before UV irradiation, tackling nucleic acids that may be shielded from UV light [26]. |
| Personal Protective Equipment (PPE) | Gloves, masks, and clean suits act as a physical barrier to prevent the introduction of human-associated contaminants (skin cells, hair, respiratory droplets) into the sample or workstation [26]. |
| Negative Control Samples | Sterile swabs, empty collection vessels, and samples of pure reagents processed alongside actual samples. They are critical for identifying the source and level of background contamination in a study [26]. |
Experimental Workflow and Contamination Pathways
PCR Contamination Pathways
Contamination Control Workflow
In molecular biology, particularly in sensitive applications like Polymerase Chain Reaction (PCR), the integrity of your samples is paramount. Contamination from exogenous DNA, RNA, or nucleases can compromise experiments, leading to false positives, unreliable data, and wasted resources. Decontamination is a critical practice to protect your work, and the choice between ultraviolet (UV) irradiation and chemical methods depends on your specific application, the materials you need to decontaminate, and the required turnaround time. This guide provides troubleshooting and FAQs to help you implement effective decontamination protocols within your research on UV irradiation for PCR workstations.
Frequently Asked Questions (FAQs)
What is the primary mechanism of UV decontamination?
UV-C light, in the range of 200-280 nm, inactivates microorganisms and degrades contaminating nucleic acids by damaging their DNA or RNA. The energy from UV light causes the formation of cyclobutane pyrimidine dimers (CPDs) between adjacent pyrimidine bases (such as thymine or cytosine). This DNA damage disrupts the base pairing and prevents polymerases from replicating the nucleic acid strand, thereby rendering the contaminant non-amplifiable [30] [31]. The efficacy peaks at wavelengths around 263-270 nm [31].
When should I use chemical decontamination over UV?
Chemical decontamination, particularly with sodium hypochlorite (bleach), is highly effective for surface decontamination of workbenches and equipment and for direct addition to liquid waste to degrade DNA [32]. It is a direct, rapid method for cleaning spills and for materials that UV light cannot penetrate. Chemical methods are also crucial during the sampling of low-biomass environments, where surfaces should be treated with agents like sodium hypochlorite to remove cell-free DNA that can persist even after autoclaving [26].
Can UV damage my PCR workstation or reagents?
Prolonged UV exposure can degrade certain materials, such as plastics, and the acrylic shields of some workstations may yellow over time. Furthermore, UV light is a surface decontaminant and will not penetrate liquids or solid objects. It is crucial to ensure that sensitive reagents and samples are not left inside the workstation during the UV decontamination cycle, as the radiation can degrade nucleic acids and enzymes [33].
How do I know if my decontamination methods are working?
Routine use of negative controls is the most direct way to monitor contamination in your experiments. A negative control, which contains all PCR components except the template DNA, should show no amplification. If a PCR product is detected, it indicates contamination is present [34]. Furthermore, for UV lamps, regular intensity testing is recommended as part of a scheduled maintenance program to ensure they are delivering the required dose for effective decontamination [33].
What are the key considerations for working with low-biomass samples?
Low-biomass samples (e.g., from human tissues, treated drinking water, atmospheric samples) are disproportionately affected by contamination. Key practices include:
- Using Personal Protective Equipment (PPE): Wear gloves, cleansuits, and masks to limit contamination from the researcher [26].
- Decontaminating Equipment: Use DNA-free, single-use equipment when possible. Otherwise, decontaminate with 80% ethanol followed by a nucleic acid degrading solution like sodium hypochlorite [26].
- Including Extensive Controls: Process "sampling controls" (e.g., empty collection vessels, swabs of the air) alongside your samples to identify contamination sources [26].
Troubleshooting Guides
Problem: Persistent False Positives in PCR
Potential Cause: Amplicon (PCR product) contamination in your workspace or reagents.
Solutions:
- Chemical Decontamination of Surfaces: Generously spray workstations, pipettes, and equipment with a 10% dilution of commercial bleach (e.g., Clorox). Let it sit for 15-30 minutes, then wipe and rinse with water to remove corrosive residues [32].
- Implement a Unidirectional Workflow: Maintain physically separate, dedicated areas for pre-PCR (reagent preparation, sample setup) and post-PCR (amplification, analysis) activities. Never bring PCR products back into the pre-PCR area [34].
- Use UV Decontamination: Run a full UV cycle in your PCR workstation between experiments to degrade any aerosolized amplicons on the work surface [35] [36].
- Discard Contaminated Reagents: If contamination is widespread, discard all opened reagents, including master mixes, primers, and water. Prepare fresh aliquots from sterile stocks [34].
Problem: Inconsistent PCR Results or Amplification Failure
Potential Cause: Nuclease contamination or reagent degradation.
Solutions:
- Eliminate Nucleases: Wipe down workbenches, pipettes, and your gloves with 70% ethanol before setting up reactions. Nucleases on skin or surfaces can digest DNA templates [37].
- Use Filter Tips and Aliquot Reagents: Always use aerosol-resistant filter tips. Aliquot all reagents (water, buffers, dNTPs, enzyme) into single-use amounts to prevent contaminating your entire stock [37].
- Ensure Proper Tube Closure: Close PCR tube caps carefully and slowly. Incompletely closed tubes can lead to evaporation, altering reaction concentrations and causing failure or inconsistency [37].
Problem: Suspected Loss of UV Decontamination Efficacy
Potential Cause: UV lamp intensity has degraded over time.
Solutions:
- Scheduled Maintenance and Certification: Perform annual certification of your PCR cabinet. This includes a UV intensity test to measure the light output along the work surface. Replace the UV lamp according to the manufacturer's schedule, typically every one to two years [33].
- Adhere to a Maintenance Checklist: Follow a comprehensive maintenance schedule that includes regular surface decontamination, pre-filter replacement, and inspection for physical abnormalities [33].
Experimental Protocols & Data
Protocol: Evaluating UV-Induced DNA Damage via RT-qPCR
This method assesses UV efficacy by measuring the degradation of a specific DNA target.
- Sample Preparation: Use a standardized sample containing the target nucleic acid (e.g., SARS-CoV-2 RNA genes like E or RdRp).
- UV Irradiation: Expose the sample to a range of UV-C doses (e.g., 0, 60, 300, 600, 900 mJ/cm²) in a controlled irradiation chamber [30].
- RNA Extraction and Reverse Transcription: Extract the RNA and perform reverse transcription to generate cDNA.
- Quantitative PCR (qPCR): Run the cDNA in a qPCR assay targeting the specific genes.
- Data Analysis: Calculate the cycle threshold (Ct) values. A significant increase in Ct value (ΔCt) for the UV-treated samples compared to the control (0 mJ/cm²) indicates successful UV-induced nucleic acid degradation [30].
Protocol: Surface Decontamination with Sodium Hypochlorite
- Prepare Fresh Bleach Solution: Dilute commercial bleach to a 10% (v/v) solution in clean water. For tougher contamination, a 5% solution (a 1:10 dilution of standard household bleach) can be used [32]. Note: The potency decreases over time; make fresh dilutions weekly.
- Application: Generously spray or wipe the solution onto the surface to be decontaminated (e.g., workbench, pipette exteriors).
- Contact Time: Allow the bleach to remain on the surface for 15-30 minutes [32].
- Rinsing: Due to its corrosiveness, wipe the surface with a cloth dampened with sterile water to remove any bleach residue [32].
- Safety: Wear appropriate PPE, including a lab coat, gloves, and safety glasses [32].
Quantitative Data Comparison
The tables below summarize key efficacy data for UV and chemical decontamination methods.
Table 1: UV-C Inactivation Efficacy Against Microorganisms This data demonstrates the wavelength-dependent nature of UV inactivation, with peak efficacy around 265 nm [31].
| Microbial Type | Example Strain | Log Reduction (approx.) | Most Effective Wavelength | Key Mechanism |
|---|---|---|---|---|
| Gram-Negative Bacteria | Escherichia coli | 3-log | 263-270 nm | DNA Damage (CPD formation) [31] |
| Gram-Positive Bacteria | Staphylococcus aureus | 3-log | 263-270 nm | DNA Damage (CPD formation) [31] |
| Bacterial Spores | Bacillus subtilis | 3-log | 263-270 nm | DNA Damage (CPD formation) [31] |
| Virus | SARS-CoV-2 | RNA degradation (Increased Ct value) | ~254 nm | RNA damage & strand breaks [30] |
Table 2: Chemical Decontamination Methods This table outlines the common uses and important considerations for two primary chemical agents.
| Chemical Agent | Typical Concentration | Primary Use | Key Advantage | Key Consideration |
|---|---|---|---|---|
| Sodium Hypochlorite (Bleach) | 0.5% - 5% (0.05-0.5% free chlorine) [32] | Surface decontamination, liquid waste | Effectively causes nicking and degradation of DNA, preventing amplification [32] | Corrosive; requires rinsing with water after use [32] |
| Ethanol | 70% | Inactivating microorganisms on surfaces, gloves [37] | Rapid evaporation, non-corrosive | Does not effectively remove pre-existing DNA traces [26] |
Workflow and Pathway Diagrams
UV Decontamination Decision Workflow
PCR Contamination Prevention Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagents and Materials for Decontamination
| Item | Function in Decontamination |
|---|---|
| PCR Workstation | Provides a controlled environment with HEPA-filtered laminar airflow to protect samples from airborne particles and a UV-C lamp for surface decontamination between uses [35] [36]. |
| Sodium Hypochlorite (Bleach) | A chemical agent that degrades DNA through oxidative nicking, preventing its amplification by PCR. Used for surface and liquid waste decontamination [32]. |
| 70% Ethanol | Used for wiping down surfaces, gloves, and equipment to inactivate microorganisms and remove nucleases. It is less effective than bleach at destroying free DNA [37]. |
| HEPA Filter | A high-efficiency particulate air filter that removes airborne contaminants (e.g., dust, spores, aerosols) from the air supplied to the PCR workstation [35] [36]. |
| Filter Pipette Tips | Contain an aerosol barrier to prevent cross-contamination of samples and reagents by pipetting [34] [37]. |
| UV-C Light Meter | A device used to measure the intensity of UV-C light emitted by a lamp, crucial for validating and certifying that the decontamination system is functioning correctly [33]. |
Implementing Effective UV Decontamination Protocols in Your Lab
In the context of molecular biology research, particularly in drug development, the integrity of PCR experiments is paramount. Contamination can lead to inaccurate results, failed experiments, and costly delays. This technical support center guide focuses on the two primary types of PCR workstations—UV-only and UV with HEPA filtration (Positive Pressure)—framed within ongoing research on UV irradiation for decontamination. Understanding the mechanisms, appropriate applications, and troubleshooting of these enclosures is a critical component of ensuring experimental validity and advancing scientific discovery.
Core Principles and Definitions
What is a PCR Workstation?
A PCR workstation, also known as a PCR cabinet or hood, is a self-contained, sterilized work zone that provides a contaminant-free environment for preparing PCR samples and reagents. Its primary function is to prevent cross-contamination between samples and from external sources (e.g., dust, microbial spores, or ambient DNA/RNA) during the sensitive steps of nucleic acid amplification and manipulation [5] [38]. By maintaining sustained air quality, it protects the integrity of samples until they are ready for use.
The Role of UV Irradiation in Decontamination
Ultraviolet Germicidal Irradiation (UVGI), specifically within the UV-C spectrum (200–280 nm), is a well-established decontamination method. Its primary mechanism of action is the absorption of UV-C photons by microbial DNA and RNA, which induces the formation of cyclobutene pyrimidine dimers (CPDs). These photoproducts disrupt the nucleic acid structure, preventing replication and transcription, thereby inactivating the microorganisms [29] [39] [40]. Research has shown that the efficacy of UVGI is wavelength-dependent, with peak bacterial inactivation occurring at 263–270 nm [39]. In a PCR workstation, UV lamps (typically rated at 254 nm) are used to decontaminate all exposed interior surfaces before and after use, eliminating contaminants that may have been introduced previously [5].
Comparative Analysis: UV-Only vs. UV+HEPA Workstations
The choice between a UV-only and a UV+HEPA workstation hinges on the specific contamination risks and procedural requirements of your experiments. The table below provides a structured comparison of these two types.
Table 1: Comparison of UV-Only and UV+HEPA PCR Workstation Types
| Feature | UV-Only (Still Air/Dead Air) Workstation | UV+HEPA (Positive Pressure) Workstation |
|---|---|---|
| Primary Contamination Control | UV surface decontamination and still air to minimize disturbance [5]. | Positive pressure and HEPA-filtered laminar airflow, supplemented by UV surface decontamination [5]. |
| Airflow Principle | Still (or "dead") air; no active circulation [5] [38]. | Positive pressure; blower forces HEPA-filtered air into the chamber, creating outward airflow [5]. |
| Ideal Application | Procedures highly sensitive to air currents; general tissue cultures where sample disturbance must be minimized [5]. | Preparation of PCR master mixes and handling of sensitive reagents; applications requiring the highest level of protection from airborne contaminants [5] [33]. |
| Contaminant Removal | Does not remove airborne particles; relies on stillness and UV to mitigate risk. | Actively removes airborne particles; the HEPA filter is rated at 99.999% efficiency for particles ≥0.3 microns [5]. |
| Temperature Uniformity | Poor; can experience stratification and layers of air, leading to temperature variations across the chamber [5]. | Excellent; forced air circulation ensures consistent temperature throughout the workspace [5]. |
| Key Limitation | Does not protect against airborne contaminants introduced during user access. | Not designed to protect the user from hazardous vapors or samples [5]. |
Experimental Protocols for Performance Validation
To ensure your PCR workstation is functioning correctly, regular performance validation is essential. The following protocols are standard in the field and should be conducted by trained professionals during annual certification [33].
UV Intensity Test
Objective: To verify that the UV lamp is emitting sufficient germicidal energy to decontaminate the work surface. Methodology:
- Ensure the workstation has been off and the chamber is closed.
- Using a calibrated UV radiometer, measure the UV light intensity (in µW/cm²) at several pre-defined points along the front-to-back centerline of the work surface.
- Compare the measured values against the manufacturer's specified minimum intensity.
- If the readings are below the specification, the UV lamp should be replaced, even if it has not reached its rated hourly lifespan [33].
Filter Integrity Test
Objective: To ensure the HEPA filter is properly sealed and has no leaks that could allow unfiltered air into the workspace. Methodology:
- A challenge aerosol (e.g., dioctyl phthalate DOP or salt) is generated upstream of the HEPA filter.
- A particle counter probe is used to scan the entire surface of the filter, its frame, and the gaskets for leaks.
- The output is measured, and the filter is deemed integral if the penetration is below the required threshold (e.g., 0.01%) [33].
Airflow Velocity Test
Objective: To confirm that the blower is providing the correct volume of HEPA-filtered air to maintain the positive pressure barrier. Methodology:
- Use a calibrated anemometer to measure the velocity of the air exiting the HEPA filter.
- Take multiple readings across the face of the filter to calculate an average airflow velocity.
- This measurement provides insight into the blower's performance and ensures it meets the design specifications for the cabinet [33].
The logical sequence and purpose of these certification tests are summarized in the diagram below.
Frequently Asked Questions (FAQs)
Q1: My PCR results are showing contamination. How can I determine if my workstation is the source? A1: First, run a negative control (no-template control) through your PCR process. If it shows amplification, contamination is confirmed. To isolate the workstation as the source, perform a surface swab test inside the cabinet after UV decontamination and use a particle counter to check for airborne particulates. Ensure your UV timer is set for an adequate duration (typically 15-30 minutes) and that the UV intensity has been recently certified [5] [33].
Q2: Can I use UV-C to decontaminate my samples or reagents directly inside the workstation? A2: No, you should never expose your samples or reagents to direct UV-C light. UV-C radiation is highly effective at damaging nucleic acids. Its purpose in the workstation is to decontaminate the static surfaces before you introduce your samples. Direct exposure will degrade the DNA/RNA in your samples and any enzymes (e.g., polymerase) in your master mix, ruining your experiment [5].
Q3: How often should I replace the UV lamp and HEPA filter in my workstation? A3: UV lamps have a finite lifespan and lose intensity over time. They should be replaced approximately every year or as per the manufacturer's guidance, but this should be verified with an annual UV intensity test [33]. HEPA filters do not have a set replacement schedule and are typically changed only when they fail the integrity test or when the airflow velocity drops below an acceptable level due to clogging [33].
Q4: What are the critical installation factors for a new PCR workstation to ensure optimal performance? A4: Proper installation is crucial. Key factors include:
- Location: Place the cabinet away from doors, high-traffic pathways, and direct airflow from air-conditioning vents to minimize disruptive air currents [33].
- Electrical: Use a dedicated power outlet, and consider a surge protector or UPS to ensure stable voltage and allow for safe shutdown during a power failure [33].
- Space: Ensure there is adequate clearance for maintenance and certification access.
Operational Procedures and Best Practices
Standard Decontamination Workflow
A consistent decontamination routine before and after every use is non-negotiable. The following workflow integrates both UV and HEPA systems.
Troubleshooting Guide
Table 2: Common PCR Workstation Issues and Solutions
| Problem | Potential Cause | Solution |
|---|---|---|
| Consistent contamination in samples. | 1. Ineffective UV decontamination.2. HEPA filter leak or failure.3. Poor aseptic technique. | 1. Test UV intensity and replace lamp if needed.2. Perform a filter integrity test.3. Review and reinforce sterile technique (e.g., frequent glove changes) [5] [33]. |
| Unusual noise from the blower. | 1. Blower motor failure.2. Obstruction in the air intake or pre-filter. | 1. Contact service technician.2. Check and clean or replace the pre-filter as per the maintenance schedule [33]. |
| UV lights do not turn on. | 1. Door/sash interlock sensor is activated.2. Lamp has reached end of life.3. Electrical fault. | 1. Ensure the sash is fully closed.2. Replace the UV lamp.3. Check power connection and contact electrician if needed [5]. |
| Reduced airflow from the HEPA filter. | 1. Clogged pre-filter or HEPA filter.2. Blower motor issue. | 1. Replace the pre-filter (quarterly or as needed).2. Perform airflow velocity test and contact for service [33]. |
The Scientist's Toolkit: Essential Research Reagents & Materials
The following table details key materials used in conjunction with PCR workstations to maintain a sterile environment and validate their performance.
Table 3: Essential Materials for PCR Workstation Operation and Validation
| Item | Function | Application Note |
|---|---|---|
| DNA/RNA Decontamination Solution | To chemically degrade contaminating nucleic acids on surfaces before UV irradiation. | Used for wiping down the interior work surface. Inactivates amplicons that may be resistant to UV [5]. |
| 70% Ethanol or Isopropanol | A general disinfectant to reduce microbial load on surfaces. | Used for routine cleaning of the workstation interior and exterior. Allows for physical removal of debris [33]. |
| Nuclease-Free Water | A critical negative control reagent to check for contamination in the PCR process. | Included in every PCR run to verify the sterility of reagents and the workspace [38]. |
| Calibrated UV Radiometer | A device used to measure the intensity of UV-C light emitted by the workstation's lamps. | Essential for the annual certification and performance validation of the UV decontamination system [33]. |
| Particle Counter | An instrument that counts and sizes airborne particles to verify the cleanliness of the HEPA-filtered air. | Used during certification to perform the particle count test and filter integrity test [33]. |
Fundamental Principles of UV Decontamination
How does UV light decontaminate a PCR workstation?
Ultraviolet Germicidal Irradiation (UVGI) utilizes short-wavelength UV-C light, primarily at 254 nm, to inactivate microorganisms. This wavelength is near the optimal germicidal effectiveness peak of 260-265 nm, which corresponds to the peak absorbance of nucleic acids [8] [41] [42]. The radiation damages microbial DNA and RNA through the generation of bipyrimidine photoproducts, such as thymine dimers. This photodamage inhibits cellular replication and prevents microorganisms from reproducing, effectively rendering them harmless [8] [43] [42]. For PCR work, this is crucial to prevent the amplification of contaminating nucleic acids, which can lead to false positive results.
What is the difference between UV decontamination and HEPA filtration?
UV decontamination and HEPA filtration are complementary technologies that address contamination in different ways:
- UV Decontamination: This is a "no-touch" surface and air disinfection method. It inactivates microorganisms by damaging their genetic material but does not remove them from the workstation [8]. Its efficacy can be limited by shadowing, where objects shield contaminants from direct UV exposure [8].
- HEPA Filtration: This is an air-cleaning process. HEPA (High-Efficiency Particulate Air) filters physically remove airborne particles, including bacteria and spores, from the air entering the workstation. In PCR workstations, this often creates a positive pressure environment, preventing external contaminants from entering the clean zone when the cabinet is open [5].
Many advanced PCR workstations integrate both technologies: HEPA filters maintain particulate-free air during use, while UV cycles are used to decontaminate surfaces before and after procedures [44] [5].
Step-by-Step Decontamination Protocol
Pre-Decontamination Preparation
- Clear the Work Surface: Remove all items from the workstation, including pipettes, racks, and tubes. UV light requires a direct line of sight to be effective; objects left inside will create shadows where contamination can persist [8].
- Wipe Down Surfaces: Thoroughly clean the work surface and interior walls with an appropriate disinfectant (e.g., 70% ethanol, isopropanol) followed by a rinse with sterile water. This removes chemical residues and physical debris that could shield microorganisms [33].
- Ensure Safety: Verify that the workstation's safety features, such as automatic UV shut-off switches, are functional. These switches should turn the UV lamps off immediately when the door or sash is opened to protect users from harmful UV exposure [44] [33].
Execution of UV Cycle
- Close the Access Door: Ensure the workstation is completely sealed.
- Set the UV Timer: Use the cabinet's touch screen or control interface to set the decontamination time. The required time depends on the UV intensity and the desired dose.
- Initiate the Cycle: Start the UV cycle. Do not open the workstation during this process. The UV indicator light, often red, will typically illuminate to show that the cycle is active [44].
- Allow Cycle Completion: The system will automatically turn off the UV lamps once the timer expires.
Post-Decontamination Verification
- Visual Inspection: After the cycle, visually confirm that the UV lights are off before opening the workstation.
- Routine Performance Validation: Regularly measure the UV intensity using a calibrated light meter (e.g., an ILT-2400 with a 254-nm sensor) to ensure the lamps are delivering the intended dose [43] [33]. Annual certification by a qualified professional is recommended to validate overall system performance, including UV intensity, HEPA filter integrity, and airflow [33].
Quantitative Data and Parameters
The effectiveness of UV decontamination is quantified by the UV Dose, which is a function of intensity and exposure time [42].
UV Dose (μJ/cm²) = UV Intensity (μW/cm²) × Exposure Time (seconds)
The following table summarizes key parameters for effective decontamination:
| Parameter | Typical Value / Range | Importance & Notes |
|---|---|---|
| Wavelength | 254 nm | Standard output of low-pressure mercury vapor lamps; close to DNA/RNA absorption peak [44] [41] [42]. |
| UV Intensity | Varies by device and distance | Measured in μW/cm². Decreases with the square of the distance from the lamp (Inverse Square Law) [8] [42]. Must be measured periodically. |
| Minimum Effective Dose | 20,000 - 40,000 μJ/cm² | A common target for surface decontamination. The required dose varies by microorganism type and strain [43] [42]. |
| Typical Exposure Time | 15 - 30 minutes | Time required to achieve the target dose depends on the initial intensity. Timers are often built into the workstation [44]. |
| Safety Exposure Limit | 6 mJ/cm² over 8 hours | The NIOSH recommended maximum occupational exposure to protect skin and eyes [8]. |
Troubleshooting and FAQs
My PCR results show contamination. Why did the UV cycle fail?
Contamination after a UV cycle can result from several factors:
- Shadowing: The most common cause. Ensure all items are removed from the work surface before the UV cycle. UV light cannot penetrate or bend around objects [8].
- Insufficient UV Dose: The exposure time might be too short for the given intensity. Recalculate the dose (Intensity × Time) and confirm your lamps are outputting sufficient intensity. Old or dirty lamps can lose effectiveness [42].
- Lamp Age: UV lamps degrade over time. Even if they still emit visible light, their UV output diminishes. Replace UV lamps according to the manufacturer's schedule, typically annually or after 8,000-9,000 hours of use [33] [42].
- Surface Contamination: Organic matter on the work surface can shield microbes. Always perform a chemical wipe-down before the UV cycle [33].
How often should I replace the UV lamps?
UV lamps should be replaced at least annually or after the manufacturer's specified number of service hours (e.g., 8,000 hours) [33]. The lamp's UV output decays over time. Relying on old lamps means your decontamination cycles are delivering a lower dose than intended, risking incomplete decontamination. Some protocols recommend changing lamps prophylactically every 9 months to ensure consistent performance.
Is the UV light in my workstation safe for my eyes and skin?
Yes, but only when safety protocols are followed. UV-C light at 254 nm is hazardous to skin and eyes and can cause photokeratitis (like a welder's flash) and skin burns [8] [45]. Modern PCR workstations are equipped with critical safety features:
- Automatic Shut-off: A safety switch must automatically cut power to the UV lamps the moment the door is opened [44] [5].
- UV-Blocking Viewing Panels: The viewing window is made of materials like Makrolon that block UV wavelengths below 400 nm, allowing you to see inside without risk [44].
- Warning Indicators: Red LED lights indicate when the UV cycle is active [44].
Never operate a UV workstation with disabled safety interlocks.
Experimental Protocol for Validating UV Efficacy
Researchers may need to validate the decontamination efficacy of their UV workstation. The following protocol, adapted from standardized methods, provides a framework for this validation [43].
Materials and Reagents
The following table details key reagents and materials required for the validation experiment:
| Item | Function |
|---|---|
| Test Microorganisms | Staphylococcus aureus (ATCC 14458, Gram-positive) and Escherichia coli (ATCC 25922, Gram-negative). Represents different UV sensitivities [43]. |
| Tryptic Soy Agar (TSA) / Broth (TSB) | Standard media for culturing and enumerating the test organisms [43]. |
| Phosphate Buffered Saline (PBS) | Used for serial dilutions and washing cell pellets to maintain osmotic balance [43]. |
| Plastic Coupons (e.g., 17.35 cm²) | Non-porous, inert surfaces on which microbes are placed for UV exposure, simulating workstation surfaces [43]. |
| UV Intensity Meter | Calibrated radiometer (e.g., ILT-2400) with a sensor specific for 254 nm to measure irradiance at the test surface [43]. |
Workflow and Procedure
The experimental workflow for testing UV efficacy involves preparing test organisms, exposing them to UV light, and then quantifying the reduction in viable cells.
Detailed Steps:
- Culture Preparation: Propagate test organisms (S. aureus and E. coli) in Tryptic Soy Broth (TSB) at 35±2°C for 24±2 hours. Centrifuge the culture, discard the supernatant, and wash the cell pellet three times in sterile PBS. Resuspend the final pellet in 0.1% Buffered Peptone Water to a concentration of ~10^8 CFU/mL [43].
- Coupon Inoculation: Aseptically dispense 100 µL of the cell suspension onto a sterile plastic coupon, forming multiple small droplets. Air-dry the inoculated coupons inside a biosafety cabinet to create a surface-bound biofilm [43].
- UV Exposure and Dosimetry: Place the dried coupons inside the PCR workstation at a specified distance from the UV lamps. Use a calibrated UV intensity meter to measure the irradiance (µW/cm²) at the coupon surface. Calculate the exposure time needed to deliver the target UV dose (e.g., 40,000 μJ/cm²) [43] [42].
- Post-Exposure Analysis: After UV exposure, immediately transfer each coupon to a known volume of recovery medium (e.g., 0.1% BPW) and vortex to resuspend the cells. Perform serial dilutions and spread plate onto Tryptic Soy Agar (TSA) plates. Incubate plates at 35±2°C for 24-48 hours and count the resulting colonies (CFU/mL) [43].
- Calculation: Compare the microbial counts from UV-treated coupons to untreated controls. The reduction is reported as Log Reduction = Log10(N₀/N), where N₀ is the CFU/mL from the control and N is the CFU/mL after UV treatment [43]. A 3-log reduction represents a 99.9% kill rate.
The polymerase chain reaction (PCR) is a cornerstone technique in molecular biology, renowned for its exceptional sensitivity in amplifying specific DNA sequences [46]. However, this great strength is also its primary vulnerability; the amplified DNA products, known as amplicons, are potent sources of cross-contamination that can compromise experimental results. A single aerosolized droplet containing billions of copies of a target sequence can contaminate reagents, equipment, and samples in subsequent experiments, leading to false positives, uninterpretable data, and eroded confidence in research outcomes. The most critical defense against this threat is the strategic spatial separation of pre- and post-PCR activities. This protocol, framed within research on UV irradiation for decontamination, establishes a comprehensive framework for laboratory design and practice to safeguard the integrity of PCR-based science. By creating distinct physical zones for sample preparation and amplification product analysis, laboratories can construct a formidable barrier against the most common source of PCR contamination.
Scientific Rationale: Why Spatial Separation is Non-Negotiable
The Nature of the Contaminant
PCR functions by exponentially amplifying a target DNA sequence, generating an immense quantity of copies [46]. These amplicons are stable, readily aerosolized during routine laboratory procedures like pipetting, tube opening, or centrifugation, and are present in concentrations that dwarf the initial target DNA in a sample. When introduced into a pre-PCR setup, they become unwanted templates, outcompeting the intended target and leading to amplification even in negative controls. The profound sensitivity of PCR, capable of detecting a single DNA molecule, means that even invisible levels of contamination are sufficient to ruin experiments [46].
The Mechanism of UVGI in Decontamination
Ultraviolet germicidal irradiation (UVGI), particularly in the UV-C spectrum (200-280 nm), is a well-established decontamination method that complements physical separation [47] [24]. Its mechanism of action is the photochemical degradation of nucleic acids. UV-C light is absorbed by nucleobases, inducing the formation of cyclobutene pyrimidine dimers and other photoproducts [48]. These lesions disrupt the base-pairing and helix structure, effectively preventing the DNA polymerase from replicating the template [49]. As research on UV irradiation for PCR workstation decontamination confirms, this inactivation renders amplicons non-amplifiable, thereby neutralizing the contamination risk [4]. It is crucial to note that UV-C's effectiveness is limited to surfaces and air it directly irradiates; it cannot decontaminate items inside drawers, cabinets, or other shadowed areas, underscoring the necessity of spatial segregation.
Implementing Spatial Separation: A Practical Guide
Defining the Physical Zones
A robust separation protocol mandates the establishment of three distinct, physically separated areas with unidirectional workflow.
- Pre-PCR Area (Clean Zone): This dedicated space must be reserved exclusively for activities involving pre-amplification steps. This includes reagent preparation, sample handling, and reaction setup. Access should be restricted and strictly controlled.
- Amplification Area (PCR Instrument Zone): This area houses the thermal cyclers. While the instruments themselves are contained, amplicon generation occurs here, classifying the entire area as a contamination risk.
- Post-PCR Area (Contaminated Zone): All activities involving amplified DNA must be confined to this zone. This includes opening reaction tubes, gel electrophoresis, and any downstream analysis.
Workflow and Traffic Control
The movement of personnel and materials must follow a unidirectional flow: from the Pre-PCR area to the Amplification area, and finally to the Post-PCR area. Reverse flow is strictly prohibited. Researchers should not enter the Pre-PCR area after having been in the Post-PCR area on the same day without a complete change of lab coat and gloves. A clear visual representation of this workflow and its critical control points is essential for training and compliance.
UVGI Decontamination Protocol for PCR Workstations
PCR workstations, including biosafety cabinets and dedicated hoods in the Pre-PCR area, are critical control points. Regular decontamination with UVGI is a key strategy to maintain an amplicon-free environment.
Detailed Methodology
- Pre-Cleaning: Before UV irradiation, thoroughly clean all interior surfaces of the workstation with a recommended laboratory disinfectant (e.g., 10% bleach, followed by 70% ethanol to remove residue). Use lint-free cloths to avoid scratches [4].
- UV-C Lamp Operation: Ensure the cabinet's UV-C lamps are free of dust. Close the cabinet sash and activate the UV-C sterilization cycle.
- Exposure Time: The decontamination efficacy is a product of irradiance and time (dose = irradiance × time). A minimum exposure time of 30 minutes is generally recommended to ensure sufficient UV-C dose is delivered to all surfaces [4].
- Safety Interlock: Never operate the UV-C lamps when the cabinet is occupied. UV-C exposure is harmful to skin and eyes [47]. Ensure safety interlocks are functional.
Monitoring and Maintaining UVGI Efficacy
UV-C lamps have a finite lifespan and their output degrades over time. Regular maintenance is non-negotiable for guaranteed decontamination.
- Lamp Lifespan: The functional lifespan of a UV lamp is typically 9,000 hours, but efficiency begins to decline after 5,000 hours [4].
- Replacement Schedule: Establish a proactive replacement schedule based on recorded usage hours. For labs without automated counters, a six-monthly replacement is a prudent practice [4].
- Verification: Use UV-C dosimeters or indicator cards periodically to verify that critical surfaces are receiving the intended germicidal dose.
Table 1: UV-C Lamp Efficiency Degradation Over Time
| Hours of Operation | Relative Output Efficiency | Recommended Action |
|---|---|---|
| 0 - 5,000 | 100% - 85% | Normal operation |
| 5,000 - 6,000 | ~85% | Monitor performance |
| 6,000 - 7,000 | ~70% | Plan for replacement |
| 7,000 - 9,000 | <70% | Replace lamp |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagent Solutions for UVGI and PCR Decontamination Research
| Item | Function/Description | Application in Protocol |
|---|---|---|
| UV-C Dosimeter | A device (e.g., ILT2400-UVGI) calibrated to measure UV-C irradiance (W/cm²) and cumulative dose (J/cm²) [49]. | Critical for validating and quantifying the UV-C dose delivered to surfaces, ensuring decontamination efficacy. |
| Biological Indicators (BIs) | In-house prepared coverslips plated with indicator organisms (e.g., E. coli DH5α) or commercial spores [49]. | Used to empirically test the germicidal effectiveness of a UVGI system by quantifying log-reduction in viable organisms. |
| Molecular Biology Grade Water | Nuclease-free, DNA-free water. | Essential for preparing PCR reagents in the Pre-PCR area to prevent introduction of nucleases or contaminating DNA. |
| 10% (v/v) Bleach Solution | A solution of sodium hypochlorite that degrades DNA. | Primary decontaminant for cleaning surfaces and equipment before UVGI treatment. Inactivates DNA through chemical oxidation. |
| 70% (v/v) Ethanol | A common laboratory disinfectant. | Used for wiping down surfaces after bleach to remove residue and for general decontamination of non-porous surfaces. |
Frequently Asked Questions (FAQs) and Troubleshooting
Q1: Our laboratory space is limited. Is spatial separation still achievable? A: Yes, with rigorous procedural controls. If dedicated rooms are impossible, use separate, dedicated cabinets or hoods for pre- and post-PCR work, placed at maximum distance. Implement strict temporal separation by performing pre-PCR work at the start of the day and post-PCR work at the end. The use of closed-system PCR platforms can also reduce amplicon release.
Q2: We follow the protocol but are still seeing sporadic contamination in our negative controls. What are the most likely causes? A: Sporadic contamination often points to a specific vector. Key areas to investigate include:
- Reagents: Test all PCR master mix components by preparing a "reagent-only" control.
- Contaminated Pipettes: Aerosols can contaminate the exterior and internal mechanisms of pipettes used in the Post-PCR area. Dedicate pipettes to each zone and decontaminate them regularly with UVGI or DNA-degrading solutions.
- Lab Coats and Gloves: Amplicons can accumulate on sleeves and gloves. Change lab coats frequently and always change gloves when moving from post-PCR to pre-PCR areas.
- Ineffective UV Decontamination: Verify that your UV-C lamps are within their operational lifespan and have been correctly maintained. A decline in lamp efficiency is a common, overlooked failure point [4].
Q3: Can I use UV-C to decontaminate my PCR reagents or samples? A: No, absolutely not. UV-C radiation will degrade the nucleic acids in your samples and the primers/enzymes in your reagents, destroying their functionality. UVGI is strictly for decontaminating surfaces and air by inactivating contaminating amplicons.
Q4: What is the minimum UV-C dose required to inactivate SARS-CoV-2, and does this relate to PCR amplicons? A: Studies have shown that a UV-C dose of 126 mJ/cm² at 254 nm can achieve complete viral RNA clearance [24]. While viral RNA and double-stranded DNA amplicons have different structures, this provides a benchmark. Research on N95 decontamination showed significant viral RNA degradation at doses as low as 60-300 mJ/cm² [48]. A dose of 100-1000 mJ/cm² is typically effective for general nucleic acid inactivation in a laboratory setting, but validation with biological indicators is recommended.
The strategic spatial separation of pre- and post-PCR areas is not merely a recommendation but a critical protocol for any laboratory dedicated to generating reliable molecular data. This systematic approach, integrating physical barriers, unidirectional workflow, and validated UVGI decontamination procedures, forms an integrated defense system against contamination. By adhering to these guidelines and maintaining vigilant monitoring and maintenance of equipment, researchers and drug development professionals can uphold the highest standards of data integrity and propel their work forward with confidence.
This technical support center article provides detailed troubleshooting and FAQs for the essential safety and decontamination features of UV PCR workstations, specifically addressing safety interlocks, timers, and reflective surfaces. Proper understanding and maintenance of these components are critical for preventing contamination in sensitive molecular biology experiments and ensuring user safety.
Core Technical Specifications
The table below summarizes the key technical features and their specifications as found in modern UV PCR workstations and cabinets.
| Feature Category | Specific Feature | Technical Specification & Performance Data |
|---|---|---|
| Safety Interlocks | UV Automatic Shut-off | Safety switch automatically turns UV light off when door is opened [28] [50]. |
| Physical Sash Protection | Sash position switch and overlap prevent UV light exposure [51]. | |
| UV Blocking Materials | Makrolon panels block all UV light below 400 nm [28]. | |
| Status Indicators | Red LED ambient light indicates when UV is on [28]. | |
| Timers | Digital Timer Control | Touch screen interface or digital timer to control all system functions [28] [50]. |
| Programmable Durations | Typical preset options (e.g., 15 min, 30 min) and custom settings up to 99 minutes or 24 hours [28] [50]. | |
| Reflective Surfaces & Materials | Work Surface | Antimicrobial coated stainless steel and aluminum for easy cleaning and contamination control [28]. |
| Interior Enclosure | Reflective polycarbonate and polypropylene construction [51]. | |
| UV Protective Glass | Glass panels with UV-protective film on sides and front [50]. |
Workstation Functionality and Workflow
The following diagram illustrates the logical relationship and workflow between the core safety and decontamination features of a UV PCR workstation.
Frequently Asked Questions (FAQs) and Troubleshooting
Safety Interlocks
Q1: The UV lamps do not turn on when I activate the timer. What could be wrong? First, check the position of the workstation's door or sash. All safety interlocks are designed to prevent UV lamp operation if the door is not completely closed [28] [51]. Ensure the door is firmly shut. If the problem persists, inspect the door switch mechanism for any visible obstruction or damage and contact technical service.
Q2: Is the blue light I see inside the cabinet the UV-C light? No. The human eye cannot see UVC wavelengths (254 nm) [52]. The blue light is from an inert gas in the lamp and serves as a visual indicator that the unit is powered on [52]. The primary, germicidal UVC output is invisible, so you must always rely on the safety interlocks and status indicators, not the visible light, to determine if UV is present.
Timers and UV Exposure
Q3: What is the recommended UV decontamination time for the workstation surface before I begin my experiment? A common effective decontamination time is between 15 to 30 minutes of UV exposure [28] [50]. The optimal duration can depend on the specific model and the level of contamination. Always refer to your manufacturer's manual for precise recommendations.
Q4: The touch screen timer interface is unresponsive. What should I do?
- Try a soft reset by turning the main power to the workstation off and on again.
- Check if the unit is receiving power by verifying that the power cord is securely connected and the outlet is functional.
- If the interface remains unresponsive, note any error codes and contact your manufacturer's technical support, as the control panel may require service or replacement.
Reflective Surfaces and Maintenance
Q5: What is the best practice for cleaning the reflective interior and work surface?
- Frequency: Clean before and after each use or as frequently as needed [53].
- Materials: Use a lint-free cloth and a compatible cleaning solution [53].
- Key Precaution: Avoid abrasive materials and aggressive chemicals [53]. For models with acrylic surfaces or special UV-protective coatings, avoid organic solvents and high-alcohol content cleaners (>20%), as they can damage the material [53]. Always wipe down with distilled water after decontamination to remove residue [53].
Q6: How often should I replace the UV lamps, and why is it necessary even if they still light up? UV lamps have a finite functional lifespan, typically around 9,000 hours [53]. However, their germicidal efficiency begins to decline significantly after approximately 5,000 hours of use [53]. A lamp that still emits visible light can have drastically reduced UVC output, leading to inadequate decontamination and compromised experiments. Implement a bi-annual replacement schedule or track usage hours to ensure consistent sterilization performance [53].
The Scientist's Toolkit: Essential Research Reagent Solutions
The table below lists key consumables and materials essential for the operation and maintenance of a PCR workstation.
| Item Name | Function / Application |
|---|---|
| HEPA Filter | In HEPA-equipped models, provides Class 100 vertical laminar flow air to maintain a particle-free work area and protect samples from airborne contaminants [28] [51]. |
| Carbon Pre-Filter | Protects the main HEPA filter by capturing larger particles and extending its operational lifespan [28]. |
| 254 nm UV Lamp | The primary germicidal source. Its 254 nm wavelength is optimal for damaging nucleic acids, effectively decontaminating surfaces and inactivating DNA/RNA fragments to prevent cross-contamination [28] [50]. |
| Lint-Free Wipes | Essential for cleaning interior surfaces without introducing fibers or scratches that could harbor contaminants [53]. |
| Compatible Cleaning Solution | A lab-approved disinfectant that effectively decontaminates surfaces without damaging the workstation's reflective polycarbonate, acrylic, or specialized coatings [53]. |
A Technical Support Guide for PCR Workstation Decontamination
This guide provides troubleshooting and best practices for researchers combining UV irradiation with chemical cleaning for PCR workstation decontamination, a critical process for preventing nucleic acid contamination in sensitive molecular biology experiments.
FAQs on Combined UV and Chemical Disinfection
1. Does combining UV-C light with bleach reduce disinfection efficacy?
A 2023 laboratory study found no significant difference in the efficacy of disinfection against Pseudomonas aeruginosa when using a combination of UV-C light and bleach compared to bleach alone. While there is a biologically plausible hypothesis that UV-C could decompose the active ingredient in bleach (sodium hypochlorite) via photolysis, the experimental data did not support this under the tested conditions. The combined treatment resulted in a >6.3 log₁₀ reduction, statistically equivalent to the >5.6 log₁₀ reduction from bleach alone [54].
2. Can UV light be used synergistically with other chemicals, like ethanol?
Yes, synergistic effects have been observed with ethanol. One study demonstrated that combined ethanol-UV treatments resulted in greater reductions in bacterial counts of foodborne pathogens than either treatment alone. The synergistic effect varied by microorganism, with log reductions ranging from 0.02 to 2.32 log CFU/ml greater than the expected additive effect [55].
3. What are the primary safety concerns when using UV-C disinfection systems?
- Human Exposure: UV-C radiation can harm skin and eyes and must not be used in occupied spaces [8] [56].
- Ozone Production: Some UV-C lamps emitting below 240 nm can generate ozone, which can cause respiratory tract irritation. Ensure proper ventilation or use lamps that do not produce ozone [8].
- Material Degradation: Prolonged UV exposure can degrade materials like certain plastics and high-pressure acrylics [8].
4. Why should UV be considered an adjunct to chemical cleaning, not a replacement?
UV technology is a powerful tool but is highly dependent on factors like line-of-sight, distance, and exposure time. Shadows, crevices, and soiled surfaces can shield microorganisms from UV light. Therefore, protocol-driven manual cleaning is essential to remove physical debris and reduce bioburden before UV application ensures comprehensive decontamination [8] [57].
Troubleshooting Common Issues
| Issue | Possible Cause | Solution |
|---|---|---|
| Inconsistent Decontamination | Surfaces not pre-cleaned before UV exposure. | Always perform manual cleaning with an appropriate disinfectant to remove organic soil and dust before UV treatment [8]. |
| UV Ineffective on Surfaces | UV lamps blocked from line-of-sight; shadowed areas. | Reposition the UV device or the items being treated. Use multiple UV sources for complex spaces [8]. |
| Reduced UV Intensity | Aging UV lamps or fouling on quartz sleeves. | Adhere to a strict lamp replacement schedule. Implement routine inspection and cleaning of quartz sleeves [58]. |
| Residual Contamination in PCR Workstation | Incorrect order of operations. | Establish and validate a strict protocol: 1. Manual cleaning with chemical disinfectant. 2. Allow surface to dry. 3. Apply UV-C decontamination cycle [54]. |
Experimental Data on Combined Disinfection Strategies
The following table summarizes key quantitative data from a study investigating the combined use of UV-C and bleach against Pseudomonas aeruginosa on stainless steel surfaces [54].
Table: Log Reduction of P. aeruginosa with Different Disinfection Methods
| Disinfection Method | Mean Log₁₀ Reduction | Statistical Significance (vs. Positive Control) |
|---|---|---|
| Positive Control (Untreated) | 0 | N/A |
| UV-C Light Only | > 2.5 log₁₀ | P < 0.001 |
| Bleach Only | > 5.6 log₁₀ | P < 0.001 |
| Bleach + UV-C Light | > 6.3 log₁₀ | P < 0.001 |
Conclusion from Data: While highly effective individually, the addition of UV-C to bleach did not yield a statistically significant improvement in disinfection efficacy under these laboratory conditions [54].
Detailed Experimental Protocol
Below is a methodology adapted from a study evaluating the combined efficacy of bleach and UV-C, providing a template for your own validation experiments [54].
Methodology for Testing UV-C and Bleach Combination
1. Surface Preparation:
- Use stainless steel coupons (e.g., 4 cm x 4 cm).
- Pre-clean all surfaces with warm water and disinfect with 70% ethanol to remove any residual bioburden. Validate cleaning with sterile swab samples cultured on nutrient agar [54].
2. Test Microorganism and Inoculum Preparation:
- Use a stationary-phase culture of a relevant organism like Pseudomonas aeruginosa PAO1, suspended in 3% Bovine Serum Albumin (BSA) to simulate organic soil.
- Confirm the inoculum concentration (e.g., ~10⁶ CFU/ml) via serial dilution and viable counts [54].
3. Surface Inoculation and Drying:
- Inoculate defined areas on the test surfaces (e.g., 160 µL for stainless steel).
- Spread the inoculum evenly with a sterile spreader and allow it to air-dry for one hour under controlled conditions [54].
4. Disinfection Arms:
- Arm 1: Negative Control. Surface is not contaminated.
- Arm 2: Positive Control. Surface is contaminated but not treated.
- Arm 3: UV-C Only. Expose contaminated surface to a measured dose of UV-C (e.g., 254 nm at 0.120 J/cm²).
- Arm 4: Bleach Only. Wipe the contaminated surface with a bleach germicidal wipe (e.g., 0.55% sodium hypochlorite) using a defined number of strokes.
- Arm 5: Bleach + UV-C. Wipe the surface with bleach, wait a set time (e.g., 2 minutes), then expose to the same UV-C dose [54].
5. Post-Treatment Quantification:
- Sample each treated area immediately with a pre-moistened sterile swab.
- Place each swab in a recovery medium, vortex to release bacteria, and incubate.
- Perform serial dilutions and plate on nutrient agar to determine the viable cell count (CFU/ml) [54].
6. Data Analysis:
- Calculate the mean log₁₀ reduction for each treatment arm compared to the positive control.
- Use statistical analysis (e.g., one-way ANOVA with a post-hoc test) to compare the efficacy across different treatment strategies [54].
Experimental Workflow for Combined Disinfection Testing
The Scientist's Toolkit: Research Reagent Solutions
Table: Essential Materials for Disinfection Efficacy Studies
| Item | Function / Specification | Example from Literature |
|---|---|---|
| UV-C Emitter | Provides germicidal irradiation at 254 nm. | Cross-Linker CL-508 (UVItec) [54]. |
| Chemical Disinfectant | Bleach-based wipe for manual cleaning. | Clorox Healthcare Bleach Germicidal Wipes (0.55% sodium hypochlorite) [54]. |
| Test Surfaces | Non-porous, defined material for consistent testing. | Stainless steel sheets [54]. |
| Organic Soil Simulant | Mimics dirty conditions; critical for realistic testing. | 3% Bovine Serum Albumin (BSA) [54]. |
| Test Microorganism | Relevant, resilient pathogen for challenge tests. | Pseudomonas aeruginosa PAO1 [54]. |
| Neutralizing Buffer | Halts disinfectant action post-treatment for accurate microbial counts. | Implied use in swab sampling and serial dilution [54]. |
| Culture Media & Agar | Supports growth for viable count quantification. | Nutrient agar plates [54]. |
For optimal decontamination of PCR workstations, establish a validated protocol that leverages the strengths of both chemical and UV methods: manual cleaning to physically remove contaminants followed by UV-C irradiation to inactivate residual nucleic acids, ensuring a pristine environment for your sensitive reactions [59] [8].
Technical Support Center
This technical support center provides troubleshooting guides and FAQs to help researchers implement robust unidirectional workflow and aseptic technique, specifically within the context of UV irradiation for PCR workstation decontamination research. These protocols are essential for maintaining the integrity of sensitive experiments in molecular biology and drug development.
Troubleshooting Guides
Problem: Recurring contamination in negative controls during PCR setup.
- Potential Cause 1: Breach in unidirectional workflow, introducing amplicons into the pre-PCR area.
- Solution: Strictly enforce a one-way movement of personnel and materials from pre-PCR to post-PCR areas only. Technologists who have worked in the post-PCR area should not re-enter the pre-PCR area on the same day [60].
- Potential Cause 2: Inadequate decontamination of the PCR workstation or reagents.
- Solution: Implement a rigorous cleaning protocol. Wipe all work surfaces, pipettors, and touchpoints with a 10-15% bleach solution (0.5-1% Sodium Hypochlorite), leave for 15 minutes, then remove residue with distilled water followed by 70% ethanol [60] [61]. Ensure the UV lamp in the PCR cabinet is functional and its efficiency has not degraded [4].
Problem: Inconsistent or failed PCR amplification despite sterile techniques.
- Potential Cause: Degradation of PCR reagents due to contaminated or improperly maintained equipment.
- Solution: Verify the sterility of all reagents and consumables. Use only autoclaved, RNase-, DNase-, and pyrogenase-free pipette tips [61]. Ensure the HEPA filters in the laminar flow cabinet are not clogged, as this reduces airflow efficiency and compromises the aseptic environment [4] [62].
Problem: UV irradiation in the biosafety cabinet is suspected to be ineffective.
- Potential Cause 1: UV lamp has exceeded its functional lifespan.
- Solution: UV lamps have a limited lifespan. Their functional efficiency begins to decline after 5,000 hours of operation, decreasing by about 15% for every additional 1,000 hours. The lamps should be replaced after 9,000 hours, or approximately every six months if used continuously [4].
- Potential Cause 2: Lamp is dirty or obstructed.
- Solution: Clean the UV bulb weekly with a suitable solvent to remove dust and grime that can block germicidal radiation [63].
- Potential Cause 3: Environmental factors reducing efficacy.
- Solution: Be aware that high humidity can absorb UV radiation, reducing its effectiveness. UV light also only provides surface-level disinfection and cannot penetrate shaded areas or layers of dust [63].
Frequently Asked Questions (FAQs)
Q1: What is the single most critical principle for preventing PCR contamination?
- A: Establishing and严格遵守 (strictly adhering to) a unidirectional workflow. This means physically separating pre-PCR (reagent preparation, sample handling) and post-PCR (amplification, analysis) activities. Never bring amplified products (amplicons) back into the pre-PCR area [60].
Q2: Can UV irradiation in my PCR cabinet replace chemical decontamination?
- A: No. UV light is a useful supplementary tool but is not a replacement for proper chemical cleaning. UV radiation has limitations: it is less effective in high humidity, only disinfects exposed surfaces, and can give a false sense of security if a "blue glow" is mistaken for germicidal efficacy [63]. Always perform a chemical decontamination routine before and after work.
Q3: How often should I change my gloves during PCR setup?
- A: Change gloves frequently. Always wear fresh gloves when starting work in the PCR area and change them immediately if you suspect they have become contaminated with template DNA or amplicons [60].
Q4: What type of laminar flow hood is suitable for PCR reagent preparation?
- A: A PCR cabinet or a Class II Biosafety Cabinet (BSC) is appropriate. These provide a HEPA-filtered, laminar flow of clean air over the work surface to protect the sample from environmental contamination. Note that "clean benches" which blow air toward the user are not safe for handling biological reagents and should not be used [64] [62].
Q5: What control is essential to monitor for contamination?
- A: Always include a No Template Control (NTC) in your PCR run. This is a reaction mix containing all reagents except the sample DNA. A positive result in the NTC indicates that one of your reagents or consumables is contaminated with template DNA or amplicons [60].
The table below consolidates key maintenance metrics for PCR workstations, crucial for scheduling and ensuring effective decontamination.
Table 1: PCR Workstation Maintenance Schedule and Specifications
| Component | Key Metric | Recommended Maintenance Frequency / Value | Notes / Purpose |
|---|---|---|---|
| UV Lamp Lifespan | Functional Lifespan | 9,000 hours [4] | |
| Efficiency Decline | Begins after 5,000 hours [4] | Efficiency drops ~15% per 1,000 hours after this point. | |
| UV Lamp Replacement | Based on Timer | Replace when 9,000-hour limit is reached [4] | For cabinets with automatic usage counters. |
| Without Timer | Replace every 6 months [4] | A standard prophylactic replacement interval. | |
| UV Radiation Intensity | At Work Surface | At least 40 μW/cm² [62] | Check manufacturer's specifications for your model. |
| Dust Filters | Inspection | Weekly visual check [4] | Check for dirt or damage. |
| Cleaning | Rinse with warm tap water monthly [4] | Allow to air dry completely before reinstalling. | |
| Surface Decontamination | Bleach Contact Time | 15 minutes [60] | Use 10-15% bleach on surfaces before wiping with water and 70% ethanol. |
| HEPA Filter | Shelf Life (unused) | Up to 10 years [62] | When stored properly in original packaging. |
Experimental Protocols
Detailed Methodology: Assessing UV Lamp Efficacy for Decontamination
This protocol is designed to validate the effectiveness of a PCR workstation's UV decontamination cycle, a critical step for any thesis research on UV irradiation.
1. Objective: To quantitatively assess the reduction of microbial load on exposed surfaces inside a PCR cabinet after a standard UV decontamination cycle.
2. Materials:
- PCR Cabinet with UV lamp and timer [62]
- Tryptic Soy Agar (TSA) plates or other general-purpose microbial growth media
- Sterile swabs
- Incubator (set at 30°C or 37°C)
- Laboratory notebook
3. Procedure:
- Pre-Decontamination Sampling:
- Before activating the UV light, open a TSA plate and place it in the center of the workstation's base. Remove the lid and leave it exposed for one hour [61].
- Simultaneously, moisten a sterile swab with a neutralizer (like Letheen broth to stop UV action post-sampling), swab a defined area (e.g., 5x5 cm) near the front grille, and streak it onto a separate TSA plate. Label this "Pre-UV - Surface."
- UV Decontamination:
- Close the cabinet sash and run the UV decontamination cycle for the standard duration (e.g., 30 minutes) [62].
- Post-Decontamination Sampling:
- After the cycle is complete and the UV lamp is off, open the sash and immediately place a new, open TSA plate in the same location for another hour.
- Repeat the surface swabbing in an adjacent area with a new swab and streak onto a new TSA plate. Label this "Post-UV - Surface."
- Incubation and Analysis:
- Seal all plates with parafilm and incubate them inverted at the appropriate temperature for 24-48 hours.
- Count the Colony Forming Units (CFUs) on each plate. A significant reduction (ideally to zero) in CFUs on the post-UV plates indicates effective decontamination.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for Aseptic Work
| Item | Function / Purpose |
|---|---|
| 10-15% Bleach Solution | Primary chemical decontaminant for work surfaces. It destroys DNA/RNA contaminants and inactivates microorganisms on benchtops and equipment [60]. |
| 70% Ethanol | Used for wiping down non-corrosive surfaces, tools, and gloves. It is effective against many bacteria and fungi and evaporates without leaving a residue [61]. |
| HEPA-Filtered PCR Cabinet | Provides an ISO Class 3 [62] or cleaner work zone by removing airborne contaminants through vertical laminar flow, protecting samples during PCR setup [62]. |
| UV-C Germicidal Lamp | Provides secondary, non-chemical decontamination of the workstation air and exposed surfaces by disrupting the DNA of microorganisms, preventing replication [4] [29] [63]. |
| Autoclaved Pipette Tips & Tubes | Sterile, aerosol-free consumables are fundamental to preventing the introduction of nucleases and microbes that can degrade samples or cause false results [61]. |
| No Template Control (NTC) | A critical quality control reagent containing all PCR components except the template DNA. It is essential for verifying the absence of contamination in reagents and the environment [60]. |
Workflow and Process Diagrams
Unidirectional PCR Workflow
UV Lamp Maintenance Check
Solving Persistent Contamination: Advanced Troubleshooting and Optimization
FAQ: Understanding NTCs and Contamination
What does amplification in my No-Template Control (NTC) indicate? Amplification in your NTC signifies contamination in your PCR reagents or reaction setup. An NTC reaction contains all real-time PCR components except the template, so any positive signal indicates the presence of contaminating nucleic acids [65]. This contamination can stem from various sources, including amplicon carryover from previous reactions, contaminated reagents, or laboratory environmental contamination.
What is the difference between random contamination and systematic reagent contamination?
- Random Contamination: Occurs sporadically when loading DNA templates into the PCR plate, showing amplification in some or all NTCs at varying CT values [66].
- Systematic Reagent Contamination: Affects one or more specific reagents, resulting in consistent amplification across all NTC replicates with similar CT values because the same amount of contaminating template is present in each reaction [66].
Why is UV irradiation important for PCR workstation decontamination? UV light induces thymidine dimers and other covalent modifications in DNA, rendering contaminating nucleic acids inactive as templates for further amplification [67]. This method sterilizes potential contaminants in the reaction setup and prevents their subsequent amplification. UV irradiation should be an integral feature of any PCR laboratory, with all pipettes and disposable devices stored in a UV light box after opening [67].
How does uracil-N-glycosylase (UNG) prevent carryover contamination? UNG is a DNA repair enzyme that recognizes and removes uracil residues from DNA. The technique relies on substituting uracil (dUTP) for thymine (dTTP) during PCR to generate amplification products with distinguishing characteristics. UNG selectively hydrolyzes and removes any contaminating amplification products containing dUTP from the PCR mix before amplification begins. The enzyme is subsequently inactivated at 95°C, allowing new amplification to proceed [67].
What are the limitations of UV decontamination? UV irradiation exhibits sub-optimal efficacy in sterilizing short (<300 nucleotides) and G+C-rich templates [67]. Its effectiveness also depends on the distance of nucleic acids from the light source, and nucleotides present in the PCR reaction mix may protect contaminating amplification products from UV irradiation. Additionally, UV irradiation may have deleterious effects on the enzyme Taq polymerase and oligonucleotide primers [67].
Troubleshooting Guide: NTC Contamination
Step 1: Interpret NTC Amplification Patterns
| NTC Amplification Pattern | Probable Contamination Source | Immediate Actions |
|---|---|---|
| Consistent amplification across all NTCs with similar CT values [66] | Contaminated master mix, water, or primers/probes [66] | Replace all reagents systematically; use new aliquots |
| Variable amplification in some NTCs with different CT values [66] | Cross-contamination during plate loading or aerosol contamination [66] | Improve pipetting technique; use filter tips; decontaminate work area |
| Late amplification (beyond cycle 34 for SYBR Green) [68] | Primer-dimer formation rather than true contamination [68] | Perform melt curve analysis; optimize primer concentrations |
Step 2: Implement Reagent Testing Protocol
- Prepare fresh NTCs with alternative reagents to identify the contaminated component
- Test each reagent individually by substituting with a new aliquot or different lot
- Include additional controls such as positive controls and internal positive controls to rule out inhibition [65]
- Document all reagent lot numbers and tracking information for traceability
Step 3: Execute Workspace Decontamination
- Clean work surfaces with 10% sodium hypochlorite (bleach), followed by ethanol to remove residue [67]. Note: 10% sodium hypochlorite must be made fresh daily and require a minimum contact time of 10 minutes [69].
- Decontaminate equipment with 70% ethanol followed by UV irradiation [69]
- UV irradiation protocol: Expose closed work areas (e.g., safety cabinets) to UV light for 30 minutes [69]. Ensure regular maintenance of UV lamps, as their efficiency declines after approximately 5,000 hours of operation [4].
Step 4: Verify Resolution
- Repeat NTC testing with fresh reagents in decontaminated workspace
- Monitor subsequent runs to ensure contamination does not recur
- Implement preventive measures to avoid future contamination events
Experimental Protocols
Protocol 1: Systematic Reagent Testing via NTC
Purpose: To identify the specific source of contamination when NTC amplification occurs.
Materials:
- Fresh aliquots of all PCR reagents (master mix, water, primers, probes)
- Sterile, DNA-free tubes and filter tips
- New personal protective equipment (gloves, lab coat)
Procedure:
- Prepare individual NTC reactions for each reagent combination:
- Test current master mix + new water
- Test new master mix + current water
- Test current master mix + current water + new primers
- Include a complete NTC with all new reagents as control
Amplify according to standard PCR protocol
Analyze results:
- If contamination persists only with specific reagent, replace that component
- If contamination persists across multiple combinations, proceed to full workspace decontamination
Document findings and update laboratory records
Protocol 2: PCR Workstation UV Decontamination
Purpose: To effectively decontaminate PCR workstations using UV irradiation as part of routine maintenance.
Materials:
- PCR workstation with functional UV lamp
- 70% ethanol or commercial DNA-destroying decontaminant
- Lint-free cloths
- UV protective equipment
Procedure:
- Pre-cleaning:
- Remove all reagents, samples, and equipment from the workstation
- Wipe all interior surfaces with 70% ethanol or validated DNA-decontaminant [69]
- Allow surfaces to air dry completely
UV decontamination:
- Close workstation shield completely
- Activate UV lamp for 30 minutes [69]
- Ensure no personnel are exposed to UV light during operation
Post-decontamination:
- Allow 5-minute ventilation period after UV cycle completes
- Verify UV lamp function by checking operational hours (replace after 9,000 hours) [4]
- Document decontamination in laboratory maintenance log
Quality Control:
- Monthly: Inspect dust filters, clean or replace as needed [4]
- Quarterly: Measure UV intensity along front-to-back centerline work surface [33]
- Annually: Perform full certification including airflow velocity, filter integrity, and particle count tests [33]
Workflow Visualization
Systematic NTC Contamination Troubleshooting Workflow
Research Reagent Solutions
Essential Materials for Contamination Control
| Reagent/Equipment | Function in Contamination Control | Implementation Notes |
|---|---|---|
| UNG (Uracil-N-Glycosylase) | Enzymatically destroys contaminating amplicons from previous reactions containing dUTP [67] | Add to PCR mix; incubate at room temperature for 10 min before amplification |
| 10% Sodium Hypochlorite | Causes oxidative damage to nucleic acids, preventing reamplification [67] [69] | Prepare fresh daily; 10 min contact time; not for metallic parts |
| DNA-Decontaminating Solutions | Commercial products validated to destroy DNA on surfaces [69] | Use according to manufacturer instructions; alternative to bleach |
| Filter Pipette Tips | Prevents aerosol contamination during pipetting [69] | Use in all pre-PCR areas; confirm fit with pipette brand |
| HEPA Filters | Removes particulate contamination from air supply in PCR workstations [5] | 99.9997% efficiency at 0.3 microns; replace according to schedule |
| UV Germicidal Lamps (254 nm) | Induces thymidine dimers in DNA, rendering it unamplifiable [67] [5] | Replace after 9,000 hours; efficiency declines after 5,000 hours [4] |
PCR Control Implementation
| Control Type | Purpose | Interpretation Guide |
|---|---|---|
| No-Template Control (NTC) | Detects reagent or environmental contamination [65] | Amplification indicates contamination; requires investigation |
| Positive Control | Verifies reaction efficiency and primer functionality [65] | Failure indicates reaction problems; success validates methodology |
| Internal Positive Control | Detects PCR inhibition in sample [65] | Absence with positive target suggests inhibition; absence with negative target validates result |
| No-RT Control (for RT-PCR) | Detects genomic DNA contamination in RNA samples [65] | Amplification indicates DNA contamination requiring DNase treatment |
| Extraction Control | Monitors nucleic acid extraction efficiency [69] | Failure indicates problems with extraction process |
Ultraviolet (UV) irradiation is a critical tool for decontaminating PCR workstations and ensuring the integrity of molecular biology experiments. Effective UV exposure inactivates contaminating DNA and RNA by creating thymine dimers and other photoproducts that block polymerase extension during amplification. This technical guide provides researchers with evidence-based protocols and troubleshooting advice for optimizing UV decontamination procedures across various experimental contexts, from eliminating pure amplicons to dealing with complex genomic DNA contaminants.
Frequently Asked Questions (FAQs)
Q1: How does UV irradiation actually decontaminate DNA in my PCR workstation?
UV irradiation, particularly at 254 nm, inactivates contaminating DNA by inducing the formation of cyclobutane pyrimidine dimers (primarily thymine dimers) in the DNA backbone. These photoproducts physically block the progression of DNA polymerase during PCR amplification, thereby preventing the replication of contaminants and eliminating false-positive results. This method effectively decontaminates surfaces, air, and reagents within the workstation when applied correctly [59] [70] [71].
Q2: Why is my PCR still showing contamination even after standard UV treatment?
Several factors could explain this:
- Insufficient Exposure Time: Short amplicons (e.g., <200 bp) require longer UV exposure times for complete inactivation compared to longer fragments [72].
- Shadow Effects: Complex workstation geometries or equipment inside the cabinet can create shadows that shield contaminants from direct UV light [73].
- Reagent Contamination: The UV source itself may be a contributor. If critical reagents like primers or water were contaminated before being placed in the workstation, UV will not fully resolve the issue, as the damage to oligonucleotides can also reduce PCR sensitivity [72] [74].
- UV Intensity Loss: UV lamps lose intensity over time. A lamp may be near the end of its operational lifespan, which is typically around 9,000 hours [70].
Q3: Can UV irradiation damage my PCR reagents?
Yes, this is a critical consideration. Prolonged or high-intensity UV exposure can damage reagents, particularly primers and polymerase. The same mechanism that inactivates contaminating DNA can also photodamage the primers, reducing their ability to anneal and extend, which ultimately diminishes the sensitivity of your PCR assay [72] [71]. It is essential to balance decontamination with reagent integrity.
Q4: What is the difference between an open UV lamp and a UV air recirculator?
Modern PCR workstations often combine both systems for comprehensive decontamination:
- Open UV Lamp: A direct, high-intensity lamp (typically 254 nm) mounted inside the cabinet. It is used for surface decontamination between experiments during unoccupied cycles. It requires the workstation to be empty of personnel due to safety risks [59] [70].
- UV Air Recirculator: A sealed unit containing a UV lamp that continuously irradiates air as it is drawn through the system by a fan. This provides continuous air decontamination during operation, inactivating airborne contaminants without exposing the user to UV light [70] [75].
Troubleshooting Guides
Problem 1: Persistent Low-Level Contamination
Symptoms: Consistent faint bands or signals in negative controls (NTCs), even after UV decontamination cycles.
| Possible Cause | Verification Method | Corrective Action |
|---|---|---|
| Insufficient UV exposure time | Check lab records for exposure duration. | Increase UV exposure time systematically, testing efficacy with NTCs after each adjustment. Target 15-30 minutes as a starting point [70]. |
| Shadowed work area | Visually inspect the cabinet for items blocking UV light. | Remove all non-essential items from the workstation during the decontamination cycle. Rearrange necessary equipment to ensure direct line-of-sight to the UV lamp. |
| Contaminated reagents | Test reagents by setting up NTCs with a fresh, unopened aliquot of each reagent. | Replace all reagents with new aliquots. Always aliquot reagents to avoid contaminating master stocks [19] [74]. |
| Ineffective UV lamp | Check the lamp's usage hours against its specified lifespan (~9,000 hours) [70]. | Replace the UV lamp if it is near or beyond its rated service life. |
Problem 2: Reduced PCR Sensitivity After UV Decontamination
Symptoms: Weaker signal from positive controls or a higher limit of detection after implementing or increasing UV decontamination protocols.
| Possible Cause | Verification Method | Corrective Action |
|---|---|---|
| Photodamage to primers | Run a PCR with a new aliquot of primers that have not been UV-exposed. | Do not expose primers to direct UV light. Prepare master mixes in the workstation after the UV cycle is complete and the area is safe, or use a separate, clean area for primer handling [72]. |
| Overexposure of sensitive reagents | Review protocol to see if polymerase or dNTPs are being exposed to UV. | Never subject enzyme mixes or nucleotides to direct UV irradiation. Use the workstation for sample and template handling, not for storing sensitive reagents. |
| General reagent degradation | Test PCR efficiency with a fresh set of all reagents. | Implement stricter aliquot practices and ensure all reagents are stored correctly at recommended temperatures [74]. |
Quantitative Data for UV Decontamination
The following table summarizes key parameters for effective UV decontamination based on equipment specifications and research findings. Note that optimal times can vary based on the specific contaminant and cabinet model.
Table 1: UV Decontamination Parameters for PCR Workstations
| Parameter | Typical Range / Value | Notes & Context |
|---|---|---|
| Optimal Wavelength | 254 nm (UV-C) | This wavelength is most effective for nucleic acid absorption and dimer formation [59] [70] [71]. |
| Surface Exposure Time | 15 - 30 minutes | Effective for surface decontamination between experimental runs; time depends on amplicon size and intensity [70]. |
| Air Recirculation Rate | 20 - 25 m³/h | The entire cabinet air volume is processed multiple times per hour for continuous decontamination [70] [75]. |
| UV Lamp Lifespan | ~9,000 hours | Lamp intensity decays over time; replace per manufacturer's schedule to maintain efficacy [70]. |
| Impact on Short Amplicons | Lower efficiency | Shorter DNA fragments (<200 bp) require longer exposure times for complete inactivation [72]. |
Table 2: Experimental Factors Influencing UV Inactivation Efficiency
| Experimental Factor | Impact on UV Efficacy | Recommendation |
|---|---|---|
| Amplicon Size | Inversely correlated; longer amplicons are inactivated more easily [72]. | For labs plagued by short amplicon contamination, consider significantly longer UV exposure times and confirm inactivation with rigorous NTCs. |
| Contaminant Type | Pure amplicons vs. complex genomic DNA may have different inactivation kinetics. | Genomic DNA from reagents is a common, challenging contaminant. A combined UV + EMA strategy has been shown to be effective [72]. |
| Primer Presence | UV exposure damages primers, reducing PCR sensitivity [72] [71]. | Keep primer stocks and master mixes containing primers shielded from all UV light sources. |
Experimental Protocols for Validation
Protocol 1: Validating UV Efficacy in Your Workstation
This protocol helps you determine the minimum effective UV exposure time for your specific setup.
Research Reagent Solutions:
- Target DNA: A known amplicon you commonly use (e.g., a 500 bp plasmid or PCR product).
- PCR Master Mix: Your standard mix, including primers for the target.
- Nuclease-free Water.
Methodology:
- Prepare Contaminated Surfaces: Pipette 1 µL aliquots of your target DNA (at a concentration typical of your experiments) onto several small, clean, UV-transparent surfaces (e.g., sterile microscope slides or PCR tube caps). Allow to air dry.
- UV Exposure: Place the contaminated surfaces inside your PCR workstation. Expose them to UV light for different time intervals (e.g., 0, 5, 10, 15, 30 minutes). Include an unexposed control (0 min) that remains outside the cabinet.
- Recovery: After exposure, elute the DNA from each surface by rinsing with a fixed volume of nuclease-free water.
- Amplification: Use the eluate as a template in your PCR reaction.
- Analysis: Run the PCR products on an agarose gel. The minimum UV exposure time that results in complete loss of the amplification band is your effective decontamination time for that specific amplicon.
Protocol 2: A Combined UV-EMA Decontamination Strategy
For persistent contamination, especially from reagents, a combined approach can be more effective than UV alone [72].
Research Reagent Solutions:
- Ethidium Monoazide (EMA) or Propidium Monoazide (PMA): Photoreactive DNA intercalating dyes.
- PCR Reagents: Master mix (without template), primers, water.
- Photolysis Device: A light source at 465-475 nm for activating EMA/PMA.
Methodology:
- UV Treat Reagents: Expose the bulk PCR master mix and water to UV light in the workstation for a predetermined time. Do not expose primers to UV.
- EMA Treat Primers: Mix your primer working stock with a low concentration of EMA (e.g., 50 µM). Incubate in the dark for 10 minutes, then expose to 465-475 nm light for 10 minutes to photo-activate the dye and cross-link any contaminating DNA.
- Assemble Reaction: Combine the UV-treated reagents with the EMA-treated primers and template.
- Amplification: Proceed with PCR and analysis. This two-pronged attack can achieve high sensitivity with a very low contamination rate [72].
Workflow and Relationship Diagrams
Diagram 1: A logical flowchart for troubleshooting PCR contamination using UV irradiation, guiding the user based on the type of contaminant identified.
Diagram 2: Experimental workflow for empirically determining the minimum effective UV exposure time for a specific amplicon in a given PCR workstation.
Troubleshooting Guides
Why is my PCR workstation failing to decontaminate samples effectively?
Several factors related to UV lamp performance can lead to ineffective decontamination of your PCR workstation.
- Reduced UV Intensity: UV lamps naturally degrade over time, leading to a lower output of germicidal 254 nm light. This reduced intensity may not deliver the lethal dose of energy required to inactivate contaminating DNA templates [76] [77].
- Devitrification (Clouding): The quartz glass of the lamp can develop microscopic white spots, a process known as devitrification. This clouding acts as a barrier, significantly reducing the transmission of UV light, particularly the shorter wavelengths critical for decontamination [76].
- Aging Electrodes and Blackening: Over the lamp's lifetime, the electrodes erode due to the high arc temperatures. Tungsten from the electrodes sputters and deposits on the inside of the quartz body, causing blackening at the ends, which compromises UV output [76].
- Mirror Coating Effect: Over-cooling can cause a combination of tungsten and mercury to condense on the inside of the lamp ends, creating a mirror-like coating that blocks UV radiation [76].
- Frequent Power Cycling: Frequently turning the UV lamp on and off accelerates degradation. The largest current surge and stress on the electrodes happen at ignition. Using a standby mode (e.g., keeping the lamp at 50% power) instead of full power cycles can prolong its life [76].
How can I diagnose a decaying UV lamp?
Diagnosis can be performed through a combination of visual inspection, performance testing, and equipment monitoring.
- Visual Inspection: Regularly inspect the lamp for clear signs of failure, including white spotting (devitrification), black deposits at the ends (electrode erosion), or a metallic, mirror-like coating on the glass interior [76] [78].
- Performance Validation with Experimental Controls: The most definitive method for a PCR lab is to run a decontamination validation assay. Expose a control sample containing a known amount of a common contaminant (e.g., a previous PCR amplicon) to the workstation's UV light for the standard duration. If subsequent PCR amplification of this control is still successful, it indicates ineffective UV decontamination [79].
- Monitor Operational Hours: Keep a strict log of the lamp's cumulative operational hours. Most lamps have a finite lifespan, and performance declines significantly as the end of that lifespan approaches [79] [77].
- Use UV Intensity Meters: If your workstation is equipped with a UV sensor or light intensity meter, monitor its readings. A gradual decline in intensity indicates the lamp is nearing the end of its useful life [80].
Frequently Asked Questions (FAQs)
How often should I replace the UV lamps in my PCR workstation?
Replacement schedules should be based on operational hours rather than calendar time. The general guideline for PCR workstations is to replace UV lamps every 8,000 to 12,000 hours of operation [77]. However, specific applications may require more frequent changes; for instance, some manufacturers recommend replacement after 1,500 hours to ensure no loss of intensity in sensitive environments [76]. For typical daily use in a lab, an annual replacement is a common and safe practice [79].
Can I still use a UV lamp that appears to be working?
It is not recommended. A UV lamp can continue to emit visible blue light long after its UV-C output has degraded to ineffective levels [78]. The lamp's ability to inactivate DNA is dependent on its UV intensity at 254 nm, which degrades faster than the lamp's physical life. Using a lamp beyond its rated hours poses a significant risk of DNA contamination in your experiments.
What is the impact of not replacing a UV lamp on schedule?
The primary impact is a greatly increased risk of false-positive PCR results due to carryover contamination from amplicons or other DNA templates in the workstation [81]. This can lead to wasted reagents, lost time repeating experiments, and compromised research data. One study noted that contaminated amplification products can compete with sample DNA for primers, potentially causing unexpectedly low PCR sensitivities or even false-negative results [81].
What maintenance can I perform to extend the life of my UV lamp?
- Proper Cleaning: Regularly clean the lamp and quartz sleeve with isopropyl alcohol wipes to remove dust, fingerprints, and other contaminants that can cause hot spots and accelerate devitrification. Always wear gloves and ensure the lamp is cool and powered off before cleaning [76] [82].
- Avoid Frequent On/Off Cycling: Minimize thermal stress on the electrodes by using the system's standby or idle mode instead of turning the lamp completely off between uses [76].
- Ensure Adequate Cooling: Verify that the workstation's cooling system is functioning correctly to prevent the lamp from overheating, which can cause bowing or deformation of the quartz tube [76].
The following tables consolidate key quantitative data for UV lamp operation and replacement in a research context.
Table 1: UV Lamp Lifespan and Performance Guidelines
| Application | Typical Lifespan (Hours) | Replacement Guideline | Key Performance Metric |
|---|---|---|---|
| PCR Workstations | 1,000 - 12,000 [76] [77] | Annually or per operational hours [79] | Effective DNA deactivation [79] |
| General UV-C Disinfection | 8,000 - 12,000 (Low-Pressure) [77] | When output degrades below efficacy threshold [77] | UV-C Intensity at 254 nm |
| Water Purification | 9,000 - 12,000 [77] | Annually [80] | Minimum UV Dose (e.g., 40 mJ/cm²) |
Table 2: UV Lamp Failure Modes and Diagnostic Signs
| Problem | Visual Signs | Impact on PCR Work |
|---|---|---|
| Devitrification | White, cloudy spots on quartz glass [76] | Reduced UV transmission; ineffective DNA decontamination [76] |
| Electrode Erosion / Blackening | Dark black deposits at lamp ends [76] | Significant drop in UV intensity [76] |
| Mirror Coating | Shiny, metallic coating on lamp interior [76] | Blocked UV light; failure to decontaminate [76] |
| Bowing/Deformation | Lamp tube sagging or bending [76] | Potential for lamp failure; uneven UV exposure [76] |
Experimental Protocols
Protocol 1: Validating UV Decontamination Efficacy
This protocol is used to verify that your PCR workstation's UV lamp is effectively inactivating DNA contaminants.
- Preparation of Target DNA: Resuspend a known quantity (e.g., 10-50 ng) of a previous PCR product in nuclease-free water [79].
- UV Exposure: Aliquot the DNA solution into a series of PCR tubes. Place the tubes, on their sides and capped, inside the PCR workstation.
- Irradiation Time Course: Expose the tubes to UV light for a set time course (e.g., 0, 10, 20, and 30 minutes) [79].
- PCR Amplification: After irradiation, use each aliquot as a template in a new PCR reaction with primers specific to the original amplicon.
- Analysis: Run the PCR products on an agarose gel. Successful decontamination is confirmed by the disappearance of the amplification band after a specific irradiation time (e.g., 20-30 minutes under the tested conditions) [79].
Protocol 2: Monitoring UV Lamp Output Degradation
This procedure provides a quantitative measure of the lamp's decaying performance.
- Baseline Measurement: When a new lamp is installed, use a UV radiometer to measure the light intensity at a standard distance and location within the workstation. Record this baseline value.
- Regular Monitoring: At regular intervals (e.g., monthly), repeat the intensity measurement under identical conditions.
- Data Tracking: Plot the intensity values over time. A decrease of 20% or more from the baseline intensity indicates the lamp should be replaced soon [79].
Workflow and Relationship Diagrams
The Scientist's Toolkit
Table 3: Essential Reagents and Materials for UV Decontamination Research
| Item | Function / Application |
|---|---|
| Isopropyl Alcohol Wipes | For cleaning the external surface of UV lamps and quartz sleeves to remove contaminants that can cause devitrification [76] [82]. |
| UV-Blocking Gloves | To protect skin from UV exposure and prevent oil contamination from fingerprints during lamp handling [76]. |
| UV Radiometer / Sensor | A device to quantitatively measure the intensity of UV-C light (254 nm) to track lamp performance and validate decontamination doses [80]. |
| Pre-amplified DNA Template | A known PCR product used as a positive control in experimental protocols to validate the efficacy of UV decontamination [79]. |
| UV Test Strips / Dosimeters | Chemical indicators that change color in response to cumulative UV exposure, providing a simple, instrument-free check of UV dose delivery [76]. |
In the sensitive world of polymerase chain reaction (PCR), the immense power of DNA amplification is a double-edged sword. The same technique that can detect a single DNA molecule is also vulnerable to false positives from minuscule contaminants, including the products of its own previous reactions. Advanced laboratories therefore employ a dual-armed defense strategy that combines physical decontamination with biochemical safeguards.
- Physical Decontamination: This involves the use of UV irradiation within PCR workstations to destroy nucleic acids on surfaces and in the air, providing a clean environment for setting up reactions [28] [83].
- Biochemical Safeguards: This utilizes the UNG/dUTP system (Uracil-N-Glycosylase/deoxyuridine triphosphate) to enzymatically destroy contaminating PCR products from earlier experiments, preventing their re-amplification [84] [85].
Integrating these two techniques provides a robust, multi-layered defense, significantly reducing the risk of contamination and ensuring the integrity of experimental results. This technical support center outlines the protocols and troubleshooting necessary to implement this combined approach effectively.
Frequently Asked Questions (FAQs)
Q1: What is the specific function of UNG in PCR, and how does it differ from UV decontamination?
UNG is a DNA repair enzyme that specifically targets and removes uracil bases from DNA strands. In PCR, it is used to degrade any contaminating PCR products that were previously synthesized using dUTP instead of dTTP. This enzymatic degradation occurs during the initial PCR step at 50°C, before the amplification cycle begins, effectively "cleaning" the reaction mix of carryover contamination [84]. UV decontamination, in contrast, is a physical method that uses 254 nm ultraviolet light to inactivate exposed DNA and RNA fragments on surfaces and in the air within a PCR workstation, typically before the experiment begins [28] [83]. While UV light provides a broad-spectrum clean-up of the work environment, UNG acts with precision inside the reaction tube itself.
Q2: Can I use a standard PCR workstation with UV light, or do I need a special model for optimal decontamination?
For optimal protection, a PCR workstation equipped with a HEPA filtration system is highly recommended alongside UV lighting [28] [5]. While a UV-only cabinet can effectively decontaminate surfaces, a HEPA filter continuously cleans the air entering the workstation, providing an ISO Class 5 (Class 100) clean environment. This is critical for protecting your samples from airborne contaminants and particulates. Many advanced workstations combine both features, offering surface decontamination via UV lamps and airborne contamination control via HEPA filtration, creating a positive pressure environment that keeps contaminants out [28] [5].
Q3: Are there any situations where I should NOT use the UNG/dUTP system?
Yes, there are several key scenarios where the UNG/dUTP system is not suitable:
- One-Step RT-PCR: If you are performing a one-step reverse transcription PCR, the UNG enzyme can degrade the newly synthesized cDNA that incorporates dU nucleotides during the reverse transcription step. A heat-labile UNG is a potential workaround [84].
- Post-PCR Analysis: If you plan to use the PCR amplicon for downstream applications like cloning or sequencing at a later date, UNG should be avoided as it can slowly degrade the product over time [84].
- Nested PCR: The system is not suitable for nested PCR protocols where the product from a first-round dUTP-containing PCR is used as a template for a second round, as UNG will degrade it [84].
- Bisulfite-Treated DNA: UNG will degrade bisulfite-converted DNA templates because bisulfite treatment converts unmethylated cytosine to uracil [84].
Q4: How long should I run the UV decontamination cycle in my PCR workstation to ensure it is effective?
While the exact time can depend on the specific workstation model and the intensity of its UV lamps, a common guideline is to decontaminate the chamber for at least 15 to 30 minutes before use [83] [75]. Some manufacturers design their UV bulbs for 1000 disinfection cycles to deliver adequate decontamination energy [86]. It is crucial to follow the manufacturer's recommendations for your specific equipment and to use the built-in digital timer to automate this process [28].
Q5: What safety precautions are necessary when working with UV-equipped workstations?
UV-C light (254 nm) is dangerous and can damage cells. Always ensure the workstation has safety interlock features that automatically turn off the UV lights when the door or sash is opened [28] [5]. The viewing panels should be made of materials like Makrolon or glass with a UV-protective film that blocks UV wavelengths, protecting the user from exposure during decontamination cycles [28] [75]. Never operate the UV lights with the cabinet open or the safety shields removed.
Troubleshooting Guides
UNG/dUTP System Troubleshooting
| Issue | Possible Cause | Solution |
|---|---|---|
| Poor or No Amplification | Too much dUTP in the dNTP mix, inhibiting PCR efficiency [85]. | Use a balanced dUTP:dTTP ratio. A mix of 175µM dUTP + 25µM dTTP is often effective [85]. |
| Residual UNG activity degrading new products during later PCR cycles [84]. | Ensure the initial denaturation step is sufficiently long and hot (e.g., 95°C for 2-10 minutes) to fully inactivate UNG. | |
| Incomplete Contamination Control | Contamination is from native (dTTP-containing) DNA, not previous dUTP-containing amplicons [84]. | UNG only degrades uracil-containing DNA. Implement rigorous lab practices and UV decontamination to control all contamination sources [83]. |
| Primer design is suboptimal for UNG degradation of primer-dimers [84]. | Design primers with dA-nucleotides near their 3' ends to ensure efficient degradation of primer-dimers by UNG [84]. |
UV Decontamination System Troubleshooting
| Issue | Possible Cause | Solution |
|---|---|---|
| Reduced UV Decontamination Efficiency | Aging UV lamp with diminished output [58]. | UV bulbs are typically designed for a finite number of cycles (e.g., 1000 cycles) [86]. Replace lamps as per the manufacturer's schedule. |
| Dirty or fouled quartz sleeves or lamp surfaces [58]. | Clean the UV lamps and quartz sleeves regularly with an appropriate solvent like 70% ethanol to remove dust and debris that can block UV light [58]. | |
| Shadows or obstacles in the work chamber. | Organize the interior to ensure a clear line of sight from the UV lamp to all critical surfaces. Avoid storing unnecessary items in the workstation. | |
| Positive Control Failure | UV light is decontaminating reagents or samples. | Never expose your master mix, primers, dNTPs, or DNA templates to UV light. Only use UV for decontaminating the empty workstation and non-porous equipment. |
Experimental Protocols & Workflows
Protocol: Establishing a dUTP-UNG Workflow
This protocol is adapted from the method used with GoTaq DNA Polymerase to ensure compatibility between the polymerase, dUTP incorporation, and UNG activity [85].
1. Reagent Preparation (In a UV-decontaminated HEPA PCR Workstation)
- Prepare a PCR master mix on ice. For a 50µl reaction:
- 10µl 5X Reaction Buffer
- 1µl Primer Mix (forward and reverse, 50µM)
- 5µl dNTP Mix (containing 2mM each of dATP, dCTP, dGTP)
- Variable: dUTP:dTTP Mix (See Table below for options)
- 0.25µl DNA Polymerase
- 1µl UNG Enzyme
- Nuclease-Free Water to 49µl
- Gently mix and briefly centrifuge.
- Aliquot 49µl of the master mix into each PCR tube.
Recommended dUTP:dTTP Ratios for Robust Amplification [85]:
| dUTP (µM) | dTTP (µM) | Final Concentration | Relative Amplification Efficiency |
|---|---|---|---|
| 0 | 200 | dTTP only (Control) | ++++ |
| 100 | 100 | 1:1 Ratio | +++ |
| 150 | 50 | 3:1 Ratio | ++ |
| 175 | 25 | 7:1 Ratio (Recommended) | ++ |
| 200 | 0 | dUTP only | Inconsistent |
2. UNG Contamination Clean-up Step
- Place the tubes in the thermocycler.
- Incubate at 50°C for 2-10 minutes. This allows UNG to actively degrade any contaminating uracil-containing DNA [84] [85].
3. PCR Amplification
- Proceed with a standard PCR protocol, ensuring the first step is a long denaturation at 95°C for 2-10 minutes to completely inactivate the UNG enzyme before cycling begins [84].
- Example cycling parameters:
- 95°C for 2 min (UNG inactivation & initial denaturation)
- 35-40 cycles of: 95°C for 30s, 55-60°C for 30s, 72°C for 1 min/kb
- 72°C for 5 min (final extension)
Workflow: Integrating UV and UNG/dUTP in the PCR Laboratory
For maximum contamination control, physical separation of laboratory areas and a unidirectional workflow are essential [83]. The following diagram illustrates how UV decontamination and the UNG/dUTP system fit into this overall workflow.
Integrated Contamination Control Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Function | Key Considerations |
|---|---|---|
| UNG/UDG Enzyme | Enzymatically degrades carryover contamination from previous dUTP-containing PCRs [84]. | Standard E. coli UNG is heat-inactivated. For one-step RT-PCR, use a heat-labile version [84]. |
| dUTP Nucleotide | A substitute for dTTP; incorporated into PCR products, making them susceptible to future degradation by UNG [85]. | Use in a mixture with dTTP (e.g., 175µM dUTP:25µM dTTP) for consistent amplification efficiency [85]. |
| PCR Workstation with UV & HEPA | Provides a clean air environment (HEPA) and surface decontamination (UV) for setting up reactions [28] [5]. | Look for automatic UV shut-off, a timer, and a low-noise blower (<40 dBA). HEPA filtration is critical for airborne contamination control [5]. |
| Aerosol Barrier Pipette Tips | Prevent the creation and uptake of aerosols, reducing sample-to-sample cross-contamination during pipetting [83]. | Essential in all pre-PCR areas. Use in conjunction with good pipetting technique. |
| Surface Decontaminants | Destroy DNA on benchtops and equipment. | 10-15% sodium hypochlorite (fresh bleach) is effective. If using 70% ethanol, follow with UV irradiation for full decontamination [83]. |
The exquisite sensitivity of the Polymerase Chain Reaction (PCR) is a double-edged sword; while it enables the detection of minute quantities of nucleic acids, it also makes the technique exceptionally vulnerable to contamination from previously amplified products (amplicons) or other foreign DNA [67]. A single PCR can generate as many as 10^9 copies of a target sequence, and the aerosolized amplicons can contaminate laboratory reagents, equipment, and ventilation systems, leading to false-positive results [67]. Ultraviolet (UV) irradiation has long been established as a critical technique for decontaminating PCR workstations and reagents to prevent such false positives [71]. The method relies on the property of UV light, particularly at 254 nm, to induce thymidine dimers and other covalent modifications in DNA. These lesions render contaminating nucleic acids unable to function as templates for the DNA polymerase, thus sterilizing the reaction setup [67]. However, the efficacy of this decontamination is not guaranteed and hinges on avoiding common pitfalls related to inadequate exposure, suboptimal workflow, and insufficient measures against cross-contamination. This guide outlines these pitfalls and provides detailed, actionable protocols to ensure robust and reliable PCR decontamination.
Troubleshooting Guide: UV Decontamination Issues
This section addresses frequently encountered problems in UV-based decontamination, their likely causes, and evidence-based solutions.
Table: Troubleshooting Common UV Decontamination Problems
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Persistent PCR Contamination | 1. Inadequate UV Exposure Time or Distance: Short amplicons (<300 bp) or G+C-rich templates are more resistant [67].2. Shielding by Reagents: Nucleotides in the PCR mix can protect contaminating DNA from UV rays [67].3. No Pre-Irradiation of Reagents: Contaminants are present in master mix, water, or primers [71]. | 1. Optimize Exposure: Validate and use longer exposure times (e.g., 15-30 min) [70] [75]. Ensure the workstation is designed for minimal distance between the lamp and work surface [70].2. Irradiate Before Mixing: Expose all non-template reagents (water, buffer, dNTPs, primers) to UV light in a thin layer before adding the template enzyme [71].3. Use Enzymatic Control: Incorporate uracil-N-glycosylase (UNG) into the PCR protocol for a dual sterilization approach [67]. |
| Nonspecific Amplification or Smears | 1. Carryover Contamination: Aerosolized amplicons from post-PCR areas have contaminated the pre-PCR area [87].2. Suboptimal PCR Conditions: Issues unrelated to UV, such as primer design or cycling parameters [25]. | 1. Enforce Unidirectional Workflow: Strictly separate pre-PCR (reaction setup) and post-PCR (analysis) areas. Never bring equipment or reagents from the post-PCR area back [87].2. Decontaminate Surfaces: Clean workstations with 10% sodium hypochlorite (bleach), which causes oxidative damage to nucleic acids, followed by ethanol to remove the bleach [67].3. Optimize PCR Protocol: Increase annealing temperature, use hot-start polymerases, or redesign primers [25]. |
| Reduced PCR Sensitivity | 1. UV Damage to Critical Reagents: Overexposure of primers or Taq polymerase to UV light [67].2. Residual UNG Activity: If using UNG, incomplete inactivation can degrade newly synthesized amplicons [67]. | 1. Protect Sensitive Reagents: Do not expose template DNA or the DNA polymerase to UV irradiation. Add these components after the decontamination step [67].2. Ensure UNG Inactivation: Follow manufacturer guidelines for UNG. Ensure a 95°C incubation step is performed to fully inactivate the enzyme before amplification begins. Store PCR products at -20°C or 72°C if UNG is a concern [67]. |
| Uneven or Inconsistent Decontamination | 1. Poor UV Cabinet Design: Lack of air recirculation leads to "shadow" areas [70].2. Old or Uncalibrated UV Lamps: Lamp intensity diminishes over time [88]. | 1. Use a UV Cabinet with Recirculation: Select a workstation with a patented UV air recirculator that continuously processes the internal air (e.g., 20-25 m³/h) for uniform decontamination [70] [75].2. Regular Maintenance: Adhere to a maintenance schedule. Monitor lamp life (typically ~9,000 hours) and replace lamps proactively. Use a UV dosimeter to validate surface irradiance [70] [88]. |
Experimental Protocols for UV Decontamination
Protocol: Validating UV Efficacy in Your Workstation
This protocol describes a method to experimentally verify the decontamination efficiency of a UV workstation.
Purpose: To confirm that the UV irradiation cycle in a PCR workstation effectively destroys a known quantity of contaminating DNA amplicons.
Principle: A solution containing a specific amount of a previous PCR product (amplicon) is exposed to UV light. The ability to re-amplify this product post-exposure is tested. A successful decontamination will show no amplification.
Materials:
- UV PCR workstation (e.g., UVP, Biosan) [59] [70]
- Purified PCR amplicon (e.g., 100 pg of a common target like a plasmid clone) [67]
- Complete PCR master mix (excluding template)
- Equipment for PCR and gel electrophoresis
Method:
- Preparation: Dilute the purified amplicon to a concentration of 100 pg/µL.
- Spotting: Pipette 10 µL droplets (containing 1 pg of amplicon) onto the clean, dry work surface of the UV cabinet at several locations, including the center and corners.
- Drying: Allow the droplets to air dry completely under ambient conditions inside the closed cabinet.
- UV Exposure: Initiate a standard UV decontamination cycle (e.g., 15-30 minutes at 254 nm) [70] [75]. Ensure the safety interlock is functional and the UV light turns off when the door is opened.
- Recovery: After the cycle, resuspend each dried spot with 20 µL of nuclease-free water by pipetting up and down.
- Amplification Test: Use 5 µL of the resuspended liquid as a template in a fresh 25 µL PCR reaction. Include two controls:
- Positive Control: A reaction with 1 pg of the same, non-UV-exposed amplicon.
- Negative Control: A reaction with nuclease-free water.
- Analysis: Run the PCR and analyze the products on an agarose gel. Successful decontamination is confirmed by the absence of a band in the test reactions, while the positive control shows a strong band.
Protocol: Incorporating UV and UNG for Robust Contamination Control
This protocol combines pre-amplification UV sterilization of reagents with enzymatic sterilization using uracil-N-glycosylase (UNG), providing a powerful dual barrier against carryover contamination [67].
Purpose: To establish a PCR setup workflow that eliminates potential DNA contaminants present in reagents and from previous amplifications.
Principle: UV irradiation damages environmental contaminants on surfaces and in reagent droplets. UNG selectively degrades any contaminating amplicons that contain dUTP (incorporated instead of dTTP), while leaving the native (dTTP-containing) target DNA untouched.
Materials:
- UV PCR workstation
- PCR reagents: dNTP mix including dUTP, UNG enzyme, hot-start DNA polymerase, primers, reaction buffer
- Template DNA
Method:
- Workstation Preparation: Turn on the UV lamp in the empty, closed workstation for a minimum of 15 minutes to decontaminate the interior surfaces [70].
- Master Mix Preparation: Inside the UV-irradiated cabinet, prepare the PCR master mix on ice. Include all components except the template DNA and the DNA polymerase:
- Nuclease-free water
- Reaction Buffer
- dNTP/dUTP mix
- Forward and Reverse Primers
- UNG enzyme (e.g., 0.5 - 1.0 U per reaction)
- Pre-Irradiation of Master Mix (Optional but Recommended): Dispense the master mix (without enzyme or template) into the reaction tubes and expose these open tubes to an additional 5-10 minutes of UV light inside the cabinet to sterilize the liquid contents [71]. Note: This step may affect very short primers and should be validated.
- Add Template and Polymerase: After the UV cycle, remove the tubes from the workstation. Add the template DNA and hot-start DNA polymerase to each tube. Cap the tubes.
- UNG Incubation: Place the tubes in the thermal cycler and run a hold at 25°C for 10 minutes. During this time, UNG will hydrolyze any uracil-containing contaminating DNA.
- UNG Inactivation and PCR: Heat the samples to 95°C for 2-5 minutes. This step simultaneously inactivates the UNG and activates the hot-start polymerase, initiating the PCR cycles.
Workflow & Safety Diagram
Diagram: Optimal UV Decontamination Workflow for PCR. This flowchart illustrates the strict unidirectional process for preventing amplicon carryover contamination, integrating both physical separation and UV decontamination steps.
The Scientist's Toolkit: Essential Reagents & Equipment
Table: Key Materials for Effective UV Decontamination and Contamination Control
| Item | Function & Rationale |
|---|---|
| UV PCR Workstation | Creates a contained environment for reaction setup. Features a 254 nm UV lamp for surface/air decontamination, a safety interlock to disable UV when open, and UV-blocking panels (e.g., Makrolon) for user protection [59]. |
| HEPA/Filtration System | (Optional in some models) Provides continuous air recirculation and filtration (e.g., carbon pre-filter + HEPA) to remove airborne contaminants, working in concert with UV irradiation [59]. |
| Uracil-N-Glycosylase (UNG) | A bacterial enzyme used for pre-amplification sterilization. It selectively degrades any contaminating PCR amplicons from previous reactions that contain dUTP, while leaving the native target DNA (with dTTP) intact [67]. |
| dUTP | A nucleotide that is incorporated into newly synthesized PCR products in place of dTTP. This "tags" the amplicons, making them susceptible to degradation by UNG in subsequent reaction setups, thus preventing carryover contamination [67]. |
| Sodium Hypochlorite (Bleach) | A chemical decontaminant used to clean work surfaces. A 10% solution causes oxidative damage to nucleic acids, rendering them unamplifiable. It is crucial for cleaning spills and decontaminating equipment that must move between zones [67]. |
| UV Dosimeter | A colorimetric indicator used to visually validate the dose of UV irradiation delivered to surfaces. It is a critical tool for quality control, ensuring that the UV lamps are functioning correctly and that all areas of the work surface receive adequate exposure [88]. |
Frequently Asked Questions (FAQs)
Q1: Why is my PCR still contaminated even after using a 15-minute UV cycle? A: The efficacy of UV light depends on several factors. Short amplicons (<300 bp) and G+C-rich templates are more resistant to UV inactivation [67]. Furthermore, the presence of reagents can shield contaminating DNA. The solution is to optimize and potentially lengthen the exposure time and, critically, to expose all non-template reagents (master mix without enzyme) to UV light in a thin layer before adding your template and polymerase [71]. For maximum protection, combine UV with enzymatic methods like uracil-N-glycosylase (UNG) [67].
Q2: Can UV light damage my PCR reagents? A: Yes, this is a critical consideration. Prolonged or intense UV irradiation can damage primers and inactivate the DNA polymerase [67]. Best practice is to use the UV cycle to decontaminate the empty workstation and then prepare your reactions inside it. If you choose to irradiate the master mix directly (a technique for sterilizing the reagents themselves), you must first omit the DNA polymerase and template, and you should empirically validate that this step does not inhibit your subsequent PCR.
Q3: What are the most important lab practices to prevent PCR contamination beyond using a UV cabinet? A: The single most important practice is maintaining strict physical separation of pre-PCR and post-PCR areas [87]. This must be a unidirectional workflow: once you or any equipment enters the post-PCR area (where amplified products are handled), you must not return to the pre-PCR area (where reactions are set up) without rigorous decontamination. Use dedicated equipment, lab coats, and aerosol-filter pipette tips for each area. Consistently include a negative control (no template) in every run to monitor for contamination [87].
Q4: How often should I replace the UV lamp in my workstation? A: UV lamps have a finite lifespan, and their intensity diminishes over time, leading to inadequate decontamination. Consult your manufacturer's specifications (e.g., some models cite a ~9,000-hour lamp life) [70]. The most reliable method to determine replacement need is to use a UV dosimeter, which provides a visual confirmation that the required irradiance dose is being achieved on the work surface [88].
Troubleshooting Guides
Troubleshooting UV Decontamination
Problem: Inconsistent Decontamination Results Post-decontamination cultures show contamination, or PCR controls indicate contamination in your workstation.
| Possible Cause | Diagnostic Steps | Recommended Solution |
|---|---|---|
| UV bulb exhaustion | Verify bulb usage hours against manufacturer's 1000-cycle typical lifespan [86]. | Replace UV bulb if it has exceeded its rated lifetime. |
| Insufficient exposure time | Confirm that the UV timer is set correctly to ensure adequate exposure [62]. | Follow a validated decontamination protocol; do not reduce the calculated UV exposure time. |
| Shadowed or obstructed areas | Audit the chamber interior for items that might block UV light from reaching all surfaces. | Remove all unnecessary items before decontamination and ensure the UV lamp is clean and unobstructed. |
| Poor chamber seal | Check the gaskets and hinges for damage or wear that could break the airtight seal [5]. | Ensure doors close completely; service or replace damaged seals to maintain the controlled environment. |
Problem: Positive Control Failure in Environmental Monitoring Your positive process control (synthetic RNA) fails to amplify, indicating potential inhibition in your sample.
| Possible Cause | Diagnostic Steps | Recommended Solution |
|---|---|---|
| Swab or collection kit interference | Test different swab types (e.g., synthetic tip) and transport media with your positive control. | Use validated, DNAse/RNAse-free swabs and transport media designed for molecular applications [89]. |
| Inhibition from surface residues | Re-swab the same surface area with a smaller swabbed area (e.g., 50 cm² instead of 100 cm²). | Re-train staff on standardized swabbing technique; consider diluting the extracted nucleic acid to overcome inhibition [89]. |
| RNA degradation | Check RNA extraction kit expiration dates and ensure cold chain during sample storage. | Ensure samples are stabilized in a guanidine-based solution immediately after collection and stored at -20°C until extraction [89]. |
Frequently Asked Questions (FAQs)
What is the primary purpose of a PCR workstation? A PCR workstation provides a clean, contaminant-free environment primarily to protect your samples (reagents and preparations) from cross-contamination, especially during sensitive procedures like PCR mastermix preparation. It is crucial to note that standard PCR cabinets do not provide user protection and should not be used with infectious materials [62].
How does a UV + HEPA PCR workstation differ from a dead air box? A dead air box is a non-ventilated enclosure with stationary, or "dead," air inside [62]. A UV + HEPA PCR Workstation actively circulates air through a HEPA filter, providing an ISO Class 5 clean environment, and includes UV light for surface sterilization, offering superior protection against contamination [86].
What does "positive pressure" mean in a PCR workstation, and why is it important? Positive pressure means the air pressure inside the workstation chamber is greater than the pressure in the surrounding room. This pressure differential causes clean, HEPA-filtered air to flow out of any small openings in the chamber, preventing unfiltered room air (and potential contaminants) from entering. This creates a protective environment for your samples [5].
How often should I replace the HEPA filter in my PCR workstation? The lifetime of a HEPA filter varies based on the laboratory environment and usage. Manufacturers generally suggest replacement between 1 to 3 years. A common technical indicator for replacement is when the pressure drop across the filter is twice the amount measured at installation [86]. Properly stored, unused HEPA filters have an estimated shelf life of up to 10 years [62].
Is the UV light in my workstation dangerous? Yes, UV light can be dangerous as it can damage cells. The UV-C light used for decontamination is particularly hazardous. Always ensure the workstation's polycarbonate shield, which naturally reflects UV-C light, is in place during the decontamination cycle to protect the operator. Never look directly at the UV light source [86].
Experimental Protocols for Validation
Protocol: Environmental Surface Sampling for Viral RNA Contamination
This protocol is adapted from a study monitoring respiratory viruses on surfaces in a university setting and can be adapted for general contamination monitoring [89].
1. Objective To detect the presence of contaminating nucleic acids on high-touch surfaces within a PCR workstation before and after UV decontamination to validate the decontamination efficacy.
2. Materials and Reagents
- Synthetic tip swabs with plastic shafts (DNAse/RNAse-free)
- Guanidine-based viral transport medium (VTM) for nucleic acid inactivation and stabilization
- Nucleic Acid Extraction Kit
- PCR reagents for a multiplex assay targeting common contaminants
- Positive control (synthetic RNA) and negative control (nuclease-free water)
3. Methodology
- Swabbing Technique: Swab a standardized surface area of 100 cm². Swab the entire surface thoroughly, moving horizontally and vertically while rotating the swab throughout the process [89].
- Sample Collection: Place the swab immediately into a tube containing 500 µL of VTM.
- Storage: Keep samples at -20°C until the nucleic acid extraction step is performed.
- RNA Extraction: Extract nucleic acids from 250 µL of the VTM sample using the designated kit, following the manufacturer's instructions.
- Quality Control: Include a synthetic RNA control added directly to the sample to monitor extraction efficiency and detect PCR inhibitors [89].
- Molecular Detection: Analyze the extracted RNA using a sensitive real-time PCR platform capable of detecting low copy numbers of target sequences.
Protocol: Validating UV Decontamination Efficacy
1. Objective To quantitatively assess the effectiveness of the workstation's UV decontamination cycle.
2. Materials
- Bacterial spores (e.g., Bacillus atrophaeus) or other UV-resistant biological indicators
- Sterile phosphate-buffered saline (PBS)
- Tryptic soy agar (TSA) plates
- Sterile swabs
3. Methodology
- Inoculation: Apply a known concentration (e.g., 10^6 CFU) of biological indicators at several predetermined locations within the workstation, including spots that might be shadowed.
- UV Exposure: Run the UV decontamination cycle for the time period being validated.
- Recovery: After the cycle, use a swab moistened with sterile PBS to recover the biological indicators from the inoculation sites.
- Culture: Streak the swabs onto TSA plates and incubate them at 35±2°C for 48 hours.
- Analysis: Compare the growth on plates from the post-UV samples with controls (non-UV exposed indicators). A successful validation shows no growth post-decontamination.
Data Presentation
Table 1: Environmental Monitoring Results from a Non-Healthcare Setting
Data from a study detecting respiratory virus RNA on surfaces, demonstrating application and outcomes of environmental monitoring [89].
| Sampling Parameter | T1 (Nov-Dec 2023) | T2 (Jan 2024) | T3 (Feb 2024) |
|---|---|---|---|
| Total Swabs Collected | 125 | 145 | 130 |
| Positive Swabs | 25 | 27 | 10 |
| Positive Rate | 20.0% | 18.6% | 7.7% |
| Viral Targets | SARS-CoV-2, Flu A/B, RSV A/B | SARS-CoV-2, Flu A/B, RSV A/B | SARS-CoV-2, Flu A/B, RSV A/B |
| Sampling Parameter | Small Classrooms | Medium Classrooms | Large Classrooms |
|---|---|---|---|
| Total Swabs Collected | 135 | 105 | 115 |
| Positive Swabs | 30 | 15 | 13 |
| Positive Rate | 22.2% | 14.3% | 11.3% |
Table 2: PCR Workstation UV Decontamination Specifications
Summary of key technical specifications for UV decontamination in PCR workstations based on manufacturer data [62] [86].
| Specification | Typical Value / Range | Notes / Variants |
|---|---|---|
| UV Bulb Lifetime | Up to 1000 disinfection cycles [86] | Designed to deliver adequate energy for surface disinfection. |
| UV Timer Range | 0 - 18 hours (timed) [62] | Configurable in 1-minute increments. |
| UV Radiation Intensity | At least 40 µW/cm² [62] | Measured on the work surface. |
| Primary Decontamination Function | Surface disinfection of the chamber interior | For consumables and work surface. |
| Key Safety Feature | Polycarbonate shield to protect operator [86] | Shields naturally reflect UV-C light. |
Workflow Visualization
Environmental Monitoring for UV Validation
PCR Workstation Contamination Control
The Scientist's Toolkit
Table 3: Research Reagent Solutions for Environmental Monitoring
| Item | Function / Purpose |
|---|---|
| Synthetic Tip Swabs | To collect samples from surfaces without introducing contaminants from the swab itself [89]. |
| Guanidine-Based VTM | To inactivate viruses and stabilize nucleic acids immediately upon sample collection, preventing degradation during transport and storage [89]. |
| Nucleic Acid Extraction Kit | To purify and concentrate nucleic acids from the environmental sample, removing potential PCR inhibitors [89]. |
| Synthetic RNA Control | A known quantity of non-infectious RNA added to the sample to monitor the efficiency of RNA extraction and alert to the presence of PCR inhibitors [89]. |
| HEPA Filter | A high-efficiency particulate air filter that removes over 99.99% of particles at 0.3 microns from the air entering the workstation, maintaining a clean ISO Class 5 environment [62] [5]. |
| Germicidal UV Lamp | A bulb emitting UV-C light at 254 nm, which damages the DNA/RNA of microorganisms, providing surface decontamination within the workstation [5]. |
Evidence and Efficacy: Validating UV Protocols and Comparing Decontamination Methods
FAQs: UV Decontamination for PCR Workstations
Q1: What is the most effective wavelength of UV light for nucleic acid decontamination?
The most effective wavelength for inactivating microorganisms by damaging their DNA and RNA peaks at 263–270 nm [39]. This range correlates strongly with the production of cyclobutane pyrimidine dimers (CPDs), a major type of DNA lesion that inhibits replication [39]. Deconvolution analysis has confirmed that maximum bacterial inhibition occurs around 267.6 nm [39]. UV-C lamps, particularly the 253.7 nm lamps commonly used in PCR workstations, are very close to this ideal peak and are highly effective for decontamination [70].
Q2: How does a UV PCR workstation ensure my samples are protected from contamination?
PCR workstations use a combination of strategies to create a contamination-free environment [5]:
- Surface Decontamination: A UV germicidal lamp (typically 254 nm) irradiates the chamber's interior surfaces before and after use, inactivating DNA, RNA, viruses, and bacteria [5] [70].
- Air Decontamination: Many advanced workstations feature a HEPA filter and/or a UV air recirculator [5] [70]. The HEPA filter removes particulate contaminants from incoming air, while the UV recirculator continuously decontaminates the internal air, processing the entire cabinet volume up to 100 times per hour [70]. This system ensures clean working conditions without exposing the user to UV light.
Q3: What are the common problems that can reduce a UV lamp's decontamination efficiency?
UV lamp output can diminish due to several common issues [76]:
- Devitrification: The quartz glass of the lamp develops white, cloudy spots, reducing its transparency to UV light. This is often caused by surface contamination or the lamp running too hot [76].
- Blackening of Ends: Tungsten from the electrodes slowly erodes and deposits on the inside of the quartz body, blocking UV light. Frequent power cycling (switching on/off) accelerates this process [76].
- External Contamination: Fingerprints, dust, or other materials on the outside of the lamp can absorb UV radiation and cause local devitrification [76].
- Aging: The electrodes naturally deteriorate over time and eventually cannot spark the plasma arc effectively [76].
Q4: What maintenance is required to keep a UV decontamination system reliable?
Regular maintenance is crucial for consistent UV output [90]:
- Regular Cleaning: Clean the UV lamp and system reflector frequently using isopropanol wipes or glass cleaner. Always wear gloves to prevent fingerprints [76] [90].
- Performance Monitoring: Measure UV intensity regularly with a calibrated UV radiometer and log the data to track performance decline [90].
- Scheduled Replacement: Replace UV lamps proactively. While some lamps have a long rated life (e.g., ~9,000 hours), best practices in sensitive industries like pharmaceuticals dictate replacement every 1,000-1,500 hours to ensure peak intensity [70] [76].
- System Checks: Inspect and clean reflectors, as up to 60% of UV radiation reaching the target is reflected light [76].
Troubleshooting Guide: UV Decontamination Systems
| Problem | Potential Cause | Solution |
|---|---|---|
| Poor Decontamination Results | Cloudy/white spots on lamp (devitrification) [76]. | Clean lamp with isopropanol; if clouding remains, replace lamp [76]. |
| Lamp is old; electrodes eroded [76]. | Replace lamp per manufacturer schedule or if output is low [76]. | |
| Dust/dirt on lamp surface or reflector [76]. | Power down system; clean lamp and reflector with isopropanol [76] [90]. | |
| UV Lamp Fails to Ignite | Air leak in the lamp envelope [76]. | Use a high-frequency tester to confirm leak; replace lamp [76]. |
| Faulty power supply or electrical connections [76]. | Check for damaged end caps/seals; consult a technician [76] [90]. | |
| UV Lamp "Trips Out" | Lamp too close to metal housing, causing arc to jump [76]. | Ensure lamp is correctly positioned within the housing [76]. |
| High humidity causing electrical leakage [76]. | Control lab humidity and ensure proper system grounding [76]. | |
| Visible Deformation of Lamp | Lamp has overheated due to poor cooling/airflow [76]. | Check system cooling fans and airflow; replace deformed lamp [76]. |
Quantitative Data on UV Inactivation
UV Sensitivity Across Bacterial Types
The following table summarizes the fluence (UV dose) required for 1-3 log₁₀ inactivation of various bacteria, demonstrating that effectiveness is wavelength and organism-dependent [39].
| Organism Type | Strain Examples | Most Effective Wavelength (nm) | Key Finding (Fluence for 1-3 log₁₀ Inactivation) |
|---|---|---|---|
| Gram-Negative Bacteria | Escherichia coli, Pseudomonas aeruginosa [39]. | 263-270 nm [39]. | Higher initial UV sensitivity compared to Gram-positive bacteria [39]. |
| Gram-Positive Bacteria | Staphylococcus aureus, Enterococcus faecalis [39]. | 263-270 nm [39]. | Generally more resistant to UV irradiation than Gram-negative bacteria [39]. |
| Bacterial Spores | Bacillus subtilis (spores) [39]. | 263-270 nm [39]. | Significantly more difficult to inactivate than their trophozoite (vegetative) forms [39]. |
UV Efficacy Against SARS-CoV-2 on Surfaces
A 2025 study demonstrated that UV-C irradiation is effective for decontaminating filtering facepiece respirators by degrading SARS-CoV-2 RNA [48].
| UV-C Dose (mJ/cm²) | Impact on Filtration Efficiency | SARS-CoV-2 RNA Gene Degradation (ΔCt in RT-qPCR) |
|---|---|---|
| 60 - 300 | No significant change; FE remained >98% for all particle sizes (0.3-5 μm) [48]. | Significant degradation for E, RdRp2, RdRp4 genes (p ≤ 0.001) [48]. |
| 600 - 900 | Not tested in this range for FE. | Marked RNA degradation continued for E, RdRp2, RdRp4 genes [48]. |
| All Tested Doses | FE was preserved, supporting UV-C for mask decontamination [48]. | N gene stability was not significantly affected (p = 0.612) [48]. |
Experimental Protocols
Protocol: Quantifying Bacterial Inactivation by UV-LED
This protocol is adapted from a 2025 study that standardized irradiation to compare 13 different UV wavelengths [39].
Key Reagent Solutions:
- Bacterial Strains: Use frozen stocks of relevant strains (e.g., E. coli ATCC 25922, S. aureus ATCC 29213) [39].
- Culture Media: Use appropriate broths and agars (e.g., LB broth, Trypticase soy broth, Muller-Hinton broth) for the strains being tested [39].
- Irradiation Suspension Medium: Phosphate Buffered Saline (D-PBS) [39].
- UV-Irradiation System: A standardized UV-LED irradiation system that accounts for LED characteristics (peak wavelength, temperature-dependent shifts) and power supply factors [39].
Methodology:
- Culture and Harvest: Preculture bacteria from frozen stocks and grow in appropriate medium with shaking until the stationary phase is reached [39].
- Wash and Resuspend: Centrifuge the culture, discard the supernatant, and wash the bacterial pellet three times with D-PBS. Finally, resuspend in D-PBS for irradiation [39].
- UV Irradiation: Place the bacterial suspension under the UV-LED irradiation system. Expose to a specific, measured fluence (dose in mJ/cm²) for each wavelength being tested [39].
- Determine Viability: After irradiation, perform serial dilutions and a colony forming unit (CFU) assay on appropriate agar plates. Incubate plates and count colonies [39].
- Analyze Data: Generate dose-response curves by plotting log₁₀ inactivation (CFU/mL) against UV fluence for each wavelength and bacterial strain [39].
Protocol: Assessing UV Decontamination of SARS-CoV-2 RNA
This protocol is based on a 2025 study that evaluated UV-C's impact on virus detection [48].
Key Reagent Solutions:
- Viral RNA: SARS-CoV-2 positive nasopharyngeal swab samples [48].
- UV Chamber: A UV-C disinfection chamber with known, calibrated output (e.g., Flash Box with an average output of 60 mJ/cm² per minute) [48].
- Detection Method: RT-qPCR reagents and systems for detecting SARS-CoV-2 genes (E, RdRp2, RdRp4, N) [48].
Methodology:
- Sample Preparation: Aliquot SARS-CoV-2 RNA samples into a suitable vessel (e.g., a shallow, open plate) [48].
- UV Exposure: Place samples inside the UV-C chamber and expose to a range of doses (e.g., 0, 60, 300, 600, 900 mJ/cm²). Calculate exposure time based on the chamber's irradiance [48].
- RNA Quantification: Post-irradiation, use RT-qPCR to analyze the samples. The cycle threshold (Ct) value will indicate the remaining intact RNA [48].
- Analyze Data: Calculate the change in Ct (ΔCt) for each gene between exposed and control samples. A significant increase in Ct indicates successful RNA degradation [48].
Mechanism and Workflow Visualizations
Diagram 1: UV Inactivation Mechanism and Experimental Workflow.
Diagram 2: Troubleshooting Poor UV Decontamination Results.
Within molecular biology laboratories, particularly those utilizing Polymerase Chain Reaction (PCR) techniques, effective decontamination is a cornerstone of operational integrity. The prevention of contaminating DNA and RNA molecules is critical to ensure the accuracy and reliability of sensitive experiments. This technical support center resource is framed within a broader thesis on Ultraviolet (UV) irradiation for PCR workstation decontamination, providing a comparative analysis with traditional chemical methods. It is designed to assist researchers, scientists, and drug development professionals in selecting and troubleshooting decontamination protocols based on empirical evidence and practical application. The following sections provide a detailed examination of both strategies, summarized data for direct comparison, experimental protocols, and answers to frequently encountered problems.
Core Concepts and Definitions
Understanding the fundamental purpose of different laboratory equipment is essential for selecting the appropriate decontamination strategy.
- PCR Workstations are designed to protect sensitive samples from airborne contamination and cross-contamination during procedures like DNA amplification. They are not designed to protect the user from biohazards [35]. They often incorporate UV lamps (typically at 254 nm) and sometimes HEPA filtration to maintain a sterile interior environment [35] [5].
- Biosafety Cabinets (BSCs) are engineered to provide protection for the user, the environment, and the work product from biohazardous materials. They use HEPA filtration for both incoming and outgoing air to achieve this containment [35]. Using a PCR workstation for handling infectious agents is a serious safety breach.
The primary decontamination methods used within PCR workstations are:
- UV Germicidal Irradiation (UVGI): Uses short-wavelength UV-C light (200-280 nm) to inactivate microorganisms by damaging their nucleic acids, preventing replication [24].
- Chemical Decontamination: Involves the application of liquid chemical agents to surfaces to destroy or remove contaminants.
Summarized Data & Comparative Analysis
Table 1: Comparative Efficiency of Decontamination Strategies for DNA Removal
This table summarizes experimental data comparing the efficiency of various methods in removing contaminating DNA molecules from different surfaces [91].
| Cleaning Strategy | Surface Type | % of DNA Recovered (vs. Control) | Key Findings & Notes |
|---|---|---|---|
| Ethanol (70%) | Plastic | ~52% | Minimally effective; used alone, it is quite inefficient. |
| Metal | ~32% | ||
| Wood | ~27% | ||
| UV Radiation (254 nm, 20 min) | Plastic | ~52% | Ineffective as a standalone method for DNA decontamination. |
| Metal | ~32% | ||
| Wood | ~27% | ||
| Sodium Hypochlorite (0.4-0.54%) | All Surfaces | ≤ 0.3% | Most efficient method for cell-free DNA. |
| Trigene (10%) | All Surfaces | ≤ 0.3% | Most efficient method for cell-free DNA. |
| Virkon (1%) | All Surfaces | ≤ 0.8% | Most efficient method for cell-contained DNA (blood). |
Table 2: Practical Considerations for UV and Chemical Decontamination
This table compares key characteristics and practical aspects of the two decontamination approaches.
| Characteristic | UV Decontamination | Chemical Decontamination |
|---|---|---|
| Primary Mechanism | Nucleic acid damage (oxidation, strand breaks) [91] [24] | Variable: protein denaturation, cell membrane disruption, oxidation. |
| Effectiveness on Surfaces | Variable; less effective on porous materials (e.g., cardboard) and shadowed areas [92]. | Can be highly effective on all accessible surfaces, depending on the agent. |
| Speed | Rapid-cycle systems can decontaminate devices in minutes [93]. | Requires application and contact time, followed by wiping; can be slower. |
| Automation Potential | High; can be programmed for automatic cycles in PCR workstations. | Low; requires manual application. |
| Material Compatibility | Can degrade certain plastics and materials with prolonged exposure. | Some chemicals can be corrosive or damage surfaces (e.g., bleach). |
| Residual Effect | None; inactivates only during direct exposure. | No residual effect for most agents used in labs (unless specifically designed). |
| User Safety | Risk of UV exposure to skin and eyes; requires safety interlocks [5]. | Risk of inhalation or skin contact with hazardous chemicals; requires PPE. |
| Impact on Workflow | Can increase compliance and frequency of cleaning; reduces cleaning time [93]. | Manual process may lead to less frequent cleaning. |
Table 3: Key Research Reagent Solutions
This table details essential reagents and materials used in decontamination research and practice.
| Reagent/Material | Function/Description | Example Use in Context |
|---|---|---|
| Sodium Hypochlorite | An oxidizing agent that is highly effective at destroying contaminating DNA and inactivating pathogens [91]. | Diluted to 0.4-0.54% for surface decontamination; a gold standard for DNA removal. |
| Trigene | A commercial disinfectant cleaner effective against a broad spectrum of microbes and nucleic acids [91]. | Used at a 10% concentration for efficient removal of cell-free DNA from surfaces. |
| Virkon | A broad-spectrum disinfectant powder that forms a peroxygen-based solution. | Used at 1% for decontaminating surfaces contaminated with blood (cell-contained DNA) [91]. |
| Ethanol (70%) | A common laboratory disinfectant that acts by denaturing proteins. | Less effective for DNA removal alone but is often used in combination with other methods [91]. |
| Φ6 Bacteriophage | An enveloped RNA virus used as a BSL-1 surrogate for SARS-CoV-2 and other enveloped viruses in decontamination studies [92]. | Used in field and lab testing to evaluate the efficacy of UV devices against an environmentally relevant virus. |
Experimental Protocols & Methodologies
Protocol: Evaluating Decontamination Efficiency on Surfaces
This protocol is adapted from a study evaluating DNA removal [91].
Objective: To quantify the efficiency of different decontamination strategies in removing contaminating DNA from various laboratory surfaces.
Materials:
- Cell-free DNA solution or whole blood.
- Test surfaces (e.g., plastic, stainless steel, painted wood).
- Calibrated spray bottles.
- Decontamination agents (e.g., Sodium hypochlorite, Trigene, 70% Ethanol).
- UV lamp (254 nm).
- Cotton swabs, DNA extraction kit, Real-time PCR equipment.
Methodology:
- Surface Contamination:
- Deposit a known quantity of cell-free DNA (e.g., 60 ng) or whole blood (e.g., 10 µL) onto marked, replicate areas of the test surfaces.
- Allow the samples to air-dry completely (e.g., for 2 hours).
Application of Decontamination:
- For liquid chemical agents, administer a standardized volume (e.g., one spray from a calibrated bottle) onto the contaminated area.
- Wipe the area uniformly with a dust-free paper in three circular motions.
- Allow the surface to dry (typically 120 minutes).
- For UV treatment, expose the dried contaminant to a specified UV dose (e.g., 254 nm for 20 minutes at a set distance). Ensure the surface is directly visible to the UV source.
Sample Collection and Analysis:
- After decontamination, swab the entire treated area with a moistened cotton swab.
- Extract DNA from the swab using a commercial kit.
- Quantify the amount of recovered DNA using a highly sensitive real-time PCR assay (e.g., targeting mitochondrial DNA for increased sensitivity).
- Compare the quantity of DNA recovered from decontaminated surfaces to the quantity recovered from non-decontaminated control surfaces to calculate the percentage of DNA removal.
Protocol: Testing UV Device Efficacy Against Viruses
This protocol is based on methods used to test UV devices against a SARS-CoV-2 surrogate [92].
Objective: To measure the antiviral efficacy of a UV decontamination device against a dried, enveloped virus on different materials.
Materials:
- High-titer preparation of an enveloped RNA virus surrogate (e.g., Φ6 bacteriophage).
- Test coupons of relevant materials (e.g., stainless steel 304, painted surfaces, cardboard, plastics).
- UV device(s) for testing.
- Plaque assay materials for virus quantification.
Methodology:
- Virus Inoculation:
- Apply a high titer (>8.0 log10 PFU) of the virus suspension onto small, defined test coupons.
- Allow the virus to dry onto the material, creating environmentally relevant conditions that include host cell debris.
UV Exposure:
- Expose the inoculated coupons to the UV device at a specified distance for a set time.
- Measure the UV dosage delivered (in mJ cm⁻²) using a calibrated dosimeter.
Virus Recovery and Titration:
- After UV exposure, elute the virus from the coupon surface using an appropriate elution buffer.
- Determine the number of remaining infectious virus particles using a plaque assay.
- Calculate the log10 reduction in viral titer compared to non-UV-exposed control coupons.
Decision Workflow for Decontamination Strategy
The following diagram outlines a logical decision process for selecting between UV and chemical decontamination within a PCR workstation context, based on the specific contamination scenario.
Troubleshooting Guides & FAQs
FAQ 1: General Decontamination Strategy
Q: Which is better for my PCR workstation: UV decontamination or chemical cleaning?
A: Neither is universally "better"; they serve complementary roles. For routine, automated decontamination of the workstation interior between uses to prevent airborne contamination, UV is highly effective and convenient [5]. However, for reliably eliminating nucleic acid contamination from spills or on surfaces, chemical decontamination with agents like sodium hypochlorite (bleach) or Trigene has been proven to be significantly more effective [91]. A best-practice approach is to use both: clean spills with a chemical agent first, followed by a standard UV cycle.
Q: Why did I get a false positive PCR result even though I used the UV lamp in my workstation?
A: UV decontamination is not foolproof. Several factors can lead to its failure:
- Shadow Effects: UV light travels in a straight line. Any item or surface irregularity that creates a shadow will not be decontaminated.
- Surface Porosity: On porous materials like cardboard, UV efficacy drops dramatically because contaminants are shielded within the fibers [92].
- Insufficient Dosage: The required UV dose to degrade nucleic acids effectively might not have been delivered due to lamp age, distance from the surface, or insufficient time.
- Organic Load: The presence of proteins or other organic debris can protect contaminants from UV inactivation [92]. Always pre-clean surfaces with a chemical agent to remove gross contamination before using UV.
Q: Can I use UV to decontaminate my biosafety cabinet after working with infectious agents?
A: UV can be a supplementary method in a BSC, but it must not be relied upon as the primary decontamination method. The primary containment in a BSC is provided by its directional airflow and HEPA filters, not UV [35]. All surfaces must be decontaminated with an appropriate chemical disinfectant (e.g., bleach, diluted for the specific pathogen) before the cabinet is wiped down at the end of a procedure. UV may be used as an additional safety measure after chemical cleaning when the cabinet is unoccupied.
FAQ 2: UV-Specific Issues
Q: How long should I run the UV lamp in my workstation to ensure it's decontaminated?
A: There is no universal time, as it depends on the lamp's intensity, distance from surfaces, and the organism or contaminant targeted. Research shows UV device dosages and efficacy are widely variable [92]. Consult your manufacturer's manual for a recommended cycle time (often 15-30 minutes). For critical work, validate the cycle by placing biological indicators (e.g., bacterial spores on a strip) in the most challenging-to-reach spots and verifying inactivation. Remember, UV is most effective on directly exposed, non-porous surfaces.
Q: The UV lamp in my unit is still glowing blue/purple. Does that mean it's working effectively?
A: Not necessarily. The visible glow indicates the lamp is on, but the germicidal UV-C output at 254 nm can degrade over time without a visible change in the light. The intensity of UV-C emission decreases with lamp age. Manufacturers typically recommend replacing UV lamps after 8,000-9,000 hours of use, but the output should be periodically checked with a UV-C radiometer to ensure it delivers a sufficient germicidal dose.
Q: Is it safe to be in the room while the UV lamp is on?
A: No. Direct exposure to UV-C light is a health hazard and can cause damage to the skin (erythema, similar to sunburn) and eyes (photokeratitis) [5]. All modern PCR workstations are equipped with safety interlocks that automatically turn off the UV lamps when the door or sash is opened. Never override these safety features. If you need to decontaminate a room with a mobile UV unit, ensure the room is completely unoccupied and secured during operation.
FAQ 3: Chemical-Specific Issues
Q: Which chemical is the most effective for destroying contaminating DNA in my lab?
A: Based on comparative studies, sodium hypochlorite (common household bleach, diluted to 0.4-0.54%) and Trigene (at 10%) are the most effective agents, recovering a maximum of only 0.3% of contaminating DNA after treatment [91]. For cell-contained DNA, such as in blood, 1% Virkon was also highly effective.
Q: I use 70% ethanol to wipe my bench. Why is it not sufficient for DNA removal?
A: While 70% ethanol is an excellent general disinfectant for many bacteria and viruses, it is relatively inefficient at destroying contaminating DNA molecules. Studies show that after ethanol cleaning, over 30-50% of the original DNA can still be recovered from the surface, which is more than enough to cause PCR contamination [91]. Ethanol fixes rather than destroys DNA on surfaces.
Q: How should I properly apply a chemical decontaminant?
A: For maximum effectiveness:
- Pre-clean: Remove visible debris with a damp cloth.
- Apply: Spray or wipe enough of the chemical to thoroughly wet the surface.
- Contact Time: Allow the surface to remain wet for the manufacturer's recommended contact time. Do not wipe it dry immediately.
- Rinse (if required): Some agents may require a rinse with sterile water to prevent residue, especially if they are corrosive.
- Dry: Allow the surface to air dry.
In laboratories where contamination control is paramount, selecting the correct equipment is a fundamental decision. While Polymerase Chain Reaction (PCR) workstations and Biosafety Cabinets (BSCs) may appear similar, they are engineered for distinct purposes and offer different levels of protection. PCR workstations are primarily designed to protect sensitive samples from contamination during molecular biology procedures. In contrast, Biosafety Cabinets are engineered to provide comprehensive protection for the user, the environment, and the work product (the sample) from biohazardous materials [35] [94].
Understanding this distinction is critical. Using a PCR workstation for handling pathogens risks exposing the technician and the laboratory to infectious agents. Conversely, using a biosafety cabinet for routine, non-hazardous PCR setup can be inefficient and may not provide the optimal environment to prevent cross-contamination between samples [35]. This guide, framed within ongoing research on UV irradiation for decontamination, will help you select the right tool and troubleshoot common issues.
Equipment Comparison: Purpose, Protection, and Principle
The following table summarizes the core differences between a PCR Workstation and a Biosafety Cabinet.
Table 1: Fundamental Differences Between PCR Workstations and Biosafety Cabinets
| Feature | PCR Workstation | Biosafety Cabinet (Class II, Type A2) |
|---|---|---|
| Primary Purpose | Protect the sample from contamination (nucleic acids, particulates) [35] | Protect the user, environment, and sample from biohazards [35] |
| Protection Level | Sample only [35] | User, sample, and environment [35] |
| Ideal Applications | DNA amplification, PCR setup, other non-hazardous molecular biology tasks [35] | Microbiological work with pathogens (BSL-1-3), cell culture, handling hazardous materials [35] |
| Airflow Principle | Vertical laminar flow to maintain a sterile work surface [35] [15] | Combination of inflow (to protect user) and downflow (HEPA-filtered to protect sample) air [35] |
| Filtration | HEPA filters to remove particulates from air flowing over the work surface [35] [28] | HEPA filters for both incoming and outgoing air to ensure containment [35] |
| UV Sterilization | Common feature for decontaminating the work surface between uses [35] [28] | May be included, but primary protection is from airflow and filtration [35] |
| Key Takeaway | For sensitive samples without hazardous materials. | For hazardous materials, ensuring personnel and environmental safety. |
The Role of UV Irradiation in Decontamination
UV-C irradiation (254 nm) is a critical decontamination feature in modern PCR workstations, directly supporting the thesis of its importance in contamination control research. It serves to degrade contaminating nucleic acids and inactivate microorganisms on the workstation's surfaces between experiments [15] [28]. Effective decontamination typically requires a 15-30 minute exposure and can reduce microbial contamination by up to 99.9% [15]. Advanced models, like some UVP workstations, employ multiple layers of UV irradiation—in the work area, pre-filter chamber, and air recirculator—to ensure maximum decontamination [28].
Troubleshooting Guides and FAQs
PCR Workstation FAQs
Q1: My PCR reactions consistently show smeared bands or false positives. How can I use the workstation to resolve this?
This is a classic sign of contamination, either from airborne particulates or contaminated surfaces/equipment within the workstation.
- Solution:
- UV Decontamination Protocol: Before starting work, ensure the interior surfaces are decontaminated. Wipe down all surfaces with a DNA-degrading solution (e.g., 10% bleach, commercial DNA-Zap), followed by 70% ethanol to remove residual cleaning agents. Then, run a full UV decontamination cycle for 30 minutes with the door closed and no materials inside [15].
- Optimize Workflow: Implement a unidirectional workflow. Place clean reagents and consumables on one side of the cabinet, perform the PCR setup in the center, and place used tips and waste on the opposite side. This prevents cross-contamination [15].
- Check HEPA Filter: If your workstation has a HEPA filter, ensure it is functioning correctly. A clogged or failed filter will not effectively remove airborne contaminants. Replace filters according to the manufacturer's schedule [28].
Q2: The UV lamp in my workstation does not turn on. What should I check?
- Solution:
- Safety Interlock: Most workstations have a safety shut-off switch that automatically turns the UV light off when the door is opened. Ensure the door is fully closed [28].
- Timer Setting: Verify that the UV timer is set correctly for the desired duration.
- Lamp Life: UV lamps have a finite lifespan. Check the manufacturer's specifications and replace the lamp if it has exceeded its service hours [95].
- Power: Ensure the workstation is properly plugged in and receiving power.
Biosafety Cabinet FAQs
Q1: The BSC alarm is indicating low airflow. What are the immediate steps I should take?
- Solution:
- Stop Work Immediately: A low airflow alarm signifies that the cabinet's containment is compromised. Cease all work with hazardous materials immediately [96].
- Check for Obstructions: Visually inspect the front and rear air grilles for any obstructions (e.g., papers, equipment) and clear them [95] [96].
- Verify Sash Position: Ensure the sash is at the correct, marked operating height.
- Do Not Ignore: If the alarm persists after these basic checks, turn off the BSC, post a "Do Not Use" sign, and contact a qualified technician for service. The HEPA filter may be clogged or the motor may be failing [95] [96].
Q2: How do I properly decontaminate my BSC, and what is the role of UV light in this process?
- Solution:
- Primary Method: Chemical Disinfection: The primary method for BSC decontamination is thorough cleaning with appropriate disinfectants (e.g., 70% ethanol, diluted bleach). Wipe down all interior surfaces, including the work surface, walls, and window, before and after each use [95] [96].
- UV as an Adjunct: UV light in a BSC should only be used as a secondary decontamination measure. It is effective for reducing bioburden on exposed surfaces after chemical cleaning, particularly in hard-to-reach areas. It is not a substitute for manual cleaning, as dust and shadows can shield microorganisms [95] [15].
- Safety First: Never operate the UV light while the cabinet is occupied. Always ensure the UV light is off and the cabinet has been ventilated before entering.
Table 2: Troubleshooting Common Equipment Problems
| Problem | Possible Cause | Solution |
|---|---|---|
| PCR: No/Low Amplification | Contaminated reagents or workspace [23]. | Decontaminate workstation with UV and chemicals; use fresh, aliquoted reagents; optimize thermal cycling conditions [25] [23]. |
| PCR: Non-Specific Bands | Contaminants or suboptimal primer design [25]. | Use hot-start DNA polymerase; optimize Mg2+ concentration and annealing temperature; redesign primers to avoid complementarity [25] [23]. |
| BSC: Airflow Imbalance/Vibration | Clogged pre-filter, worn motor bearings, or unstable placement [95] [96]. | Replace pre-filter; contact technician for motor repair; ensure BSC is on a stable, level surface [95]. |
| BSC: UV Light Not Effective | Expired UV lamp, dirty surfaces, or shadowed areas [95]. | Replace lamp per manufacturer's schedule; always chemically clean before UV use; be aware of UV's line-of-sight limitation [95] [15]. |
Experimental Protocol: UV Irradiation Efficacy for Surface Decontamination
This protocol outlines a methodology to validate the efficacy of UV irradiation in decontaminating a PCR workstation surface, a key experiment within the broader research context.
Objective: To determine the minimum UV exposure time required to achieve effective decontamination of a PCR workstation surface inoculated with a known contaminant (E. coli containing a plasmid with a common amplicon sequence, e.g., GFP).
Materials:
- Sterile swabs
- LB agar plates
- Liquid LB media
- E. coli (with and without GFP plasmid)
- UV PCR Workstation (e.g., UVP model with 254 nm lamps) [28]
- Vortex mixer
- Incubator (37°C)
Methodology:
- Preparation: Turn on the PCR workstation's white light and wipe down the entire work surface with 70% ethanol. Allow to air dry.
- Inoculation: Create a suspension of E. coli (with GFP plasmid) in LB media. Apply a standardized volume (e.g., 100 µL) in discrete, marked spots on the workstation's surface. Allow the spots to dry completely under the laminar flow.
- UV Exposure: Close the sash and set the UV timer. Expose the entire work area to UV light for varying time intervals (e.g., 0 min control, 5 min, 10 min, 15 min, 30 min).
- Sampling: After each exposure time, use a pre-moistened sterile swab to resuspend the bacteria from a specific spot. Streak the swab onto an LB agar plate. Repeat for each time point using a new, dedicated spot.
- Incubation and Analysis: Incubate all plates at 37°C overnight. The following day, count the colony-forming units (CFUs) on each plate. The effective decontamination time is the minimum exposure that results in a 99.9% reduction in CFUs compared to the 0-minute control [15].
- PCR Validation (Optional): To specifically test for nucleic acid contamination, repeat the inoculation. After UV exposure, swab the surface and use the swab as a template in a PCR reaction targeting the GFP gene. Effective decontamination will be confirmed by the absence of a PCR amplicon on a gel.
Research Reagent Solutions
Table 3: Essential Reagents for UV Decontamination Research
| Reagent/Material | Function in the Experiment |
|---|---|
| HEPA Filter | Creates a particulate-free environment for pre- and post-UV exposure sampling, preventing airborne contamination [28]. |
| UV-C Lamps (254 nm) | The primary decontamination agent; generates germicidal UV light that damages microbial DNA and RNA [28]. |
| Microbial Culture (e.g., E. coli with plasmid) | Serves as a standardized biological indicator to quantitatively measure the efficacy of the UV decontamination process. |
| DNA-Decontaminating Solution (e.g., 10% bleach) | Provides a chemical decontamination baseline to compare against the efficacy of UV irradiation [15]. |
| Agar Plates & Growth Media | Allows for the cultivation and quantification of surviving microorganisms post-UV exposure (via CFU count). |
| Hot-Start DNA Polymerase | Used in post-decontamination PCR checks to prevent false-positive amplification from low-level contaminants, ensuring reaction specificity [25] [23]. |
Logical Workflow for Equipment Selection and Troubleshooting
The following diagram illustrates the decision-making process for selecting between a PCR workstation and a biosafety cabinet, and the primary troubleshooting paths for each.
Our research laboratory, focused on high-throughput drug development, recently faced a critical challenge: a lab-wide contamination incident was compromising sensitive PCR experiments, leading to inconsistent results and costly delays. This case study details how we systematically identified the sources of contamination and resolved the issue by implementing a comprehensive UV irradiation and maintenance protocol for our PCR workstations. Contamination in PCR workstations jeopardizes sensitive biological samples and can lead to inaccurate experimental results, making a robust decontamination protocol essential [4] [53].
Troubleshooting Guides
PCR Workstation Contamination: Diagnostic Flowchart
The following diagram outlines the logical, step-by-step process for diagnosing the root cause of contamination in a PCR workstation.
Frequently Asked Questions (FAQs)
Q1: Our lab is experiencing sporadic PCR contamination. We use our UV workstation daily. What is the most commonly overlooked maintenance task?
A: The most frequently overlooked task is the regular cleaning of dust filters [4] [53]. Clogged filters reduce air circulation and UV irradiation efficiency, increasing contamination risk. We implemented a mandatory weekly visual inspection and a monthly cleaning routine where filters are gently rinsed with warm tap water and air-dried [4].
Q2: How often should we replace the UV lamps in our PCR workstations? We wait until they burn out.
A: Waiting for complete failure is a common error. UV lamps have a functional lifespan of 9,000 hours, but their germicidal efficiency begins to decline after 5,000 hours, falling by approximately 15% for every additional 1,000 hours of operation [4] [53]. We resolved our issue by instituting a bi-annual replacement schedule, which is a proactive approach recommended for models without automatic usage tracking [4].
Q3: Are all PCR workstations the same, and could the type itself be a source of our problems?
A: No, a key factor is the presence of a HEPA filter. There are two primary types:
- UV PCR Workstations: Rely on UV light for decontamination.
- UV + HEPA PCR Workstations: Combine UV light with a HEPA filter that cleans incoming air, creating a positive pressure environment that actively excludes external contaminants [5]. For highly sensitive samples, a HEPA-filtered workstation is strongly recommended [5].
Q4: We cleaned our acrylic-surfaced PCR workstation with a 70% ethanol solution, and the surfaces are now cloudy. What went wrong?
A: Cloudiness indicates surface damage. Acrylic surfaces (e.g., Plexiglas) are permanently damaged by solvents like alcohol, acetone, and ammonia [4] [53]. For models like the UVC/T-AR, you must avoid organic solvent-based cleaners, pure alcohol, and high-concentration alcohol solutions (>20%). Always consult your user manual for compatible cleaning agents and finish by wiping with distilled water to remove residue [4].
Quantitative Data on UV Lamp Performance and Efficacy
A critical part of our new protocol was establishing data-driven maintenance schedules. The table below summarizes the key performance metrics for UV-C lamps, which are essential for planning replacements before decontamination efficacy drops.
Table 1: UV-C Lamp Performance and Replacement Guidelines
| Metric | Specification | Protocol Action |
|---|---|---|
| Functional Lifespan [4] [53] | 9,000 hours | Schedule replacement well before this point. |
| Efficiency Decline Start [4] [53] | After 5,000 hours | Monitor usage hours from this point. |
| Efficiency Decay Rate [4] [53] | ~15% reduction per 1,000 hours after 5,000 hours | For critical work, replace lamps before significant decay. |
| Recommended Replacement [4] | Every 6 months (or per automatic alarm) | We implemented a bi-annual replacement schedule for all cabinets. |
| Primary Wavelength [28] [5] | 254 nm | Ensure purchased replacement lamps emit at this germicidal wavelength. |
The Scientist's Toolkit: Research Reagent & Material Solutions
Implementing the correct materials and reagents is fundamental to the success of any decontamination protocol. The following table details the essential items for maintaining a contamination-free PCR workstation.
Table 2: Essential Materials for PCR Workstation Maintenance and Decontamination
| Item | Function / Purpose | Key Considerations & Notes |
|---|---|---|
| Lint-free Cloths [4] [53] | For wiping down interior surfaces without leaving particles. | Prevents scratching and avoids introducing new contaminants. |
| Manufacturer-Approved Cleaning Solutions [4] [53] | Surface decontamination before and after experiments. | Critical: Compatibility varies by model. Aggressive solutions can damage acrylic surfaces or special UV coatings on glass. |
| Distilled Water [4] [53] | Final rinse to remove cleaning solution residue. | Used after decontamination on acrylic surfaces to prevent clouding. |
| Replacement Dust Filters [4] | Maintains optimal airflow and protects UV lamps from dust. | Should be visually inspected weekly and cleaned monthly. Must be replaced if damaged. |
| Spare UV-C Lamps [4] [28] | Ensures consistent and effective germicidal irradiation. | Stocked based on the bi-annual replacement schedule. We keep a log of installation dates for each cabinet. |
| HEPA Filters (for equipped models) [28] [5] | Provides final filtration of incoming air, crucial for creating a positive pressure, contaminant-free environment. | Rated at 99.999% efficiency for particles ≥0.3 microns [5]. |
Experimental Protocol: Validating UV Decontamination Efficacy
To objectively validate our new UV protocol, we established a simple yet effective testing procedure.
Workflow for Protocol Validation
The experimental workflow for validating the decontamination efficacy of a PCR workstation is as follows.
Methodology
- Surface Contamination: Apply a standardized inoculum (e.g., E. coli or Staphylococcus aureus [97]) to a sterile Petri dish or a coupon of material matching the workstation's interior surface. Allow to dry.
- UV Decontamination Cycle: Place the contaminated sample inside the PCR workstation. Execute a standard UV decontamination cycle (e.g., 30 minutes at 254 nm [28] [5]).
- Surface Sampling & Microbial Culture: After UV exposure, use a neutralizer solution to swab the surface and transfer the sample to a liquid growth medium. Plate serial dilutions onto agar plates and incubate.
- Colony Count Analysis: Count the colony-forming units (CFUs) on the plates from the test sample and a positive control (contaminated but not UV-exposed).
- Calculate Log Reduction: Use the formula: Log Reduction = Log₁₀(Control CFU) - Log₁₀(Test CFU). A well-functioning UV system should achieve a significant reduction. Studies have shown that effective UVC illumination can achieve up to a 9 log₁₀ reduction on Petri dishes [97].
By adopting this comprehensive, data-driven UV protocol—encompassing rigorous troubleshooting, scheduled maintenance, staff training on proper cleaning techniques, and regular validation—we successfully eradicated the lab-wide contamination. This restored the integrity of our PCR experiments, significantly improved operational efficiency, and provided a sustainable framework for maintaining the highest standards of sample quality in our drug development research. A well-maintained PCR cabinet is fundamental to achieving consistent, reliable results and prolonging the life of valuable laboratory equipment [4] [53].
Technical Support Center: Troubleshooting UV PCR Workstations
This support center provides targeted guidance for researchers and scientists integrating UV decontamination into accredited laboratory workflows. The following troubleshooting guides and FAQs address common operational challenges to ensure compliance and experimental integrity.
Troubleshooting Common UV Workstation Issues
| Problem | Possible Cause | Troubleshooting Steps | Accreditation & Data Integrity Considerations |
|---|---|---|---|
| Reduced UV Intensity [58] | Aging UV lamps, quartz sleeve fouling, malfunctioning power supply [58]. | Monitor UV intensity levels; inspect lamps for aging; clean quartz sleeves; check power supply voltage [58]. | NATA accreditation requires demonstrated technical competence; failure to maintain intensity compromises test validity [98]. |
| Inadequate Decontamination | Insufficient UV exposure time, obstructed UV light, incorrect wavelength. | Validate exposure time for microbial kill rates; ensure work surface is unobstructed; verify lamp emits correct 254 nm UVC [59]. | Contamination leads to false positives/negatives, invalidating data. Accredited labs must have documented validation protocols [98] [99]. |
| Quartz Sleeve Fouling [58] | Accumulation of minerals, sediment, or biological growth on the sleeve [58]. | Implement a routine cleaning schedule with appropriate solutions (e.g., ethanol, specialized descaling agents) to remove deposits [58] [100]. | Fouling reduces UV transmission, creating a non-validated decontamination state. Cleaning logs are essential for audit trails. |
| Safety Interlock Failure | Worn switch, electrical fault, or control system error. | Test safety function regularly; do not operate if interlock fails; contact qualified technical service immediately [59]. | Non-functional safety features pose immediate risk to personnel and violate safety standards required for accreditation [101]. |
| Positive Control Failure | Contamination of master mix, reagents, or consumables within the workstation. | Decontaminate workstation surfaces and interior with UV; use fresh, certified nuclease-free reagents; employ single-use aliquots [102]. | QA procedures must define corrective actions for contamination events. This is critical for maintaining accredited status [98]. |
Frequently Asked Questions (FAQs)
Q1: What are the key characteristics to look for in a UV PCR workstation to ensure it meets accreditation standards? A workstation should feature effective UVC containment (e.g., automatic shut-off when opened, shielded design), a documented decontamination efficacy against common contaminants, and safety certifications such as those from UL based on relevant standards (e.g., UL 8802, IEC 62471) [101] [59]. The technical competence of the equipment is a fundamental requirement for labs seeking accreditation like NATA [98].
Q2: How often should I validate the decontamination efficacy of my UV PCR workstation? Regular validation is critical. The frequency should be determined by a risk assessment and the manufacturer's recommendations. This is part of the lab's quality assurance program, which accreditation bodies will audit [98]. Key performance indicators, such as UV lamp intensity, should be monitored regularly, as lamps degrade over time and require periodic replacement [58] [100].
Q3: My PCR experiments are showing sporadic contamination. How can I determine if my UV workstation is the source? Perform a negative control test. Run a PCR reaction with all reagents but no template DNA inside the UV workstation after a full decontamination cycle. If the negative control is positive, it indicates contamination. Next, use surface swabs and settle plates to sample the workstation's interior after decontamination to identify if contamination is airborne or on surfaces. This systematic investigation is part of a robust Quality Assurance framework [99].
Q4: Are there any materials that should not be placed inside a UV PCR workstation during decontamination? Yes. UVC radiation can degrade many materials. Avoid placing unprotected plastic consumables (e.g., pipette tips racks not designed for UV exposure), reagent tubes that are not UV-stable, and any sensitive optical components inside the chamber during the UV cycle. Always consult the manufacturer's guidelines for material compatibility [102].
Experimental Protocol: Validating UV Decontamination Efficacy
This protocol provides a detailed methodology for validating the efficacy of a UV PCR workstation's decontamination cycle, a critical requirement for laboratory accreditation.
Objective: To quantitatively assess the effectiveness of the workstation's UV cycle in eliminating nucleic acid contaminants from surfaces and the air.
Principle: The workstation is intentionally contaminated with a known amount of a non-pathogenic surrogate organism or purified DNA. After running the standard UV decontamination cycle, residual viable contaminants are cultured or detected via PCR to determine the log reduction.
Materials:
- UV PCR Workstation (e.g., model with 254 nm UV lamps and HEPA filtration) [59].
- Bacillus atrophaeus spores or purified Lambda DNA (common surrogates).
- Nutrient Agar plates (for bacterial culture) or PCR reagents for detection.
- Sterile swabs or settle plates.
- Incubator (for bacterial cultures) or Thermal Cycler (for PCR).
Procedure:
- Preparation: Turn off the workstation's white light and ensure the safety door is closed. Wipe down the entire interior work surface with 70% ethanol to remove pre-existing contaminants [102].
- Contamination (Pre-UV Cycle):
- Surface Contamination: Apply 10 µL of a
10^6 CFU/mLB. atrophaeus spore suspension or10^6 copies/µLLambda DNA solution to several pre-defined locations on the work surface. Allow to air dry completely. - Airborne Contamination (for HEPA models): Open a nutrient agar settle plate within the workstation for 30 minutes to capture airborne particles before the UV cycle.
- Surface Contamination: Apply 10 µL of a
- UV Decontamination: Initiate the standard UV decontamination cycle as per the manufacturer's instructions (e.g., 30-minute cycle) [59]. Ensure the room is vacated during this process.
- Sampling (Post-UV Cycle):
- Surface Sampling: After the cycle is complete and the UV lamps are off, use a sterile moistened swab to resample the exact locations contaminated in Step 2. Streak the swab onto a nutrient agar plate for culture or elute the nucleic acid for PCR analysis.
- Airborne Sampling: Place a fresh settle plate in the workstation for 30 minutes post-decontamination.
- Analysis:
- Culture Method: Incubate agar plates at
37°Cfor24-48 hours. Count the colony-forming units (CFUs) on pre- and post-UV plates. - PCR Method: Perform qPCR on the pre- and post-UV samples. Compare the cycle threshold (Ct) values.
- Culture Method: Incubate agar plates at
- Calculation:
- Calculate the log reduction using the formula: Log Reduction = Log10(Pre-UV CFU) - Log10(Post-UV CFU). A reduction of >4-log (99.99%) is typically considered effective for critical applications.
The workflow for this validation protocol is outlined below.
The Scientist's Toolkit: Essential Materials for UV Decontamination Research
| Item | Function in Research/QA | Relevance to Accreditation |
|---|---|---|
| NATA-Accredited Testing Lab Services [98] | Provides independent, accredited validation of UV disinfection efficacy against standards, lending credibility to in-house data. | Directly supports accreditation by providing externally verified, objective evidence of technical competence [98]. |
| HEPA-Filtered UV Workstation [59] | Provides a controlled environment combining UV surface decontamination with HEPA-filtered air circulation to prevent airborne contamination. | Essential for demonstrating a controlled, contamination-free work area as required by quality standards [99] [59]. |
| Bacillus atrophaeus Spores | A common biological indicator used to challenge and validate the efficacy of the UV decontamination cycle due to their high UV resistance. | Provides a quantitative, traceable measure of decontamination performance for audit and review purposes. |
| UV Intensity Meter | Measures the output of UV lamps at 254 nm to ensure they are operating within specified parameters for effective germicidal activity [58]. | Part of equipment calibration and monitoring, a core requirement of any accreditation system [98] [58]. |
| Nuclease-Free Water & Reagents | Certified to be free of nucleases and contaminants, serving as critical negative controls in PCR experiments to monitor for contamination. | Use of certified reagents is a fundamental best practice in an accredited lab's quality system to ensure result reliability [102]. |
UV PCR Workstation Market & Standards Data
For strategic planning and justification of equipment procurement, the following quantitative data is relevant.
Table: Global UV PCR Workstation Market Overview [99]
| Metric | Value / Forecast | Note |
|---|---|---|
| Projected Market Value (2025) | ~$350 Million | Reflects the growing adoption in labs. |
| Projected CAGR (2025-2033) | ~8% | Indicates sustained growth and technological evolution. |
| Dominant Application Segment | Scientific Research | Driven by R&D in life sciences and biotechnology. |
| Key Growth Driver | Demand for sensitive/accurate molecular diagnostics & contamination control. | Directly aligns with accreditation goals of data integrity. |
Table: Relevant Safety and Performance Standards [101]
| Standard / Outline | Applicable Equipment | Focus Area |
|---|---|---|
| UL 8802 | UV Germicidal Equipment & Systems | Overall safety and performance for commercial/healthcare settings. |
| IEC 62471 | Lamps and Lamp Systems | Photobiological safety, including UVC radiation risk assessment. |
| UL 8803 (Outline of Investigation) | Portable UV Germicidal Equipment | Safety for portable devices, often requiring critical control functions. |
Conclusion
UV irradiation remains a cornerstone of effective PCR contamination control, offering a non-chemical, reliable method to safeguard experimental integrity. Success hinges on a multi-layered strategy that combines the foundational understanding of UV mechanisms with rigorously applied protocols, proactive troubleshooting, and continuous validation. As molecular techniques evolve, future directions will likely involve the integration of smarter, sensor-based workstations and the refinement of UV-LED technologies for greater efficiency and precision. For biomedical and clinical research, mastering these decontamination principles is not merely a technical skill but a fundamental requirement for producing trustworthy, reproducible data that drives discovery and diagnostic accuracy.
References
- 1. All You Need Is Light. Photorepair of UV-Induced ... [https://www.mdpi.com/2073-4425/11/11/1304]
- 2. DNA interstrand crosslink repair and cancer - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC3560328/]
- 3. The effect of flanking bases on direct and triplet sensitized ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8096240/]
- 4. PCR workstation essential maintenance guide [https://bionordika.no/products/equipment/general-lab-equipment/pcr-workstations/pcr-workstation-essential-maintenance-guide]
- 5. Your Guide to PCR Workstations | Yamato USA [https://www.yamato-usa.com/blog/news-2/your-guide-to-pcr-workstations-28]
- 6. Three-dimensional visualization of ultraviolet-induced DNA ... [https://pubmed.ncbi.nlm.nih.gov/9457909/]
- 7. The Catalytic Mechanism of a Pyrimidine Dimer-specific ... [https://www.sciencedirect.com/science/article/pii/S0021925819873179]
- 8. A Review of Recent Evidence for Utilizing Ultraviolet ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8869636/]
- 9. Efficacy of UV-C 254 nm Light and a Sporicidal Surface ... [https://www.mdpi.com/2076-0817/13/11/965]
- 10. Effect of ultraviolet light treatment on microbiological safety ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9356256/]
- 11. Maximizing the disinfection effectiveness of 254 nm UV-C ... [https://www.frontiersin.org/journals/food-science-and-technology/articles/10.3389/frfst.2023.1223829/full]
- 12. Ultrafast inactivation of SARS-CoV-2 by 254-nm UV-C ... [https://www.nature.com/articles/s41598-023-39439-1]
- 13. Efficacy of UVC-LED radiation in bacterial, viral, and ... [https://www.sciencedirect.com/science/article/abs/pii/S0043135424012211]
- 14. PCR Workstation & UV Cabinet | Contamination Control [https://microlab-global.com/products/life-science/pcr-workstation/]
- 15. PCR Setup in Biosafety Cabinets: Contamination Control [https://qualia-bio.com/blog/pcr-setup-in-biosafety-cabinets-contamination-control/]
- 16. Design optimization and validation of UV-C illumination ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10909644/]
- 17. Efficacy of Ultraviolet-C Devices for the Disinfection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9681524/]
- 18. Troubleshooting your PCR [https://www.takarabio.com/learning-centers/pcr/faq/troubleshooting?srsltid=AfmBOorTjU1J8ygnHbeVvXIPH_UnggVwsCx2DgCfJOKo5QsVmhv2GIHq]
- 19. Could your PCR be affected by contamination? | IDT [https://www.idtdna.com/page/support-and-education/decoded-plus/could-your-pcr-be-affected-by-contamination/]
- 20. Ultraviolet dosage and decontamination efficacy were ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9578676/]
- 21. Control Contamination During PCR Amplification [https://www.airscience.com/controlling-contamination-during-pcr-amplification-preparation-best-practices]
- 22. PCR Setup—Six Critical Components to Consider [https://www.thermofisher.com/us/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/pcr-education/pcr-reagents-enzymes/pcr-component-considerations.html]
- 23. PCR Troubleshooting: Common Problems and Solutions [https://www.mybiosource.com/learn/pcr-troubleshooting-common-problems-and-solutions/]
- 24. Ultraviolet germicidal irradiation for surface cleaning of COVID ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11905184/]
- 25. PCR Troubleshooting Guide | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/pcr-education/pcr-reagents-enzymes/pcr-troubleshooting.html]
- 26. Guidelines for preventing and reporting contamination in ... [https://www.nature.com/articles/s41564-025-02035-2]
- 27. A Review of the Scientific Rigor, Reproducibility, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6959150/]
- 28. UVP PCR Workstations & Cabinets - uvp.com [https://www.uvp.com/products/lab-equipment/pcr-workstations-cabinets/uvp-pcr-workstations-cabinets/]
- 29. Ultraviolet germicidal irradiation: Advances in viral ... [https://www.sciencedirect.com/science/article/pii/S2950362025000281]
- 30. Impact of ultraviolet radiation on filtering facepiece ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11922867/]
- 31. Efficacy of ultraviolet-light emitting diodes in bacterial ... [https://link.springer.com/article/10.1007/s00203-025-04324-0]
- 32. PCR: The Right Way to Decontaminate and Eliminate ... [https://bitesizebio.com/3473/pcr-the-right-way-to-decontaminate-and-eliminate-false-positives/]
- 33. PCR Cabinets: From Installation to Maintenance [https://www.escolifesciences.com/news/from-installation-to-maintenance-a-complete-guide-to-pcr-cabinets]
- 34. How to prevent PCR contamination [https://www.minipcr.com/how-to-prevent-pcr-contamination/]
- 35. PCR Workstation vs. Biosafety Cabinet: Understanding the ... [https://www.airscience.com/pcr-workstation-vs-biosafety-cabinet-understanding-the-differences]
- 36. How Does a PCR Workstation Improve Lab Accuracy? [https://aeromech.in/how-does-a-pcr-workstation-improve-lab-accuracy/]
- 37. 10 Handy Tips to Help Keep Your PCRs Contamination Free [https://bitesizebio.com/23625/10-handy-tips-to-help-keep-your-pcrs-contamination-free/]
- 38. PCR Workstation: Everything You Should Know [https://www.igenels.com/pcr-workstation-everything-you-should-know/]
- 39. Efficacy of ultraviolet-light emitting diodes in bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12031847/]
- 40. Impact of UV-C Irradiation on Bacterial Disinfection in a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9895992/]
- 41. Ultraviolet Radiation - an overview [https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/ultraviolet-radiation]
- 42. Ultraviolet germicidal irradiation [https://en.wikipedia.org/wiki/Ultraviolet_germicidal_irradiation]
- 43. Protocol for Evaluating the Microbial Inactivation of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9332507/]
- 44. UVP PCR Workstations & Cabinets [https://www.analytik-jena.us/products/life-science/pcr-workstations-cabinets/uvp-pcr-workstations-cabinets/]
- 45. Sun Safety [https://www.hopkinsmedicine.org/health/wellness-and-prevention/sun-safety]
- 46. Polymerase Chain Reaction (PCR) - StatPearls - NCBI - NIH [https://www.ncbi.nlm.nih.gov/books/NBK589663/]
- 47. About Germicidal Ultraviolet (GUV) | Ventilation [https://www.cdc.gov/niosh/ventilation/germicidal-ultraviolet/index.html]
- 48. Impact of ultraviolet radiation on filtering facepiece ... [https://www.frontiersin.org/journals/public-health/articles/10.3389/fpubh.2025.1537742/full]
- 49. Construction and validation of an ultraviolet germicidal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8301618/]
- 50. UV cabinet/PCR workstation, steel work surface [https://www.azerscientific.com/UV-cabinet-PCR-workstation-s.-steel-work-surface-glass-sides-front-dual-UV-protection-lamp-and-UV-re]
- 51. 24" PCRPREP PCR Workstation with Class 100 Vertical ... [https://www.scilogex.com/shop/sci-pcr24-24-pcrprep-pcr-workstation-with-class-100-vertical-laminar-flow-air-and-timed-uv-light-110v-5549?srsltid=AfmBOoqoFJPwG7syUeYjLGDl-_6qVy-Y2fh_HzQGKhhQavTKkqiFxKgM]
- 52. Frequently Asked Questions: Ultraviolet Disinfection System [https://info.xtralight.com/faq-ultraviolet-disinfection-system]
- 53. Essential Maintenance Guide for Your PCR Cabinet [https://www.nordicbiolabs.se/kunskapsbank/why-is-it-important-to-care-for-your-pcr-cabinet-essential-maintenance-guide_1]
- 54. Is the combination of UV-C light and bleach less effective than ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10724481/]
- 55. Synergistic effects of ethanol and UV radiation to reduce ... [https://pubmed.ncbi.nlm.nih.gov/20202345/]
- 56. UV Disinfection vs Chemical Cleaners [https://www.environments.tech/post/uv-disinfection-vs-chemical-cleaners]
- 57. Chemical Cleaning and UV Disinfection: What to Know [https://rzero.com/blog/cleaning-and-disinfecting-with-chemicals-what-to-know/]
- 58. Troubleshooting Common Issues with UV Disinfection ... [https://www.linkedin.com/pulse/troubleshooting-common-issues-uv-disinfection-rl40c]
- 59. UVP PCR Workstations & Cabinets [https://www.analytik-jena.us/products/lab-equipment/pcr-workstations-cabinets/uvp-pcr-workstations-cabinets/]
- 60. 6 Ways to Minimize Contamination during PCR [https://www.labclinics.com/2019/07/22/six-ways-to-minimize-contamination-during-pcr/?lang=en]
- 61. Aseptic Technique - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/medicine-and-dentistry/aseptic-technique]
- 62. PCR Cabinets [https://www.escolifesciences.com/products/pcr-cabinet]
- 63. Biosafety Cabinet UV Light White Paper [https://www.nuaire.com/en/resources/biosafety-cabinet-uv-light]
- 64. Laminar Flow Hoods for Cell Culture [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-equipment/laminar-flow-hood.html]
- 65. PCR controls [https://www.qiagen.com/us/knowledge-and-support/knowledge-hub/bench-guide/pcr/reference-genes-and-controls/pcr-controls]
- 66. Amplification of the No Template Control (NTC) [https://www.thermofisher.com/us/en/home/life-science/pcr/real-time-pcr/real-time-pcr-learning-center/real-time-pcr-basics/real-time-pcr-troubleshooting-tool/gene-expression-quantitation-troubleshooting/amplification-no-template-control.html]
- 67. Preventing PCR Amplification Carryover Contamination in ... [https://www.annclinlabsci.org/content/34/4/389.long]
- 68. Avoiding false positives with PCR | IDT [https://www.idtdna.com/page/support-and-education/decoded-plus/how-to-avoid-false-positives-in-pcr-and-what-to-do-if-you-get-them/]
- 69. Dos and don'ts for molecular testing [https://www.who.int/teams/global-malaria-programme/case-management/diagnosis/nucleic-acid-amplification-based-diagnostics/dos-and-don-ts-for-molecular-testing]
- 70. UVC/T-AR, PCR cabinet [https://biosan.lv/products/uvct-ar-display/]
- 71. Eliminating PCR contamination: is UV irradiation the answer? [https://pubmed.ncbi.nlm.nih.gov/1664435/]
- 72. Removal of Contaminant DNA by Combined UV-EMA ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4501569/]
- 73. Top Tips for UV Crosslinking Polymer Processes in 2025 [https://ebeammachine.com/top-tips-for-uv-crosslinking-polymer-processes-in-2025/]
- 74. Tips for successful PCR and Troubleshooting PCR-qPCR [https://info.abmgood.com/polymerase-chain-reaction-pcr-qpcr-troubleshooting]
- 75. UV cabinet/PCR workstation, steel work surface [https://test.azerscientific.com/UV-cabinet-PCR-workstation-s.-steel-work-surface-glass-sides-front-dual-UV-protection-lamp-and-UV-re]
- 76. UV Lamp Troubleshooting [https://www.alpha-cure.com/knowledge-centre/uv-lamp-troubleshooting]
- 77. How Long Do UV Lights Last? | Guide [https://bscbulbs.com/blogs/bsc-blog/how-long-do-uv-lights-last-guide?srsltid=AfmBOoomZm_gL6WjduHF6UmGVTTKT3VaaCPDxzepElesr_FELI2byIlg]
- 78. When to Replace a UV Light in Your Air Conditioning System [https://www.acwarehouse.com/when-to-replace-a-uv-light-in-your-air-conditioning-system/]
- 79. CleanSpot PCR Workstations [https://coylab.com/coy-products/cleanspot-pcr-workstations/]
- 80. How To Maintain A UV Water Filter System [https://espwaterproducts.com/pages/uv-technical-help?srsltid=AfmBOor9WhO_ik3RdKIKh8pHH_DcBokP_UZHgu2qj7qPbqEp9boO3_Q3]
- 81. Reliability of PCR decontamination systems [https://pubmed.ncbi.nlm.nih.gov/7580883/]
- 82. Maintenance Tips for Prolonging the Life of Your UV Lamps [https://www.alpha-cure.com/news-article/maintenance-tips-for-prolonging-the-life-of-your-uv-lamps]
- 83. The Devil in the Details: Contamination in a PCR Lab [https://www.rapidmicrobiology.com/news/the-devil-in-the-details-contamination-in-a-pcr-lab]
- 84. What is UNG/UDG? [https://www.thermofisher.com/us/en/home/life-science/pcr/real-time-pcr/real-time-pcr-learning-center/real-time-pcr-basics/what-is-ung-udg.html]
- 85. Minimizing PCR Cross-Contamination using GoTaq® DNA ... [https://www.promega.com/resources/pubhub/enotes/minimizing-pcr-crosscontamination-using-gotaq-dna-polymerase-with-dutp-and-ung/]
- 86. AirClean Systems FAQ [https://aircleansystems.com/faq]
- 87. Troubleshooting your PCR [https://www.takarabio.com/learning-centers/pcr/faq/troubleshooting?srsltid=AfmBOop50HgfCdfXeGagDyQopMW8Bh8VyrQ8vmHoS4bbtWbUxOFLE3oI]
- 88. Top 4 UV Curing Challenges and Solutions for Manufacturers [https://intellego-technologies.com/top-4-uv-curing-challenges-and-solutions-for-manufacturers/]
- 89. Simultaneous detection of respiratory virus RNA on ... [https://www.nature.com/articles/s41598-025-22296-5]
- 90. UV Production & UV Curing System FAQ's [https://www.doctoruv.com/uv-maintenance]
- 91. Evaluation of Different Cleaning Strategies for Removal ... [https://www.mdpi.com/2073-4425/13/1/162]
- 92. Ultraviolet dosage and decontamination efficacy were ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2022.875817/full]
- 93. Comparative effectiveness of rapid-cycle ultraviolet ... [https://pubmed.ncbi.nlm.nih.gov/30987794/]
- 94. Is a PCR workstation the same as a biological safety cabinet? [https://www.labcleantech.com/knowledge/is-a-pcr-workstation-the-same-as-a-biological-safety-cabinet]
- 95. Common Biosafety Cabinet Issues and Solutions - QUALIA [https://qualia-bio.com/blog/common-biosafety-cabinet-issues-and-solutions/]
- 96. Troubleshooting Common BSC Problems and Errors [https://www.linkedin.com/advice/0/how-do-you-troubleshoot-common-bsc-problems-errors]
- 97. Microbiological evaluation of ultraviolet C light-emitting ... [https://www.sciencedirect.com/science/article/pii/S2405844024133129]
- 98. Microbiology testing laboratory achieves NATA ... [https://www.unsw.edu.au/news/2025/09/microbiology-testing-laboratory-achieves-nata-accreditation-for-uv-disinfectant-testing]
- 99. UV PCR Workstation Market Expansion Strategies [https://www.datainsightsmarket.com/reports/uv-pcr-workstation-1522352]
- 100. UV Disinfection System Maintenance: Pro Tips & Tricks [https://waterestore.com/blogs/news/uv-disinfection-system-maintenance-pro-tips-tricks?srsltid=AfmBOoogoiWGdxukvIeCZ20qdMxKOdYQW2zC-gKfZdch1YbGUDLkaT_O]
- 101. UVC Testing, Certification and Safety Services [https://www.ul.com/services/ultraviolet-uvc-light-testing-and-certification]
- 102. 生物技术自动化带大家了解实时荧光定量PCR仪的维护及注意 ... [https://www.jxjrsd.com/news/29.html]